Advances in dairy Research
Open Access
ISSN: 2329-888X
ISSN: 2329-888X
Research Article - (2019)Volume 7, Issue 3
The objective of this study was to compare the fatty acid profile of caprine milk or cheese from animals raised under extensive, semi-extensive or full confinement production system in México. Feeding systems particularly grazing have been shown to affect essential fatty acid profiles, particularly the ω6/ω3 balance. Utilization of concentrates probably raises the omega 6 contents, or decreases omega 3 concentration, often exceeding 4:1 ω6/ω3 ratio, diminishing beneficial effects of ω3 regarding consumer health. The present study was conducted for two years (2016 and 2017) on 5,079 lactating goats from the states of Querétaro, Guanajuato, Colima, San Luis Potosí and Michoacán in México. Average milk production was 1.5/d liters (DS ± 0.9). Of the 98 farms studied, 35% were in full confinement, feeding basically alfalfa hay and concentrate (18% CP), 22% grazed rye grass, 17% pastured alfalfa (45% of these grazing systems were supplemented), finally 26% grazed were in silvopastoral surroundings, mainly without supplement. The use of concentrate in more than 35% of total feed consumption had an effect on the profile of essential fatty acids exceeding ω6/ω3 4/1 ratio. In 36 of the studied farms levels of ω6/ω3 exceeded 4:1, all of them were supplemented with 700 g /d or more concentrate. In those units the use of the supplement decreased ω3 or increased ω6.
Goat milk cheese; Indoor feeding; Grazing; Poly-Unsaturated Fatty Acids (PUFA); Omega fatty acids
In recent years, the consumers' demand for high quality food in relation to its nutritional value and sensory characteristics has increased [1]. Animal diets can affect the quality of farm animal products [2]. It has been documented that pasture-grazed sheep and goats provide healthier milk by enhancing its composition in the desirable bioactive fatty acids, CLA cis-9, trans-11, n-3 fatty acids [3] and in volatile aromatic compounds such as terpenes [4]. Previous studies demonstrated the fatty acid composition of the milk used for the production of dairy products, affects directly their quality characteristics and subsequent consumer acceptance [5].
The two major systems of goat farming are pasture and indoor systems. Between these two managements there are wide scopes of mixed systems such as summer pasture/winter indoors or alternatively indoors/outdoors, subject to climatic differences or forages viability [6]. People consider more often pasture systems as more extensive than indoor systems [7]. However, level of intensification varies in grazing systems based on cultivated pasture versus goats fed on poor rangelands [8]. The intensification level in indoor systems is tightly linked with the nutritive value of fodders, as well as the quantity of distributed concentrates [4]. As previously discussed despite the difficulties due to the multiple types of systems, we can compare results in regards of indoor versus outdoor farming systems [4].
At present there is a growing interest in animal food products obtained from grazing and is recognized by nearly all consumers and farmers themselves as high-quality foods [9]. Dairy specialization has brought about an increase in the use of concentrates [10], thus reducing or even eliminating pasture as a feed source. Despite the goat farming intensification process, there are still areas where farms depend on pastures at least partially, to feed their animals. However, for this production model to be sustainable, apart from being economically viable, the quality of their products should also be analyzed and characterized to increase market value [11].
Recently it has been proven in the literature that a lower content of Saturated Fatty Acids (SFA) favors human health, due to its role in different diseases [12]. The effect of the presence of ω´s 3, metabolized by elongation of conjugated linoleic acid (ACL), particularly the role of arachidonic acid and Docosa Hexaenoic Acid (DHA) in the reduction of oxidative stress due to the presence of reactive oxygen substances, which is characterized by a decrease in the capacity of the endogenous system to act against oxidative attack directed to biomolecules, associated with different severe pathologies, such as cancer, cardiovascular diseases, type 2 diabetes, hypertension, and neurodegenerative diseases has been well documented [12].
In México previous studies have demonstrated the importance of grazing in the quality of goat milk, particularly oxidative compounds and phenols [9,13]. These investigations reported a total range of phenols from 780 mg/l in milk of the goats that graze against 50 mg/l to milk from full confinement, and also discussing pasteurization that could diminish almost half those contents [9]. Pasturing has been very significant. The phenols range from a minimum of 49 mg up to a maximum of 150 mg, which is sufficient to justify the influence of phenols on taste [6]. Cabiddu [2] report that total phenols exceeded 67 mg/l in milk from goats fed indoors, compared to 469 mg/l if the goats were pasturing. Chavez- Servin [9] in semi-arid Mexico have found that pasteurization of milk decreased phenol contents and that grazing, compared to the full confinement system, caused a significant increase in phenol content. Fatty acid, volatile and sensory profiles from milk and cheese raised on native semiarid pasture or confinement have demonstrated significant differences also in Brazil [14].
Several previous studies on the nutritional quality of milk in Mexico demonstrated benefits of grazing in cows, goats and sheep [15]. A low content of Saturated Fatty Acids (SFA) and high in Poly-Unsaturated Fatty Acids (PUFA) was observed in milk from grazing animals, contrasted with a significantly higher presence of SFA and lower PUFA in milk from animals in full confinement [9,15].
Food, in general, and in particular free grazing in a silvopastoral environment, allows each goat as an individual, to compose a diet according to their own needs, which have a positive effect on the nutritional characteristics of milk [15,16]. It has been demonstrated that milk from grazing livestock could have a positive impact on human health, producing milk or cheese of better nutritional quality for the consumer, due to the balanced presence of ω6/ ω3 [17].
Previous work demonstrated the importance of ω6/ω3 relationship Simopoulos [18,19] suggesting a 4:1 ratio to prevent cardiovascular diseases with a 70% reduction in mortality. Recently the importance of maintaining a ratio of ω6/ω3 not higher than 4:1 was documented and in modern diets the contribution of ω3 decreased and the consumption of ω6 has increased to levels higher than 10:1 [20,21]. The Poly-Unsaturated Fatty Acids (PUFA) consumed in the diet is absorbed by the intestinal cells and subsequently give rise to several metabolites. On the one hand, by elongation and desaturation, PUFAs are transformed into long chain PUFAS. By retroconversion, the number of carbon in the chain is increased by 2 in derivatives of linoleic acid (AL) and conjugated linoleic acid (ACL). On the other hand, cyclooxygenases and lipoxygenases generate different protonoids of Eicosa Pentaenoic Acid (EPA), Arachidonic Acid (AA) and Docosahexaenoic Acid (DHA) in the formation of Prosta Glandins (PG), Leukotrienes (LT) and Thrombokinase (TX), all of them involved in anti-oxidation and anti-inflammatory processes, which prevent coronary heart disease, hypertension, diabetes, arthritis and cancer, in addition to their role in the control of other inflammations and autoimmune conditions [20].
Goats adapt well to environmental conditions in semiarid lands, being able to obtain adequate diets, even when forage are scarce and they can feed over rough, and otherwise inaccessible terrain. Mexican range land is dominated by different vegetation species, which usually contain metabolites with certain benefits for human health, including alkaloids, glycosides, fatty acids, terpenes, phenols, saponins, tannins, and flavonoids [8,13,15,16]. The vegetation, consumed by goats during grazing, changes its chemical and nutritive composition seasonally, which gives cheese a different nutritive profile and flavor through the year [22,23]. Components with nutritional and beneficial health interest were found in higher concentrations in cheese made from grazing, compared to an indoor feeding system, regardless of animal species [7,8,24]. Nevertheless very few studies have detailed the nutritional qualities of grazing goat’s cheese with or without temperature treatment over a length of time [16].
Goats grazing on semi-arid brush land had more than twenty different eatable plants [25]. Beneficial vegetation metabolites to human health found in milk, has also been reported from analyses of different cheeses, including amines and alkaloids, cyanogenic glycosides, cyclitols, fatty acids, fluoroacetate, gums, non-protein amino acids, terpenes, tannins, and flavonoids [13,26]. In Mexico, there has been a long tradition of livestock grazing due to extensive pastoral areas [27].
Cheese and milk products from grazing animals have become known for their superior taste and quality compared to dairy products from indoor fed animals, cattle or goats [8,16]. This knowledge could help the economical and sociological development of small farm enterprises. Utilization of fibrous diets, roughages and brush land through grazing by ruminants can be manipulated in various ways. The use of the local resources provides economic feasibility to the dairy goat industry [8].
Recent work discussed the quality of sheep’s cheeses manufactured from grazing or indoor animals [16]. However, it is not well known whether this pastoral system affects cheese composition in different grazing systems. Therefore, the present study compares the effect of grazing and supplementation as compared to indoor feeding with alfalfa hay and concentrate, on the nutritional composition of artisan goat’s cheeses, made from raw or pasteurized milk.
The objective of this study was to evaluate the effect of grazing on the presence of essential fatty acids (PUFA) and the balance of ω6/ ω3 of grazing animals in 99 goat herds in Mexico during 2 years.
Experimental design and treatments
The experiment was conducted with a total of 5,079 goats from 98 goat farms in each unit from random two samples in Guanajuato, Michoacán, Jalisco, Querétaro and Colima. The goats were in second and third lactation. The study was conducted over two years, 2016 and 2017, with three different systems: grazing, silvopastoral and full confinement. The grazing group was fed Ryegrass (Lolium perenne), or Alfalfa (Medicago sativa), with or without concentrate supplementation of 16% CP. The grasses in the silvopastoral system were: Bouteloia curtipendula, Chloris virgata, Bothriochloa saccharoides, Leptochloa saccharoides, Leptochloa dubia, Rhyncheltythurum roseum, Panicum obtusum, Bouteloia repens, Aristida adscensionis, Setaria parviflora, Urochloa fasciculata.
Legumes: Prosopis laevigata, Acacia farnesiana, Acacia schaffneri, Mimosa biuncifera.
Scrubs: Celtis pallida, Jatropha dioica, Zalazania augusta, Verbasina serrata, Opuntia spp., Lolium perennial, Prosopis sp., Opuntia sp with or without 16% PC concentrate. While goats in full confinement indoors during the study, were fed 2.5 kg DM, and sub-divided into 1 kg (16% of CP and 2.5 Mcal/kg) grain supplement and 1.5 kg alfalfa hay/d.
Food sampling and analysis
Randomly collected 2 samples of milk or goat cheese. Also representative of each farm, simultaneously determined the mass of the grass before entering each meadow by cutting 5 squares (0.25 m/quadrant) of pasture 3 cm above the ground. The samples were grouped and used for chemical composition analyses. The samples were dried at 60°C and analyzed by the Van-Soest method to determine the content of dry matter, mineral, crude fiber, crude protein, fat and nitrogen-free extract.
Fatty acid analyses
Lyophilised samples of frozen goats milk were extracted by Soxhlet with petroleum ether/ethyl ether (1:1 v/v) to characterize the fatty acid composition. Fatty acid methyl esters were separated by gas chromatograph equipped with a flame ionization detector and split (1:24) injector. Separations were performed using a capillary column (25 cm × 0.2 mm i.d. × 0.3 um film thickness). The content of fatty acids was expressed in percentage. After determination, the sum of saturated fatty acids, mono-unsaturated, polyunsaturated, as well as the content of omega fatty acids were calculated using AOAC method 923.07 [27].
Fatty Acids Methyl Esters (FAME)
Lipids were dissolved in hexane and sodium hydroxide methanol solution for saponification, the trans-esterification of cheese fat to methyl-esters was carried out according to official method of AOAC 696.33 [27] using Boron Trifluoride (BF3). FAME was quantified by gas chromatography (GC) using a CP-3380 chromatograph equipped with a split injector, Flame Ionization Detector (FID) and auto sampler CP 8400. A DB23 column (30 m × 0.25 mm i.d.) with a film thickness of 0.25 μm was employed. Nitrogen was used as carrier gas at a flow rate of 30 ml/min. Temperatures column was held for 1 min at 120°C, then programmed at rate of 10°C/min to 200°C and held for 5°C/min to final temperature of 230°C; temperature injector and FID were 250°C and 300°C, respectively. Integration for each fatty acid was performed by a Varian Star Chromatography Workstation Software. Identification of the peaks was made on the basic of the retention times of standard methyl esters of individual fatty acid (FAME mix C4-C24 #18919-1 AMP). The final concentration of FAME was expressed as mg/100 g of cheese on fresh base.
The total milk production and the milk composition were analyzed using a one-way ANOVA design. Data analysis was carried out using the General Linear Model Procedures (Statgraphics- Centurion), calculated with Statistical Analysis System [28].
Diet and botanical composition of intake is shown in Table 1. Approximately 71% of the DMI was provided by grazing; 15% concentrate and 14% from browsing. In the confined system (CS) diets 20% of the DMI was provided by commercial concentrate and 80% by alfalfa hay (Medicago sativa).
| Diets | |||||
|---|---|---|---|---|---|
| Variables | SP | SP plus | FC | G | G plus |
| Dry matter (%) | 90 | 91.7 | 75.2 | 34.1 | 40 |
| Nitrogen (%) | 18.21 | 16.25 | 14.64 | 13.42 | 12.86 |
| Organic matter (%) | 87.5 | 92 | 94.1 | 78.8 | 87.5 |
| NDF | 73.1 | 67.8 | 71.8 | 17.8 | 73.1 |
| ADF | 42.4 | 45.2 | 43.2 | 13.5 | 42.4 |
| ME (Mcal/kg DM) | 2.15 | 2.23 | 2.52 | 2.1 | 2.24 |
SP: Silvopastoral; SP plus: Silvopastoral with Concentrate; FC: Full Confinement; G: Grazing Alfalfa or Rye Gras; G plus: Grazing Alfalfa or Rye Gras with Supplementation; NDF: Neutral Detergent Fibre; ADF: Acid Detergent Fibre; ME: Metabolizable Energy
Table 1: Herbal composition value of mixtures of forages alfalfa or rye grass, full confinement with concentrate and pasturing silvo-pastoral areas, with or without supplementation.
Crude protein was higher when goat’s diets included shrub rangeland particularly in arboreal legumes (Prosopis laevigata, Acacia farnesiana, Acacia schaffneri) which supplied large amounts of nitrogen, being comparable similar to animals fed in full confinement, due to alfalfa protein (Medicago sativa) of and 18% protein from the commercial concentrate, being slightly higher to the nitrogen content of alfalfa or rye grass, differences in fatty acid profile, could be explained on the nutrients mixtures studied, that were demonstrated given significant differences in nitrogen free extract as shown in Table 1.
A total of 196 milk samples were taken, two for each farm, goat farms had on average 52 milking females, (± 22), with a maximum of 120 goats and a minimum of 18 per farm; Milk production per herd was 1.5 l/d, (± 0.820) higher farms produced 264 l/d and small enterprises 12.5 l/d. Milk production per goat was 1.5 l/d liters (± 0.820), maximum performance 3.7 l/d minimum production 0.700 l/d. From the 98 farms studied, 35% supplemented their diet with the administration of concentrate during milking, 800 g/d (± 100) and 5 more than 1.2 kg/d, finally 26% did not provide concentrate (Table 2).
| Variables | Number of ranches | Number of animals | Total milk production per day | Daily milk production per goat |
|---|---|---|---|---|
| Silvopastoral | 24 | 1110 (± 35) | 41.4 (± 16) | 0.8 (± 0.2) |
| Silvopastoral plus | 3 | 126 (± 14) | 46.13 (± 13) | 1.00 (± 0.4) |
| Full confinement | 33 | 1562 (± 47) | 125.58 (± 27) | 2.7 (± 0.9) |
| Alfalfa or rye grass | 8 | 473 (± 18) | 58.91 (± 14) | 0.97 (± 0.2) |
| Grazing plus | 31 | 1808 (± 75) | 53.67 (± 17) | 0.91 (± 0.3) |
Table 2: Farms in silvopastoral with or without supplementation, full confinement, or grazing alfalfa or rye grass with or without supplementation.
The saturated fatty acids in grazing showed no significant difference between the two years, however, the saturated fatty acids in animals grazing plus (supplemented with concentrate) showed a difference between years, being higher in the year 2017. Differences on grazed forages due to environment were recorded (mainly rain precipitation). The results of fatty acids in full confinement did not show differences between years, for which the balance was not significant as summarized in Figure 1.
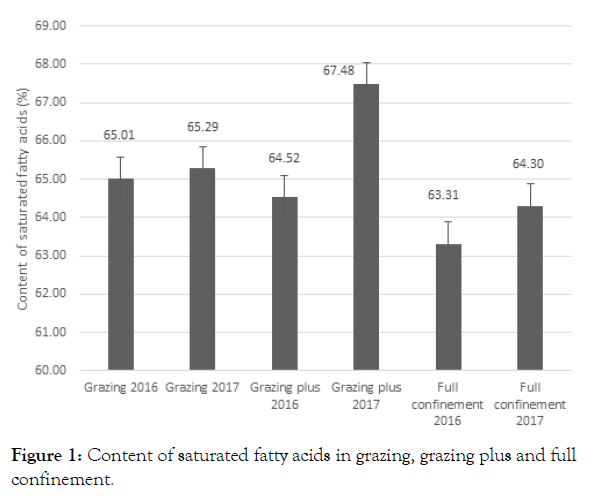
Figure 1: Content of saturated fatty acids in grazing, grazing plus and full confinement.
Grazing animals with moderate supplementation (less than 300 g/d) showed an increase of saturated fatty acids, where short chain fatty acids (caproic, caprylic, capric fatty acids) were prominent (P<.01). At the same time the animals that grazed or were in full confinement slightly increased (P<.05) from one year to another. At the same time arachidonic acid was present in all groups in low concentration without significant differences (P>0.5).
The animals in grazing plus in 2017 showed a reduction in the ratio of non-saturated fatty acids, these include omega 3 and 6, which showed omega concentration and therefore a reduction in the ratio, that allowed acid nutraceuticals to be obtained with greater availability. The animals in full confinement increased non-saturated fatty acid concentration, increasing the ratio of omega fatty acids as shown in Figure 2 particularly omega 6.
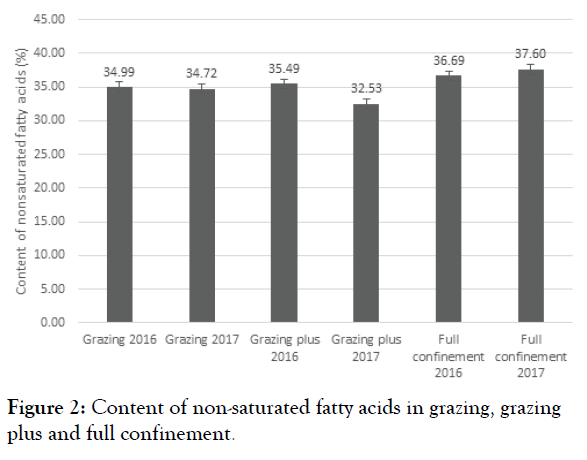
Figure 2: Content of non-saturated fatty acids in grazing, grazing plus and full confinement.
Grazing systems showed a significant difference between one year to another (P<0.5), due to the amount of nonsaturated fatty acids present, however, the omega fatty acids of the animals in silvopastoral increased from one year to the next due to the augmentation in linoleic acid demonstrated by high omega 3 content. These differences were of importance, even though that in the diet there was an increase in forage fat, showing rises in the amount of saturated fatty acids in the milk, without exceeding the maximum allowed ratio of omega 6/omega 3 of 4:1, unlike the animals in full confinement where ratio exceeded 5.3:1 in 2016 preventing classifying this milk as a nutraceutical food (Table 3).
| Variables | Grazing (2016) | Grazing (2017) | Silvopastoral (2016) | Silvopastoral (2017) | Full confinement (2016) | Full confinement (2017) |
|---|---|---|---|---|---|---|
| Linoleic ω6 | 2.51a | 2.0b | 2.48a | 2.42a | 1.98c | 2.02c |
| α Linoleic ω3 | 0.97a | 0.88a | 0.88a | 0.74b | 0.37c | 0.6b |
| Relation ω6/ω3 | 2.58a | 2.2b | 2.8a | 3.2b | 5.3c | 3.3b |
Different superscripted letters show statistical differences between groups (P<0.5).
Table 3: Relationship of omega 3 and 6 in animals in grazing, grazing plus and full confinement.
There was a huge variation in the number of animals in each herd, which averaged 52 goats (± 22), with one farmer having 254 animals and the smallest with 17, with an average of milk production per herd of 52 liters per day, (± 17), a maximum of 264 liters in one of the farms. The number of milking animals was also significantly different among farms with an average of 24 goats (± 14), the farmer who milked the largest number of animals had 90, while the smallest goat farm milked 5 animals. Chemical composition and nutritive value was reported in Table 1. The grass dry matter was 91.2% for animals with silvopastoral management; animals in grazing had 80% and feedlot had 95% DM.
The results summarized in Table 2 show the prevalence of palmitic (c16: 0), oleic (C18: 1), myristic (C14: 0), stearic (C18: 0) and caprylic (C10: 0) fatty acids. Content of short chain fatty acids (C4: 9-C10: 0) was similar for all treatments (Table 3). Amount of medium chain fatty acids was significantly higher in silvopastoril management.
Palmitic acid was significantly higher for the silvopastoral group (P<0.5), stearic was significantly lower in silvopastoral 2017 than in grazing and stabling (P<0.1), while oleic and linoleic acids were similar for all groups except for full confinement. The linoleic content was significantly higher for the grazing and silvopastoral groups compared to indoor (P<0.1). The content of CLA was significantly higher for silvopastoral grazing than for stabling (P<0.5). Feeding based on grazing and silvopastoral, feed rich in Poly-Unsaturated Fatty Acids resulted in a significant increase in this class of fatty acids to the detriment of the saturated class (P<0.5).
Despite these huge variations of grazing animals, the 26 units had an amount of ω3 higher than what was necessary to be considered as Latte Nobile (Table 3) quantitatively, but when providing concentrates 32 of them exceeded the 4: 1 ω6/ω3 ratio, indicated as a limit by previous literature to cease beneficial nutritional effect in preventing chronic degenerative diseases. In those 32 units that were offered concentrates the proportion of ω6/ω3 was increased exceeding the limit of 4: 1 while when there was not offered concentrates, it kept it at 3: 1 ω6/ω3. The average milk production was 0.9 kg/d, (± 0.3) for the farms that did not give concentrates, while it increased significantly by 2.6 kg/d (± 0.3) in the farms that offered concentrates. Results showed concentrates increased milk production but decreased milk quality.
The proportion increases to 4.5: 1 ω6/ω3 in the 37 farms that offered more than 700 g/d of concentrates, which indicates that the low or moderate use of the concentrates does not have an effect on the proportion of omega 6/omega 3, while a high utilization unbalanced the ratio of omegas of more than 4: 1 ω6/ω3.
The beneficial effect of ω3 polyunsaturated acids and its relation with ω6 in grazing systems has been abundantly documented, and its beneficial effect in relation to human health [21,24]. Particularly the role of omega 3 and 6 fatty acids as antioxidants in the biochemistry of oxidative stress [12]. Recent studies have shown the importance of maintaining a ratio of less than 4: 1 between ω6/ω3, since higher concentrations modify the beneficial effects of ω3, either as ALC (18 carbons) or particularly when elongated towards arachidonic acid (20 carbons) and docosahexaenoic acid of 22 carbons (DHA) 22: 6 decreasing the protective effects on health [20,21]. In 61 of the 98 units studied they had a ratio less than 4.1 ω6/ω3; all in pasture. All of them had values higher than 40 mg/l of ω3, but in 5 of them the values of ω3 for the ratio of ω6 would not have a protective effect on consumer health. Units higher than the 4: 1 ratio had 38 to 47 mg/lω3. Concentrate supplementation apparently was the variable for the increase of the ω6/ω3 ratio greater than 4: 1. These values coincide with those reported in the literature as quantities with beneficial effects on health and the effects of an increase in the ω6/ω3 ratio [18,19,21,24].
Recently Sant-Ana [14] have compared two feeding systems full confinement or grazing, resulted in distinct FA profile of milk and cheese produced from goats with access to pasture presenting higher PUFAs and MUFAs and lower SFAs resulting in a clear reduction of its atherogenic index, thus presumably more beneficial for human health. In their work both milk and cheese from the grazing systems are better accepted by the tasters, similar results were documented in the present observation probably due to grazing.
Even in spite of the differences that were shown among the ranches studied, the LAC values were within the parameters suggested as beneficial for health for the systems [24]. On the other hand, the ω6/ω3 ratio was higher in 37 of the studied units, which probably means a low use of ω3, which in turn were the ones with the lowest volume of ALC with concentrate use [17-29].
Soryal [30], measured protein and fatty acids in soft goat cheese comparing different feeding systems in full confinement and grazing with five different forages (wheat, sesame, sudan grass, native grass and clover) supplemented with concentrate mixed or exclusive diet. They found a protein concentration between 15.0% and 14.0% from each diet recording 14.0% to 14.5% FA, without differences among diets. They concluded that diet did not have significant effects on cheese quality. On the contrary, in the present observations, grazed raw (GR) had higher protein content 16.6% with statistical differences. Diet adding no heat treatment, impacted total protein content, even if there were not statistical differences but alfalfa hay plus concentrate showed the lowest value. However it has been proved that semi-arid forages are high in protein and utilization of non-protein supplementation with urea increased microbial protein [15]. Previous work by Park [31,32], recorded values of 20.1% fat and 18.9% protein, both being higher than those observed in the present study. High protein concentration is probably due to 50% or more concentrate offered to high producing milking goats. Management, supplementation and forage variety could explain differences, but those studies showed that diet had a direct impact in cheese quality and protein content. Ramanzini [33] and Soryal et al. [30] proved that feeding system modified cheese fat content when great amounts of concentrate (45% or more) were offered in the diet. Present results did not show this increase, probably, due to a comparatively lesser amount of concentrate supplementation (20%) in indoor feeding and no concentrate in with SIUS in grazing, in the latter probably by the large amount of microbial digestible protein from urea formed [34].
Fat content of goat products could be relatively high as demonstrated by Andrikopoulus [35] with a fat: protein ratio over 1.5, as is the case of the Greek soft cheese, specially Manouri with a relation of 2.5 above, or Feta and Teleme soft goat cheeses with a 1.29-1.68 ratio. Our observations recorded a relation of 0.82 and 0.92, being a low fat product in all observations, particularly GR, showing the highest relationship to management compared to process. Low fat cheeses have an increasing market because of health related issues, and nutritional management has a major impact in fat production [17].
Average amino acids 12.55 g/100 g in the present observation were inferior (17.7 g/100 g) to those reported by Park [32]. However present values were higher compared with the work of Gambelli [26] and Franco et al. [36] with 8.75 and 9.03 g/100 g, respectively. Variances of values could be due to measurement techniques in different studies, management diets or cheese processing methods.
Cholesterol in goat soft cheese varies as demonstrated by Park [31], who analyzed 15 varieties of soft goat cheese, showing results from 81, 125 and 146 mg/100 for each cheese. Other values reported by Park [31] with colorimeter had 121 mg/100 g of cholesterol, variation of techniques add difficulties to compare cheeses, suggesting colorimeter is not the best technique to analyze cholesterol. Standardizing Gas Chromatography (GC) was employed in the present study, with results of 80 and 90 mg/100 g of fresh product, demonstrating low cholesterol values for Mexican soft cheese, similar to Emmental cheese (89 mg/100 g) values reported by Ulberth and Reich [37], however the lowest cholesterol recorded was in row grazing, probably due to the extensive use of grazing and no concentrate in the diet.
On the other hand, Chin [38] reported higher values for soft goat cheese up to 110 mg/100 g. In other studies in Cheddar cheese, as discussed by Hamill and Soliman [39], and Stewart [40] results showed lower values of 71 and 77 mg/100 g but no information of nutritional management was discussed. Andrikopoulus [35] presented values of 68 mg/100 g of cholesterol in Feta cheese. Manufacturing process could explain differences in cholesterol values among soft cheese. In either case, Mexican soft cheese was low in cholesterol particularly from grazing goats differences could be explained by the diet and management [17].
Park [32], Jensen [41], Chapkin [42] and Banskalieva [43] discussed that worldwide, fat and cholesterol have increased in man’s diet, being a health risk due to the coronary and vascular problems. The OMS on the other hand suggest a maximum of 300 mg/cholesterol per day, suggesting that consumption of soft goat cheese is not a health risk as has been well documented for smoking, hypertension and obesity [44]. Data in the present observation confirmed the low cholesterol in Mexican cheeses.
Literature reviews are mixed in opinion, Carpino [45] observations did not show differences in total FA while the work of Soryal et al. [30] did, in a mixed diet, showed higher concentration of fatty acids (0.54 g/100 g) compared with those measured in animals in full confinement (0.42 g/100 g). In our work, total fatty acids were higher than those reported before, particularly for IP and IR. Differences in diet, management and measurement techniques could explain variations.
Other studies by Jensen [41], Chapkin [42] Banskalieva [43] demonstrated that consumption of saturated fatty acids, specially lauric (C12:0) myristic (C14:0) and palmitic (16:0), are related with an increase in blood cholesterol being hypercholesteremic due to an increase in plasma LDL leading likely to cardiovascular disorders, those same fatty acids where particularly less in grazing diets.
Previously, value of 9.8% was reported for stearic acid, similar to amounts found in GP and GR which demonstrates that management system changes cheese composition as shown by Carpino [45]. Oleic acid concentration in soft cheese was evaluated before by Zlatanos [46] as 14.8% superior to the 10.5% to 11.6% shown in the present observation.
In another study, Banskalieva [43] discussed that the consumption of stearic acid (C18:0) and oleic acid (C18:1) diminished blood cholesterol levels because it reduced LDL and augmented HDL, favoring liver formation of low density lipoproteins (VLDL) that allows cholesterol to be transformed to gall bladder salts, values similar to the ones demonstrated for grazing products in the present observation.
In relation to Poly-Unsaturated Fatty Acids precursors of ω-6 and ω-3 fatty acids, it has been shown that quantities of linoleic acid (LA) and alfa-linoleic acid (ALA) are affected by the feeding system [46,47]. Similar tendencies were demonstrated in the present study.
The cis-9,12-octadecadienoic acid (C18:2 linoleic) in IP cheese had higher concentration (2.85%) compared with cis-9,12,15- Octadecatrienoic acid (C18:3 alfa-linolenic) in the GR that had been the highest (0.9%). This result was similar compared to the results shown by Zlatanos [46] with concentration of 2% and 1% for linoleic and linolenic acids respectively, discussing that apart from feeding, cheese manufacturing process could affect FA concentration.
GR cheeses showed the higher concentration to cis-5,8,11,14- eicosatetraenoic acid (arachidonic) and production of the elongation of the linoleic acid. Jensen [41], Chapkin [42] and Bankalieva [43] reported that a higher intake of arachidonic acid, metabolized prostaglandins 12 to 1 and thromboxanes A2, elements related with cardiovascular diseases and inflammatory disorders. All cheese values in the present observation were high in healthy fatty acids related to human health.
Finally, Castro [47] estimated that the optimal relation between ω-6 and ω-3 should be 5:1 or less. Our results for soft goat grazed cheese had an average 3.2:1 relation being rich in ω-6, but did not pass the health values discussed above. Pasteurization did appear to diminish cheese quality related to fatty acids and amino acid content in the present observation.
A significant difference was found for the presence of the ratio ω6 /ω3 greater than 4:1. All farms had a level higher than 4 mg/l of milk (0.30%). In 37 of the farms studied, the ratio of ω6/ω3 exceeded the limits of 4:1. In these farms, the use of concentrate was reflected in a decrease of ω3 and an increase of ω6 for the average 25 farms. The use of concentrate had a significant correlation (-0.8241 P<0.01) between the values of ω6/ω3. In 61 farms, all of them grazing, the omega 6/omega 3 ratios were less than 4: 1, which showed the beneficial effects of grazing on the quality of milk or goat cheese.
This Research was supported by PAPPIT IN IT202014 and Catedra Faculdad de Estudios Superiores Cuautitlan, Universidad Nacional Autonoma de Mexico.
Citation: Galina AM, Pineda J, Piedrahita RIH, Vázquez P, Haenlein G, Olmos J, et al. (2019) Effect of Grazing on the Fatty Acid Composition of Goat´s Milk or Cheese. Adv Dairy Res. 7:227 doi: 10.35248/2329-888X.19.7.227
Received: 22-Aug-2019 Accepted: 09-Sep-2019 Published: 16-Sep-2019 , DOI: 10.35248/2329-888X.19.7.227
Copyright: © 2019 Galina AM, et al. This is an open access article distributed under the term of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.