Pediatrics & Therapeutics
Open Access
ISSN: 2161-0665
ISSN: 2161-0665
Research Article - (2023)Volume 13, Issue 1
Background: Pediatric oncology patients are a high-risk population for clinical deterioration that might require Pediatric Intensive Care Unit (PICU) admission. This study aims to assess the clinical characteristics of pediatric oncology patients admitted to PICU with acute events, and to identify prognostic factors related to mortality.
Materials and Methods: All PICU admissions for cancer-related acute complications in oncology patients’ ≤ 18 years followed at a Pediatric Oncology Unit (POU) of a general oncology hospital, from 2017 to 2021, were retrospectively evaluated. We assessed the relation between clinical characteristics and PICU interventions, and the in-hospital mortality during PICU admission and at 30-day after PICU discharge.
Results: Of 4225 admissions to the POU ward, 63 patients (1.6%) were admitted to PICU. Most had hematologic malignancies (83%) and were under induction/neoadjuvant chemotherapy (54%). The main reasons for PICU admission were cardiovascular collapse (53%), respiratory failure (30%), and neurologic deterioration (14%). There were no significant differences between survivors and non-survivors in relation to underlying cancer disease, disease status (including relapse or HSCT recipient status), or cause of PICU admission. The mean PICU stay was 6 ± 0.9 days. The PICU mortality rate was 19% and the cumulative in-hospital mortality at 30 days after PICU discharge was 29%. In the multivariate analysis, mechanical ventilation (OR 17.6; 95% CI: 3.8–80.1; p<0.001), confirmed infection (OR 3.6; 95% CI: 1.1–13.5; p=0.042), and younger age (OR 0.9; 95% CI: 0.7-0.9; p=0.045) were significant predictors of mortality. Mechanically-ventilated patients who were under vasoactive support had lower mortality risk when compared to those under mechanical ventilation only (OR 6.0; 95% CI: 1.9-18.2; p=0.001).
Conclusions: Although a small percentage of pediatric oncology patients required PICU admission for cancer-related acute complications, the mortality rate was still high. Mechanical ventilation was the strongest predictor of mortality. The use of vasopressors may be associated with a significant decrease in the mortality of mechanically ventilated children with cancer.
Paediatric oncology; Cancer acute complications; Paediatric intensive care; PICU mortality
Enhanced treatment and support over the last decades have increased overall survival of children with cancer from an estimated 20% at 5 years in the late 1980s to 80% nowadays [1-3]. However, the intensification of treatment protocols has also led to an increased incidence of therapy-related life-threatening toxicities and side effects [4-6]. Recent studies show that almost 40% of all pediatric oncology patients will require admission to a Pediatric Intensive Care Unit (PICU) at some point during their disease [5-8]. The PICU mortality rates of children with cancer are far higher than those observed in the general PICU population, reaching 33% for oncological non-surgical patients [8]. Furthermore, their PICU mortality has remained relatively unchanged over the past decades, in contrast with the improved survival seen in adults with cancer admitted to intensive care [8].
Several studies have reported the characteristics and outcomes of children with cancer who require PICU admission, with acute respiratory failure and sepsis being the main causes [5-7, 9]. However, most studies include post-operative admissions, which may obscure the nature of cancer-related acute complications and its prognostic factors. Moreover, although some have pointed out disease characteristics associated with worse outcomes, little information is available on the existence of potential favorable prognostic factors related to PICU interventions.
Understanding which clinical characteristics and interventions act as predictors of outcomes for emergency PICU admissions of pediatric cancer patients is essential for supporting both the oncologists and the intensivists in the optimization of supportive care and in the allocation process of PICU resources’ utilization.
Our study aims to expand the current knowledge of the clinical characteristics of pediatric oncology patients admitted to PICU with acute events, and to identify prognostic factors related to mortality in this group of patients.
We conducted a single-center retrospective cohort study of pediatric oncology patients admitted to a Pediatric Oncology Unit (POU), between January 2017 and January 2022. This POU, founded in 1960, is located in a general oncology hospital in a high-income Western European country. The referral area extends over 41.769 km2 with a total population of 4.1 million (14.8% are children aged 0-19 years); additionally this POU admits children referred from international cooperation protocols with African countries. On average there are 180 new pediatric cancer diagnoses and 850 in-hospital admissions annually. There is no PICU in the hospital; therefore the POU has protocols with three close-by PICUs (within 12 km, average travel time by pediatric emergency ambulance less than 15 minutes).
We included all patients who were admitted to the POU and transferred to a PICU due to cancer-related acute complications. Patients transferred to the PICU for perioperative management, following Hematopoietic Stem Cell Transplantation (HSCT) or any elective admissions were excluded.
Review of the clinical files included demographics and data on the underlying primary disease, treatment phase, reason for PICU admission, length of PICU stay, confirmed infection (defined by positive culture, tissue stain, or polymerase chain reaction test in normally sterile sites), therapeutic interventions (vasopressor support, mechanical ventilation, dialysis, Extracorporeal Membrane Oxygenation (ECMO), surgical procedures) and whether these were started before transfer or after arrival at the PICU, and outcomes (mortality during admission and at 30 days post PICU discharge). The primary outcome was PICU mortality. The secondary outcomes were overall in-hospital mortality, measured at 30 days after PICU discharge; and PICU Length-of-Stay (LOS).
Descriptive analysis was used for characterization of this population. Continuous variables were described as mean and Standard Deviation (SD) or as median and Interquartile Range (IQR). Categorical variables were described as Numbers (N) and percentages (%). The distribution of variables was tested by the Kolmogorov–Smirnov test. For comparisons of differences between two groups, t-Student or Mann-Whitney tests were used. Categorical variables were analyzed using the χ2 test or Fisher exact test. To estimate the magnitude of the association between several variables and mortality occurrence, Odds Ratio (OR) and 95% Confidence Intervals (CI) were calculated, using logistic regression analysis. A backward elimination method was used to establish a multivariate logistic regression model. Survival curves and rates were estimated by the Kaplan–Meier method and compared by the log-rank test. Descriptive and inferential statistical analysis was performed using Stata®(V. 16.0, StataCorp, College Station, TX). Statistical significance was considered for two-sided p-value<0.05, with a 95% CI.
The study was performed in line with the principles of the Declaration of Helsinki. Ethical approval was waived by the local Ethics Committee in view of the retrospective nature of the study and all the procedures being performed as part of the routine care.
In this 5-year interval there were 4225 admissions to the POU ward, and 66 transferals (1.6%; N=63 patients) to PICU for treatment of oncology-related acute complications. The demographic and clinical characteristics of the survivors and non-survivors are shown in Table 1. Median age was 7.1 years (IQR 9.2 years; minimum 5 months, maximum 17 years); 29 (46%) patients were male. Most children had hematologic malignancies (83%; 71% acute leukemia and 12% lymphoma), with solid tumors representing only 17% (including 5% of Central Nervous System (CNS) tumors). Most patients were under induction/neoadjuvant chemotherapy (54%); 23% were under relapse treatment and 5% post-HSCT. The median time since the initial diagnosis of cancer was 65 days (IRQ 50 days; minimum 1 day, maximum 12 years). The mean time from the admission in the oncology ward to transfer to PICU was 13 ± 2.2 days. The most common causes of PICU admission were cardiovascular collapse (N=35; 53%), respiratory failure (N=20; 30%) and neurologic deterioration (N=9; 14%). There were no significant differences between survivors and non-survivors in gender, underlying cancer disease, disease status (including relapse or HSCT recipient status), or cause of admission.
| Characteristics | Survivors (N=45) |
Non-survivors (N=18) |
p |
|---|---|---|---|
| Age,† years | 4 (2-7) | 7 (5-13) | 0.02 |
| Male, N (%) | 26 (57) | 9 (50) | 0.15 |
| Underlying disease, N (%) | |||
| Acute leukaemia* | 29 (64) | 14 (78) | 0.19 |
| Lymphoma | 8 (18) | 0 | |
| Solid tumours–noncentral nervous system** | 7 (16) | 2 (11) | |
| Solid tumours–central nervous system | 1 (2) | 2 (11) | |
| Disease status, N (%) | |||
| Newly diagnosed | 26 (58) | 7 (39) | 0.89 |
| In remission | 10 (22) | 2 (11) | |
| In relapse | 11 (24) | 3 (17) | |
| HSCT recipient | 3 (7) | 0 | |
| Cause of admission***, N (%) | |||
| Cardiovascular dysfunction | 26 (58) | 9 (50) | 0.17 |
| Respiratory failure | 11 (24) | 9 (50) | |
| Neurologic deterioration | 9 (20) | 0 | |
| Miscellaneous**** | 5 (4) | 0 | |
| PICU interventions, N (%) | |||
| Mechanical ventilation | 6 (13) | 8 (44) | <0.01 |
| Vasoactive agents | 20 (44) | 9 (50) | 0.02 |
| Pleural effusion drainage | 3 (7) | 2 (11) | 0.34 |
| Emergent surgery | 4 (9) | 0 | 0.22 |
Note: †Continuous variables are presented as median (IQR) values. *Acute lymphoblastic leukemia (N=28), acute myeloblastic leukemia (N=15). **Soft tissue tumors (N=4), bone tumors (N=2), kidney tumors (N=2), hepatoblastoma (N=1). ***Some patients had more than one cause of admission. ****Acute renal failure (N=3), electrolyte disturbances (N=2).
Table 1: Demographic and clinical characteristics of children (0-18 years) with cancer after admission to a Pediatric Intensive Care Unit, according to survival status at 30 days post-PICU discharge, 2017-2021 (N=63).
On PICU admission, the most frequent diagnoses were septic shock (62%), pneumonia (33%), acute CNS thrombosis, pleural effusion and acute renal failure (9% each) (Figure 1). Three patients developed Hemophagocytic Lymphohistiocytosis (HLH), and three other developed Posterior Reversible Encephalopathy Syndrome (PRES). While in the PICU, mechanical ventilation was required in 14 (22%) patients, for a mean of 6 ± 1.5 days; and 29 (46%) patients needed vasoactive agents for a mean of 4 ± 0.8 days (Table 1). Other major interventions, such as emergent surgery and catheter drainage for pleural effusion, were performed in 8% and 6% of patients, respectively. No patient required dialysis or ECMO. The mean PICU LOS was 6 ± 0.9 days.
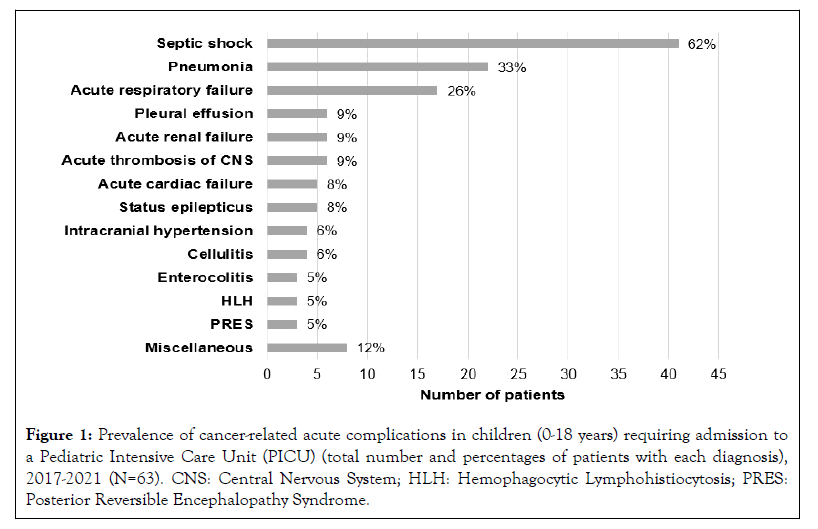
Figure 1: Prevalence of cancer-related acute complications in children (0-18 years) requiring admission to a Pediatric Intensive Care Unit (PICU) (total number and percentages of patients with each diagnosis), 2017-2021 (N=63). CNS: Central Nervous System; HLH: Hemophagocytic Lymphohistiocytosis; PRES: Posterior Reversible Encephalopathy Syndrome.
In 31(49%) patients infection was confirmed, and these had a higher mortality rate (40% in survivors vs. 58% in nonsurvivors; p=0.03). There was a clear predominance of bacterial infections (82%), and a high incidence of gram-negative agents (68%) with Klebsiella sp. as the most frequent pathogen (Figure 2).
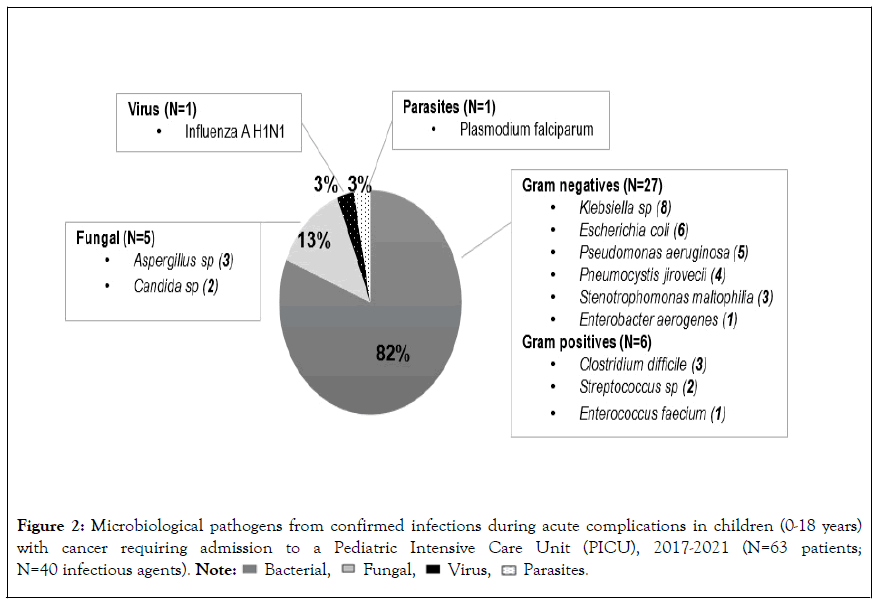
Figure 2: Microbiological pathogens from confirmed infections during acute complications in children (0-18 years)
with cancer requiring admission to a Pediatric Intensive Care Unit (PICU), 2017-2021 (N=63 patients;
N=40 infectious agents). 
Additionally, 26% (N=8) of patients had confirmed infection by 2 or more agents. While most patients (N=27, 87%) were on multidrug broad-spectrum antibiotics prior to PICU transferal, step-up of treatment during PICU admission was required in 68% of cases.
The PICU mortality rate was 19% (12/63 patients) and the overall in-hospital mortality, i.e. the cumulative mortality after PICU discharge, was 29% (18/63 patients). The main cause of death in the PICU was massive pulmonary hemorrhage (N=4), followed by refractory respiratory failure, refractory septic shock, and multiorgan failure (N=2); one patient died from massive gastrointestinal bleeding and another from intracranial bleeding. Children with acute leukemia or newly diagnosed disease under induction/neoadjuvant treatment had longer ICU LOS (8.8 ± 3.9 days, p=0.013; and 7.9 ± 2.7 days, p=0.030, respectively), but not increased mortality (p=0.110, and p=0.522, respectively).
On multivariate regression analysis, mechanical ventilation (OR 17.6; 95% CI: 3.8–80.1; p<0.001), confirmed infection (OR 3.6; 95% CI: 1.1–13.5; p=0.042), and younger age (OR 0.9; 95% CI: 0.7-0.9; p=0.045) were independent predictors of PICU mortality (Table 2). While the use of inotropes was associated with increased mortality on univariate analysis (OR 11.9; 95% CI: 1.5-105.5; p=0.026), it was not a risk factor for mortality when the several clinical factors were analyzed together (p=0.141). On the contrary, patients who were under mechanical ventilation and vasoactive support performed better with decreased mortality (OR 6.0; 95% CI:1.9-18.2; p=0.001), when compared to those under mechanical ventilation only (Figure 3).
| Clinical factors | Univariate analysis | Multivariate analysis | ||
|---|---|---|---|---|
| OR (95% CI) | p | OR (95% CI) | p | |
| Age | 0.8 (0.7-0.9) | 0.034 | 0.9 (0.7-0.9) | 0.045 |
| Gender | 0.3 (0.1-1.4) | 0.160 | _ | _ |
| Acute leukaemia | 5.6 (0.6-47.6) | 0.110 | _ | _ |
| Relapse treatment | 1.2 (0.2-5.1) | 0.824 | _ | _ |
| Sepsis and/or pneumonia | 1.2 (0.5-2.7) | 0.687 | _ | _ |
| PICU interventions | ||||
| Mechanical ventilation | 27.2 (2.1-92.1) | <0.001 | 17.6 (3.8-80.1) | <0.001 |
| Vasoactive agents | 11.9 (1.5-105.5) | 0.026 | 2.6 (0.7-9.5) | 0.141 |
| Mechanical ventilation+Vasoactive agents | 8.8 (1.2-16.7) | 0.001 | 6.0 (1.9-18.2) | 0.001 |
| Confirmed infection | 4.1 (1.2-13.3) | 0.017 | 3.6 (1.1-13.5) | 0.042 |
| PICU LOS | 0.9 (0.9-1.0) | 0.848 | _ | _ |
Note: LOS: Length-of-Stay.
Table 2: Logistic regression analyses of mortality for children (0-18 years) with cancer during admission to a Pediatric Intensive Care Unit for treatment of cancer-related acute complications, 2017-2021 (N=63).
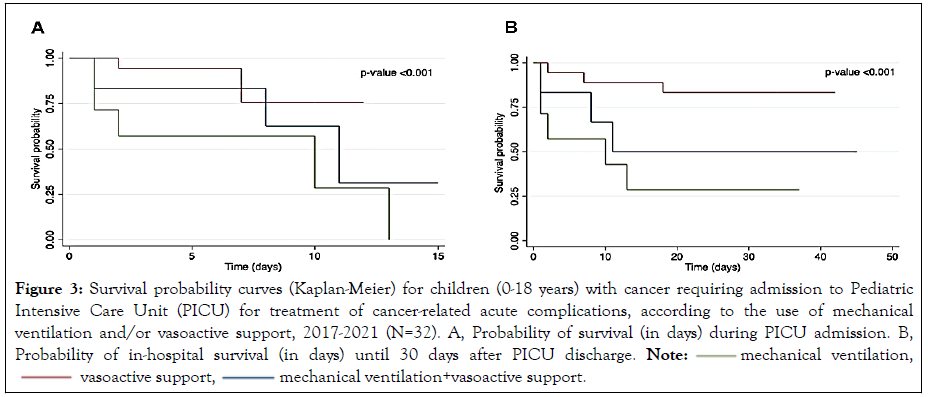
Figure 3: Survival probability curves (Kaplan-Meier) for children (0-18 years) with cancer requiring admission to Pediatric
Intensive Care Unit (PICU) for treatment of cancer-related acute complications, according to the use of mechanical
ventilation and/or vasoactive support, 2017-2021 (N=32). A, Probability of survival (in days) during PICU admission. B,
Probability of in-hospital survival (in days) until 30 days after PICU discharge. 

This retrospective 5-year study of acute complications in pediatric cancer patients requiring PICU admission showed a high PICU mortality, of 19%. This is, however, lower than in other studies [13,14] and in a meta-analysis [8] that reports mortality rates in the PICU of 33% when excluding postoperative patients, as in our study.
These findings may be related to several factors. First, we classified PICU mortality apart from overall in-hospital mortality. We found an overall mortality rate of 29% at 30 days after PICU discharge, closer to that reported in other studies. The main cause of death over this period was disease progression. This may signify that these patients are being offered the required intensive care treatment, yet when the acute complication is controlled they return to the POU to continue supportive treatment. This strategy is helpful not only for the management of limited PICU resources but, most importantly, it may create the optimal conditions for patients and families to go through the terminal phase with better comfort, supported by the oncology and palliative care teams that have known them for longer.
Secondly, the improved survival may be related to significant advances in PICU interventions, such as earlier therapy for sepsis and lung-protective ventilation strategies [8, 15, 16]. These specific approaches target the two most frequent cancer-related acute complications (septic shock and pneumonia/respiratory failure), which accounted for more than 60% of all PICU admissions in our study. The analyzed period is very recent (2017-2021) and may indeed reflect a contemporary trend for change of practices. Contrary to earlier studies (conducted between 1987-2017) [5, 7, 10-13], we found the use of inotropes to be associated with increased mortality only in univariate regression, not confirmed in the multivariate analysis. Most surprisingly, the use of vasoactive support in patients under mechanical ventilation showed a significant effect of improved survival. This can be explained by the enhanced cardiovascular support in a critical patient in respiratory failure, improving organ perfusion and indirectly compensating the balance of oxygen towards less consumption.
Thirdly, our findings may also be related to an earlier use of inotropes in our population. Over the recent years, it has become increasingly frequent to start vasoactive support while in the oncology ward, prior to transferal to a PICU. This practice follows the most recent international recommendations for pediatric hemodynamic stabilization with earlier start of vasoactive infusions [15]. In our setting, this practice is suitable to be implemented because: (1) Most cancer patients have a central catheter in place; and (2) There is a national specialized pediatric ambulance service that coordinates patient management since the moment the transport is activated for transferal to the PICU, guiding the oncologists in the on-site hemodynamic stabilization of patients, even before the ambulance arrival. In line with these findings, our mortality associated with septic shock was significantly lower than in reported in the literature. In a recent meta-analysis of 31 observational studies, the estimated pooled PICU mortality among patients with sepsis was 46%, whereas ours was 23% (9/40 patients) [8]. This can further support the rationale for the earlier beginning of vasoactive support in our practice.
To our knowledge, this is the first time that a beneficial effect of vasoactive support is shown in a study of pediatric cancer patients with acute life-threatening events. To note, Wöstenvan Asperen et al. [8], reported a significant improvement in intervention-specific PICU survival over the last 30 years in patients who required inotropic support. Together with our findings, this highlights the need for urgent prospective interventional studies in this population, to further unfold the potential for early and effective therapeutic interventions.
Although lower than in similar studies, our 19% PICU mortality rate is still higher than the current mortality rate of the general PICU population, ranging from 2.5% to 5% [17]. This strongly emphasizes the fact that pediatric oncology patients are a vulnerable population in the PICU. Their increased vulnerability derives in great measure from aggressive chemotherapy regimens, which puts them at a high risk of developing serious infections. Our results show that half of the patients had confirmed infection during the PICU admission; positive cultures were significant predictors of mortality in these patients. As children with cancer are immunocompromised and are at high risk for multidrug-resistant pathogens, early recognition of septic shock or other sepsisassociated organ dysfunction is critical, allowing for early implementation of empiric multidrug therapy [15].
Our study has three limitations. This is a single institution study with a relatively small population size. Studies including larger numbers of pediatric oncology patients from multiple centers would be helpful for a further assessment of the prognostic factors and the PICU interventions predictive of mortality and other adverse outcomes. We also lack formal calculation of a severity score on PICU admission, owing to the retrospective nature of the analysis. Finally, based on bed availability, during the study period patients were transferred to three PICUs, where different practices and therapeutic approaches cannot be excluded but that we were unable to address.
In conclusion, mortality of pediatric cancer patients during PICU admission for acute events is still very high. In our study the need for mechanical ventilation was the strongest predictor of mortality, in line with the literature. We showed that the use of vasopressors may be associated with a significant decrease in the mortality of mechanically ventilated children with cancer, probably reflecting improved cardiovascular support in patients in respiratory failure and shock. On the other hand, the underlying cancer diagnosis, the disease status (including relapse or previous HSCT), and the cause for PICU admission were not risk factors for mortality during the episode. These patients should therefore be given full consideration for intensive treatment and PICU admission during these acute episodes, supported by a multidisciplinary team discussion. This will allow these children to overcome the acute event, resume their cancer treatment after recovery and achieve the best long-term outcomes for their oncological disease.
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
Citation: Castelão M, Martins T, Gonçalves P, Lacerda A (2023) Characteristics and Predictors of Mortality after Intensive Care Admission of Children with Cancer. Pediatr Ther. 13.484.
Received: 05-Jan-2023, Manuscript No. PTCR-23-21272; Editor assigned: 09-Jan-2023, Pre QC No. PTCR-23-21272 (PQ); Reviewed: 23-Jan-2023, QC No. PTCR-23-21272; Revised: 30-Jan-2023, Manuscript No. PTCR-23-21272 (R); Published: 06-Feb-2023 , DOI: 10.35841/2161-0665.23.13.484
Copyright: © 2023 Castelão M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.