Forest Research: Open Access
Open Access
ISSN: 2168-9776
ISSN: 2168-9776
Research Article - (2015) Volume 4, Issue 2
The aim of this study was to examine the effect of rainfall on wood anatomical structures of Acacia senegal (L.) Willd located in the sandy soil. Samples of wood were collected from sites representing three rainfall (low, medium and high) conditions throughout the gum belt of North, West Kordofan States. Microscopic slides of wood samples were prepared to measure the size and percentage of wood cells. Analysis of variance was used to determine the differences in wood cells from different sites affected by rainfall. The differences in rainfall isohyets in sandy soil did not significantly affect the wood anatomical structures.
<Keywords: Acacia Senegal; Anatomy; Wood; Rain fall
In Sudan, Acacia senegal var. senegal grows in various environments ranging from semi-desert with under 100 mm rainfall to the fringes of the moist savanna with up to 900 mm rainfall and soils ranging from sand to heavy clays. It is found in pure stands or in mixture with other species [1,2].
Xylem adaptation to various conditions has been studied in different ways. One of the approaches is to study ecological variation within a single species to analyze the phenotypic responses of wood formation and differences due to environmental factors [3]. Water and nutrient interactions are widely recognized as key factors in determining forest productivity [4-6]. The pattern of response of trees to change is recorded in their wood structure [7]. It is useful to know how trees respond, either directly or indirectly, to various individual contributing factors that result from varying environmental conditions [8]. Anatomical structure is one of the main methods of timber classification and it is identified as one of the methods of characterization and behavior and use of timber.
An experiment was carried out in the sandy soil in the gum arabic belt under the different rainfall levels. Zone one in the low rainfall from (150-200 mm) was represented by Bara area(13°39′ N 30° 39′ E) “zone two and three’’ were represented by Elhemera (13°8′ N 30°13′ E) and Babanosa(11°20′ N27°48′ E) in moderate (250-350 mm) and high (450- 500 mm) rainfall isohyets, respectively.
Nine Acacia senegal trees were randomly selected from each rainfall level from each of the different areas. The selected trees were marked for wood samples. The samples were taken from the mature wood. From each sample, 3 specimens 1×1×2 cm were prepared for sectioning. A transverse section was prepared from each sample. The cubes were softened and sectioned with sliding microtome. The sections were then stained with safranin and mounted in Canada balsam for microstructures. The transverse sections of wood samples were examined using a microscope camera setting attached to a computer. The wood softening method is described elsewhere [9]. Stereological counts were conducted following a procedure described elsewhere [10], using a 0.75 mm grid length under 4/0.1 magnification. The measurements of the microstructure involved random counts on the microscopic sections superimposed to nine squared on sixteen point grid. The glass slides were fixed on microscope fitted with a camera and the cross sections were projected through the computer screen to obtain the point count (Pp), the number of points of intersection with cell boundaries per unit length of test lines (PL) and the number of objects or features in the count area of the microstructure (NA) for vessels, parenchyma, fibres and rays.
The stereological equation relating fractional measurement in microstructure to point fraction according to Ifju et al., [11] is as follows:
Vessel volume fraction = Pp vl + Pp vw (1)
Parenchyma volume fraction = Pp pl + Pp pw (2)
Fiber volume fraction = Pp fl + Pp fw (3)
Ray volume fraction = Pp rl + Pp rw (4)
Where l is lumen, w is wall, v is vessel, p is parenchyma, f is fiber and r is ray.
For the general description of stem anatomy, stereological method was used to calculate average cell diameter vertical and horizontal (Dv,h), average lumen diameter (LD), the mean free path between cells (MFP), vessel shape factor (VSF), fiber density index (FDI) and double cell wall thickness (DCWT) for vessel and fiber, Rankle’s ratio and fiber hardness rate . The following equations were used:
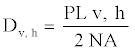 (5)
(5)
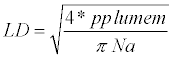 (6)
(6)
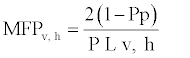 (7)
(7)
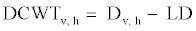 (8)
(8)
 (9)
(9)
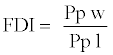 (10)
(10)
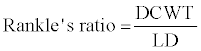 (11)
(11)
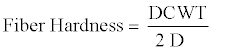 (12)
(12)
The data were analysed using the statistical analysis system (SAS), JMP programs. Analysis of variance and Duncan’s Multiple Rage tests at 0.05 probability level was used to study the significance of the differences between the mean of wood parameters from different locations of the gum belt.
The diameter (vertical and horizontal) of vessels, fibers, and parenchyma were similar in sandy soil under different rainfall levels. Also there were no significant differences between the mean free path of vessels vertical or horizontal in sandy soil under different rainfall levels (Table 1). Tables 2 and 3 show the rest of the anatomical characteristics of cells. There were no significant differences between these parameters in sandy soil under different rainfall levels except fiber double cell wall thickness, fiber hardness and rankle’s ratio which were significantly affected under different rainfall. Abd elgadir [12] reported that the diameters of vessel radial and tangential are 0.112 mm, 0.142 mm respectively, lumen diameter of vessel is 0.084 mm, parenchyma diameter radial 0.026 mm and tangential is 0.023 mm, vessel mean free path 0.5 mm radial and 0.03 mm tangential, the vessel shape factor is 1.8 mm, fiber density index is 0.9 mm, double wall thickness for vessels are 0.03 radial and 0.06 tangential, double wall thickness for fibers are 0.02 radial and 0.02 tangential, volume fraction for vessel, fiber, parenchyma, ray are 18.3 mm, 40.8 mm, 28.8 mm, 12.1 mm respectively. These results are more or less similar with results of the present study. Sahri et al. [13] reported on wood anatomical structure of Acacia mangium showed that the average percentage of fiber, vessels and rays are 85.8%, 9.1%, and 5.2%, respectively. The average fiber diameter and fiber lumen diameter fiber, wall thicknesses are 24, 17 and 3.3 μm, respectively. The vessels percentage decrease with increasing height. Fernandez et al. [14] reported that severe drought affected differentially the amount but not the function and quality of formed wood in Pinus ponderosa growing under different competition levels. He showed that some wood anatomical variables of P. ponderosa did differ in more water stressed trees; lower cell wall thickness of early wood cells and higher proportion of small-lumen cell in later wood. In this respect, Nasroun [15] reported similar trend of association in different tree species. He showed that Acacia senegal, which had large vessel diameter and small number of vessel per unit area, also had very large mean free path between vessels (0.8 mm). On the other hand, Eucalyptus microtheca [16] had a large number of vessels per unit area and small (0.4 mm) mean free path between vessels. In addition, the mean free path between vessels decreased with increase in height and increase from the pith. Means followed by the same letter in columns are not significantly different using DMRT at P=0.05.
| Rainfall | DVv | DVh | DPv | DPh | DFv | DFh | MFPVv | MFPVh |
|---|---|---|---|---|---|---|---|---|
| Low | 75.82a | 102.21a | 8.77a | 8.98a | 6.39a | 6.56a | 1046.81a | 800.97a |
| Medium | 77.76a | 104.74a | 6.95a | 7.85a | 7.36a | 7.28a | 2541a | 1033.49a |
| High | 93.59a | 91.66a | 7.03a | 6.52a | 5.44a | 5.58a | 1187.65a | 1152.52a |
| P | 0.77 | 0.55 | 0.18 | 0.35 | 0.18 | 0.13 | 0.54 | 0.49 |
| SE± | 18.98 | 8.61 | 0.68 | 1.09 | 0.63 | 0.49 | 1010.7 | 199.52 |
Table 1: Diameter of different cells and mean free path of vessels (in micron) of Acacia senegal in sandy soil different rainfall levels. Means followed by the same letter in columns are not significantly different using DMRT at P=0.05.
| Rainfall (mm) | DCWTVv | DCWTVh | DCWTFv | DCWTFh | LDV | LDF |
|---|---|---|---|---|---|---|
| Low | 9.03a | 20.47a | 1.51ab | 1.34ab | 83.96a | 7.91a |
| Medium | 30.04a | 56.96a | 1.25b | 0.51b | 47.71a | 7.54a |
| High | 17.24a | 11.55a | 2.57a | 2.35a | 84.86a | 8.02a |
| P | 0.34 | 0.11 | 0.07 | 0.02 | 0.2 | 0.45 |
| SE± | 9.31 | 13.55 | 0.34 | 0.34 | 14.56 | 0.26 |
Table 2: Lumen diameter and double wall thickness of different cells (in micron) of Acacia senegal in sandy soil under different rainfall levels. Means fallowed by the same letter in columns are not significantly different using DMRT at P=0.05.
| Rainfall (mm) | V S F | FDI | F Hardness | Rankle's ratio | Wood% | Vessel % | Parenchyma % | Fiber % | Ray % |
|---|---|---|---|---|---|---|---|---|---|
| Low | 0.77a | 2.47a | 0.1b | 0.36b | 53.33a | 12.33a | 36.33a | 25a | 26.33a |
| Medium | 0.73a | 2.88a | 0.04b | 0.18b | 48a | 7.66a | 36.33a | 24.33a | 31.66a |
| High | 1.01a | 3.38a | 0.22a | 0.62a | 54.66a | 9a | 34a | 29.66a | 28a |
| P | 0.58 | 0.27 | 0.007 | 0.01 | 0.44 | 0.43 | 0.81 | 0.41 | 0.43 |
| SE± | 0.19 | 0.35 | 0.11 | 0.06 | 3.63 | 2.44 | 2.89 | 2.86 | 2.8 |
Table 3: Vessel shape factor, fiber (density index, hardness) Rankle’s ratio and volume fraction of different cells (in micron) of Acacia senegal in sandy soil under different rainfall levels. Means fallowed by the same letter in columns are not significantly different using DMRT at P=0.05.
Generally, there are no significant differences between different compositions of anatomic cells consisting of wood under the influence of the different levels of rainfall in sandy soil. Hence, this species is well adapted with its environment without dramatic changes in the anatomical structures.