Journal of Antivirals & Antiretrovirals
Open Access
ISSN: 1948-5964
ISSN: 1948-5964
Review Article - (2021)
During inhalation oxygen molecules are drawn towards type I cells. The partial pressure difference and solubility factor act as a Drive for this movement of oxygen molecules. It is well known that the pulmonary surfactant plays an important role in gas exchange. The surfactant is thin mono-layer. The top surface of surfactant with SP-B and C proteins faces alveolar air and is hydrophobic in nature and acts as a surface tension reducer, whereas lower surface with SP-A and D proteins is hydrophilic and is adsorbed on mucosal layer. This lower surface of surfactant acts as an anti-invader, pathogen barrier. However there is a small missing link in explaining its exact mechanism or role in justifying these properties during normal conditions and during ‘viral ligand’ attack.
Similarly a unique physical property of SP-C component is listed in research papers; however its application is not researched anywhere. SP-C has a dielectric constant of 2 to 3 and plays a very critical role in drastically reducing progression of any respiratory viral disease including SARS-CoV-2. This hypothesis targets to explain both micro mechanisms with the help of basic laws of physics, and fluid mechanics. The figures/sketches, drawn also depict the physics involved and not much of a physiology or genetic codes etc. Two examples, in the industry, are briefly listed in the last paragraphs to draw some parallel with above mechanisms.
Vaccination is a proven method for containment of respiratory viral disease, if not its cure. But if focus is also given on health of pulmonary surfactant with respect to pathogenesis of any respiratory viral disease, probable loss of lot many lives can be avoided and vaccine development and vaccination management related issues can be less panicky.
Surfactant; Type I and II cells; Partial pressure; Surfactant cover
Basis 1
In a normal lung, the alveolar space is lined by mucus, which covers lung surface, including Type I and Type II pneumocytes. Type I pneumocytes are responsible for gas exchange, which is governed by gas laws of partial pressure difference and respective gas solubility. This drives the gas molecules to travel across mucus layer [1].
Type II pneumocytes generate pulmonary surfactant which floats over the mucosal fluid. This surfactant is basically amphiphillic protein-lipid combination mono-layer. The oleophillic, hydrophobic component with surfactant proteins SP-B and SP-C forms upper surface of the surfactant layer. It faces up the alveolar air. It basically reduces surface tension and facilitates smooth respiration. The hydrophilic component with surfactant a protein SP-A and SP-D forms lower surface and is adsorbed on the fluid mucus layer and acts as an anti-invader [2].
However there is no document which will illustrate how, where and why the surfactant is consumed? Why type II pneumocytes have to release surfactant regularly? Does the surfactant play role same as hemoglobin in blood? Is surfactant a mute spectator in viral attack?
Hypothesis 1
Role of surfactant in gas exchange mechanism: When an oxygen molecule moves past the surfactant, its small portion is pushed along with oxygen molecule. This can happen because there is very little surface tension. The top or upper surface of this little surfactant becomes inner surface and lower surface becomes outer face of bubble cover (Figure 1) [3]. In other words the top hydrophobic face with SP-B and C proteins now becomes inner surface and maintains low surface tension (Young-Laplace equation for thin film with outer and inner face) and the anti-invader surface of SP-A and D forms outer face of the bubble cover and provides protection against mucosal pathogens if any. As the oxygen bubble hits type I pneumocytes, a pore forms on the surfactant bubble cover [4]. While oxygen molecule diffuses in type I pneumocytes, surfactant cover breaks down as soon as the surfactant component SP-B and C comes in contact with water based mucus (Figure 1).
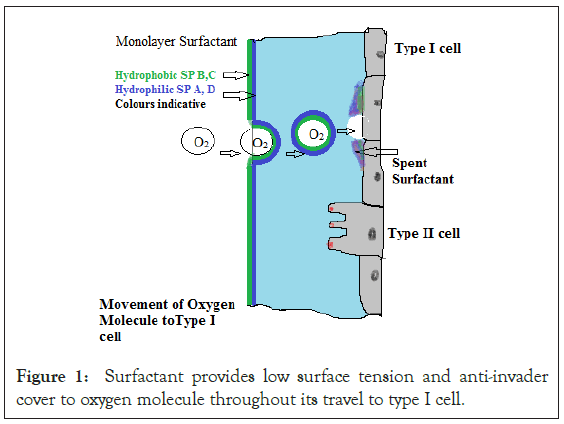
Figure 1: Surfactant provides low surface tension and anti-invader cover to oxygen molecule throughout its travel to type I cell.
During exhalation carbon dioxide is also driven to alveolar space due to partial pressure difference (mere 4 mm Hg); along with fresh or make up quantity of surfactant. The surfactant released by Type II pneumocytes, forms a thin layer between mucosal fluid and periciliary fluid just above epithelium. In this case also the hydrophilic surface adsorbs on mucosal fluid above it and forms top or upper face and hydrophobic surface forms lower face of the surfactant released by Type II pneumocytes. This surface has least surface tension. As CO2 diffuses from Type I cell, below the surfactant layer, the hydrophilic component forms upper surface and hydrophobic component, an inner surface of carbon di-oxide molecule bubble cover [3,4]. At the air-mucosal fluid interface, while carbon di-oxide molecule is released for exhalation, the surfactant takes its default position (Figure 2). Thus the surface tension reducing and anti-invader property is available during entire gas exchange.
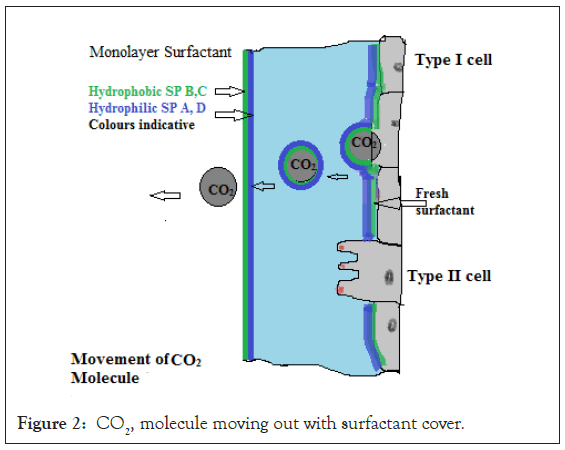
Figure 2: CO2, molecule moving out with surfactant cover.
Basis 2
Corona virus is present where ever there is a foot fall. Droplets through respiration, coughing, sneezing by an infected person are major source of active corona respiratory viral transmission. Further scientists are now confirming possibility of airborne transmission of SARS-CoV-2 [5,6].
A single corona virus; a genetic structure; is generally electrically neutral particle of an average submicroscopic size of 100 nm and will follow a Brownian motion while airborne. During this motion there are two main possibilities, it can acquire static charge by; a) Its surface becoming polar and demonstrating a charge while passing through some, electrical field or b) by hitting or bouncing off a charged surface [7,8]. Even humans get statically charged. But can get grounded and lose charge easily. However virus cannot lose charge so easily, because while airborne or resting on some non-conducting surface, getting grounded is impossible. Thus at places of crowding like, airports, airplanes, malls, stations, metro, passenger vehicles, theatres, spas, hospitals etc. possibility of virus acquiring charge and getting active are very high. The virus shall be in form of a clump and not as a single antigen.
Not all of this active viral material, which is inhaled, can reach alveolar space. Some gets stuck in the mucus of respiratory track and loose charge and gets tracked and seized by IgA and other immunoglobulin and some are segregated by unique beating motile cilia and pushed to esophagus via pharynx [9]. Being of same ‘net acquired charge’, the viral clumps repel each other within individual alveoli as per Coulomb’s law, the repelling force (F) is inversely proportional to square of the distance between two (d). In an effect viral clumps are pushed towards pulmonary surfactant. In spite of this, the viral material cannot just bond with ACE2 receptors without overcoming pulmonary surfactant. It is important to note that a virus does not know what is its destination, nor a direction to the destination i.e ACE 2 receptors. It becomes an intelligent-viral ligand-only after it is driven to the destination by some mechanism. The physics behind this nano mechanism is explained in next paragraphs.
Hypothesis 2
Role of surfactant in gas exchange mechanism during respiratory viral attack like SARS-CoV-2: The position of surfactant, type I and II cells and the gas exchange mechanism is already explained in Hypothesis 1. Type II pneumocytes generate pulmonary surfactant, which ultimately floats over the mucus. There are research papers on physical properties, structure, and composition of these surfactants. Huge data is available but none define mechanism of its functioning.
One such physical property of the SP-C component is listed in research papers although its purpose remains unexplored. SP-C is a very good dielectric material having dielectric constant of 2 to 3 as compared to hydrophilic component with SP-A and D proteins and respiratory mucus; which has dielectric constant of 50 onwards [10]. Thus apart from reducing surface tension, SP-B and SP-C shall act as a dielectric barrier between any statically charged or statically oriented viral layer or dust and the host cells i.e. Type II pneumocytes with ACE2 receptors (Figure 3).
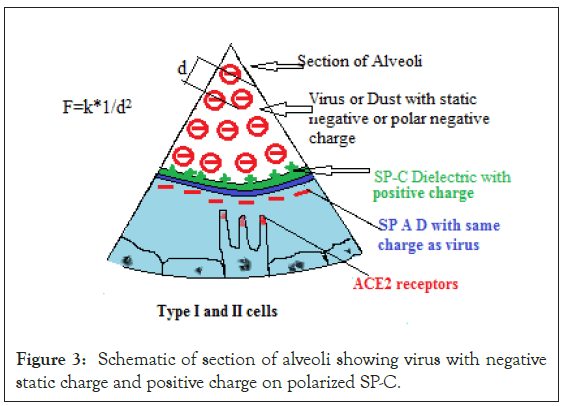
Figure 3: Schematic of section of alveoli showing virus with negative static charge and positive charge on polarized SP-C.
A dielectric material is one which is a poor conductor of electricity, but can support electrostatic fields. This means that if the material is exposed to an external electric field, it gets polarized. This allows it to store electric charge, making it a poor conductor, but a good storage medium.
The SP-C of the top surface of surfactant becomes polar and acquires opposite charge as that of the viral load, whereas the lower surface of SP-A and D displays same charge as that of the viral load (Figure 3).
Thus when oxygen molecule is driven through the surfactant the upper surface with SP-C and B forms inner face and SP-A and D outer as explained in hypothesis I. But outer face being of same charge as that of the viral or dust loads may repel it and stop it from getting driven along with it. This is also how SP-A and D may act as anti-invader in case of any respiratory viral infection. Apart from this, the charged molecules on outer surface further repel each other and increase diameter of bubble (Figure 4).
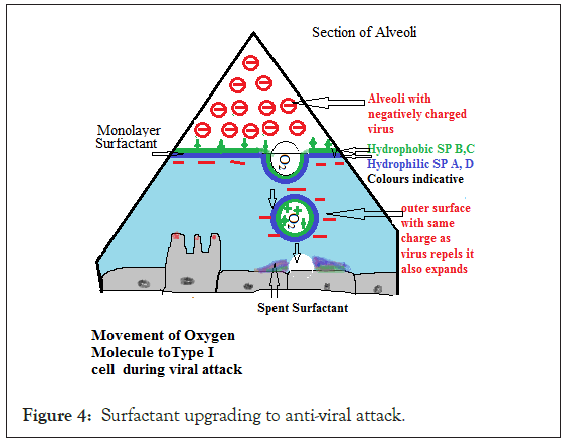
Figure 4: Surfactant upgrading to anti-viral attack.
At the air-liquid interface dielectric SP-C component of the surfactant keeps on storing charge dissipated by the viral load. Viral load loses its static charge, but at the cost of dielectric strength of SP-C. The dielectric strength is a measure of a materials ability to sustain high voltage differences without breakdown. As the SP-C loses dielectric strength, voltage across it effectively increases, and at some point, current transits through the weakened dielectric to a closest conductor (Figures 5 and 6). This clearly shows criticality of the role played by surfactant and need to focus on its health with respect to pathogenesis of any respiratory viral disease.
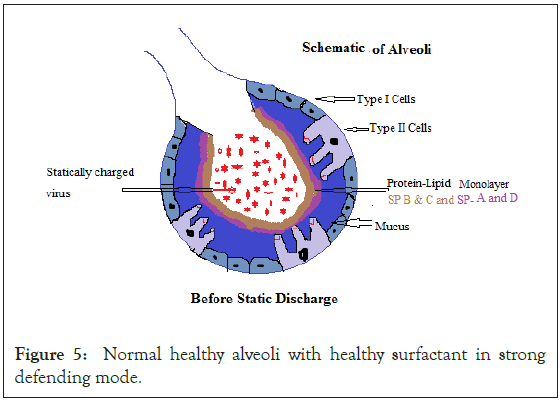
Figure 5: Normal healthy alveoli with healthy surfactant in strong defending mode.
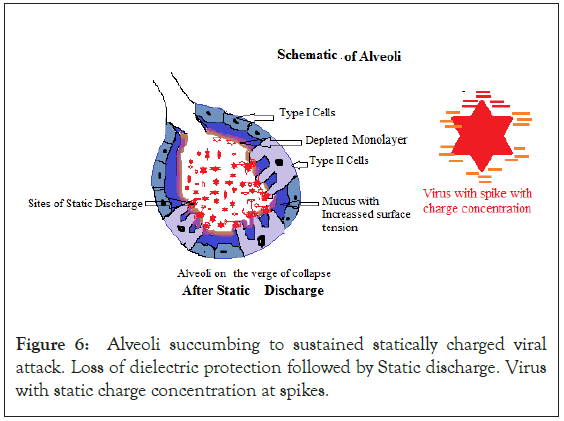
Figure 6: Alveoli succumbing to sustained statically charged viral attack. Loss of dielectric protection followed by Static discharge. Virus with static charge concentration at spikes.
The dielectric strength typically is stated in volts per centimeter. The dielectric strength of an insulating material has a highly stochastic characteristic and can only be described as a function of probability.
Thus in case of severe sustained viral attacks or a charged dust attack, the SP-C and SP-B component is SPENT rapidly as compared to replenishment from type II pneumocytes and is converted to watery liquid thereby losing its surface tension reducing property also [11]. This increases surface tension in mucosal fluid.
Increased surface tension causes shrinkage of mucosal layer from all direction, uncovering ACE2 receptors, exposing them to fresh viral attack and a state of dielectric barrier breakdown (Figure 6).
At the breakdown of dielectric barrier a static discharge; occurs through spikes of viral material. The spikes have concentration of charge at surface, because of its shape i.e., more repulsion from within as compared to surface. As the spikes are present all over the viral structure, static discharge is feasible in any direction in spherical alveolar space. The total alveolar surface area of lungs is around seventy five to one hundred square meters, which has millions of alveoli which in turn means, that many number of possible sites for static discharge. It is like a capacitor plate with huge surface area.
The capacitance of human body is around 200 pF, and voltage build up could be few thousand volts, although the discharge currents are tiny [12]. The viral clump being very light and submicroscopic moves as a charge, and chooses closest point on host cell i.e. ACE2 receptor. This is how the virus is driven to the location, where viral ligand gets activated, and enters host cell (Figure 6). The binding between SARS-CoV-2 spike glycoprotein and ACE 2 receptors takes place. This is followed by first stage replication and release.
In nutshell, in sustained viral attacks, gas exchange gets severely affected as the surfactant quantity and quality gets depleted. This ultimately leads to death or permanent collateral damages to other organs, including heart and brain where oxygen requirement is critical and also probably tongue and eyes due to gas exchange which is devoid of any surfactant protection.
Testing
If the shrinkage of mucus due to dielectric barrier dysfunction can be measured easily, then possibility of a person, contracting the disease can be predicted.
This measurement can predict whether third or subsequent waves can happen and who are going to be its targets.
A test module with simulation of inhalation, with multilayered statically charged filters can be installed at various crowding locations to measure magnitude of dominant static charge of airborne virus for further analysis and action [13].
Example from industry as a backup for the hypothesis
Bag filter: Let us look at an example of de-dusting system; commonly installed for removing dusts from metal processing plants, textile mills, mines, sugar mill, flour mill etc.; which to certain extent is similar to human inhalation. One can visit such installations at nearest industrial towns across the world. Only major difference is that, the industrial dust size shall be above 1 micron.
In a bag filter type dust collection system, gas laden with dust loads, travels through ducts into a hopper with filter membrane, across which filtration takes place. The dust laden gas is sucked in by induced draft fans, which generate negative pressure in the system. During this movement and friction with air, a static negative charge starts building up on the dust particles. And the particles remain charged, if any loss of charge while in motion is less than total charge collected. All such charged particles shall form a layer near the interface, i.e. filter membrane between air laden with dust and filtered air manifold. And a stage reaches when this charged layer tries to discharge to nearest possible conductor by breaching the filter medium (Figure 7) [14].
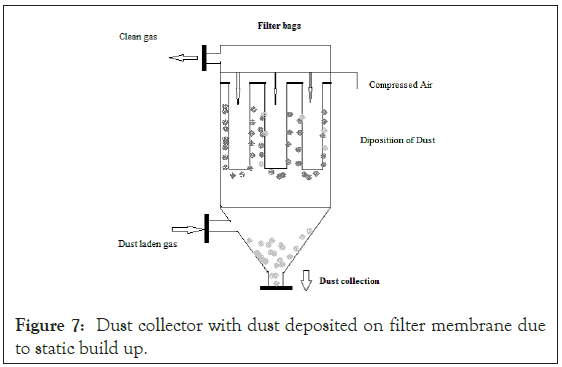
Figure 7: Dust collector with dust deposited on filter membrane due to static build up.
This can result in failure of the system and damage to the membrane or even fire. To avoid this one has to use either anti-static filter membrane or a spark arrester. By using anti-static membrane, the dust gives away its charge and does not stick to its surface and falls down in the hopper for separation by reverse pulse air. All bag filters across the world in industries listed above use antistatic bags and spark arresters to avoid loss.
Oil cooled transformers: The oil, in the oil immersed transformers which we see installed in our neighborhood, has a functional similarity with SP-C component of pulmonary surfactants. The oil insulates as a strong dielectric medium, suppresses corona discharge and acts as a coolant. The study of drawing functional similarity between SP-C and transformer oils; which can be mineral oil or even edible vegetable oils; is beyond scope of the presentation at this stage.
Thus it can be inferred that,
It is need of hour to focus line of action of medication, on health of pulmonary surfactant so that progression of any respiratory viral disease can be drastically curtailed before it can attack ACE2 receptors.
SARS-CoV-2 or any other respiratory viral infection can occur where there is, depletion or short supply of dielectric barrier consisting of SP-C protein.
At any given instance, status of pulmonary surfactant especially, the dielectric strength and surface tension reducing property shall define susceptibility to any viral attack.
The infection strength of virus shall depend on presence of spike protein and its geometry. A normal dust without spiked component, or even a viral material, without or a less projecting spike protein will have much lesser infecting strength.
Citation: Phadke MA (2021) Role of Pulmonary Surfactant in Gas Exchange Mechanism and Providing Protection against Progression of any Respiratory Viral Disease like SARS-CoV-2 and Need to Focus it as First Point of Medication for all Respiratory Diseases. J Antivir Antiretrovir. S22:005.
Received: 08-Oct-2021 Accepted: 22-Oct-2021 Published: 29-Oct-2021 , DOI: 10.35248/1948-5964.21.s22.005
Copyright: © 2021 Phadke MA. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.