Lupus: Open Access
Open Access
ISSN: 2684-1630
ISSN: 2684-1630
Case Report - (2021)Volume 6, Issue 7
Background: Although pyoderma gangrenosum (PG) -like lesions have been rarely described in adults with the antiphospholipid antibody syndrome (APS) and systemic lupus erythematosus (SLE), the occurrence of PG as a preceding manifestation of APS in children with SLE has not been reported until. We present a young girl with SLE and APS who developed progressive extstensive ulcerations that were consistent with PG.
Case: A 14-year-old girl with a 2-year history of SLE was admitted to our department, complaining painful crusted ulcerations on her legs. Skin biopsy was reported as PG. However, she did not respond to immunosuppressive therapy administered. When her skin biopsy findings is reassessed in keeping with the positive anticardiolipin antibody results, superficial small vessel micro thrombosis was observed. Diagnosis of APS and PG developing secondary to SLE were made. It was resulted in marked clinical improvement with anticoagulation therapy in addition to immunosuppressive as is recommended in APS.
Conclusion: Based in clinical, pathological and response to proposed treatment, we can state that PG -like lesions in children with SLE could be considered as a secondary form of APS.
Systemic lupus erythematosus; Antiphospholipid antibody syndrome; Pyoderma gangrenosum; Mucocutaneous
Systemic lupus erythematosus (SLE) is a multisystem chronic autoimmune inflammatory disesease. Children with SLE may have a severe multisystem disease, including cutaneous lesions. One of the most common clinical features was mucocutaneous manifestations, followed by constitutional, hematological and musculoskeletal manifestations. In recent years, antiphospholipid antibody syndrome (APS) has been increasingly recognised in various pediatric autoimmune [1]. Patients with APS may develop cutaneous ulcerations which mimic pyoderma gangrenosum (PG), particularly leg ulcers, and similar lesions have rarely been reported in patients with SLE [2,3]. The associations of these clinical conditions in the same patient, which are rarely reported in adults, have not been found in the literature in children [4]. We present a young girl with SLE and APS who developed progressive extensive ulcerations that were consistent with pyoderma gangrenosum.
A 14-year-girl was referred to pediatric rheumatology outpatient clinic because of crusted ulcerations on her legs. She had diagnosed with SLE at pediatric rheumatology clinic, 2 years before. SLE diagnosis was based on the association of weight loss, alopecia, polyarthritis, raynaud's phenomenon, recurrent oral ulcers, Antinuclear Antibodies (ANA) positivity at a 1/320 dilution, double-stranded DNA antibodies positivity, and low complement levels. Renal involvement was initially absent. Diagnostic kidney biopsy could not be performed since informed consent could not be obtained from the patient and her family. Her SLE was being treated with hydroxychloroquine, mycophenolate mofetil, amlodipin and low dose (10 mg/day) of methylprednisolone. Skin examination revealed painful crusted ulcerations with overhanging border on the erythematous base, about 15*5 cm in size, on her legs (Figure 1). The other system examinations were unremarkable. Laboratory investigations showed a white cell count of 8.14*109/L(3.8–10.8*109), an absolute lymphocyte count of 0.26*109/L, hemoglobin of 10.3 g/dL (11.1–17.1), and a platelet count of 600*109/L (140– 360*109). Serum C-reactive protein was 3.14 mg/L (0-5) and erythrocyte sedimentation rate 80 mm/h (0-20). Results of coagulation tests were within the normal range. Laboratory investigations gave positive results for ANA and double-stranded DNA antibodies (>150 IU/mL; 0–18). Complement (C3 and C4) levels were low.
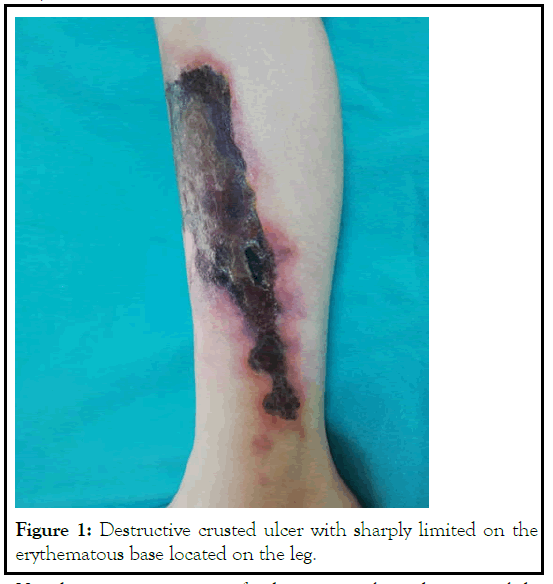
Figure 1: Destructive crusted ulcer with sharply limited on the erythematous base located on the leg.
Histologic examination of ulcerations showed neutrophilic infiltration in the entire height of the dermis. In addition, bleeding suffusions in the superficial dermis and epidermal ulceration, necrosis and leukocytoclasia were noticed (Figure 2).
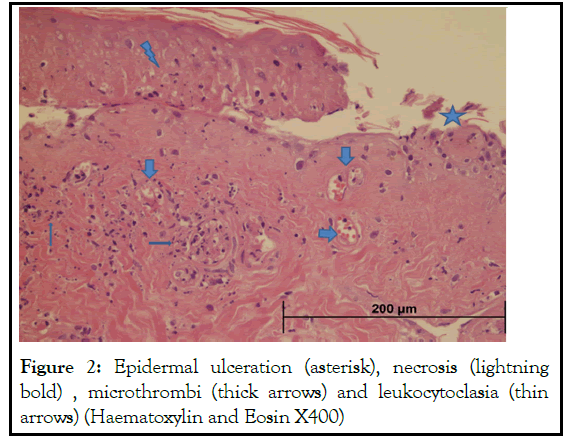
Figure 2: Epidermal ulceration (asterisk), necrosis (lightning bold) , microthrombi (thick arrows) and leukocytoclasia (thin arrows) (Haematoxylin and Eosin X400)
A skin biopsy for direct immunofluorescence was taken from the nonaffected area. Immunoreacatants (IgG, IgA, C1q) were negative. No deposits around dermal vessels were seen. Tissue cultures for fungi, mycobacteria and bacteria were negative. Skin biopsy was reported as pyoderma gangrenosum. She was started on daily intravenous methylprednisolone (30 mg/kg/day) for 3 consecutive days and oral prednisone divided doses 2 mg/kg/day was continued. Mycophenolate mofetil was changed with tacrolimus. She was discharged home on oral prednisone, hydroxychloroquine, tacrolimus and amlodipin. Unfortunately there was not improvement in her skin lesions after one month. Her antisphospholipid antibody profile was positive; anticardiolipin antibodies (IgG 17, 1 U/mL; normal range 0–12 U/mL, IgM 34, 3 U/mL; normal range 0–12 U/mL). When her skin biopsy finding is reassessed in keeping with the anticardiolipin antibody results, superficial small vessel microthrombosis was observed. Diagnosis of antiphospholipid syndrome and pyoderma gangrenosum developing secondary to SLE were made according to The Ped-APS Registry Collaborative Group and American College of Rheumatology’s classification criteria. Inherited and / or acquired prothrombotic risk factors (factor V Leiden, prothrombin 20210A gene mutation, polymorphism of methylenetetrahydrofolate reductase (MTHFR) gene, deficiencies of antithrombin, protein C, protein S, and hyperhomocysteine) were excluded [3,5]. Because the high similitude of clinical and pathological features of PG associated with occlusion of small dermal vessels by thrombi in ulcers of patients with APS, we decided to treat this patient with immunosuppressives as usual in PG in addition to ]anticoagulation therapy as is recommended in APS. She was started low molecular weight heparin (enoxaparin) for antiphospholipid syndrome. However, proteinuria (0.5 gr/day) was occured in the follow-up. Renal biopsy was not done because of risk of bleeding. Therapeutic plasmapheresis was performed for 5 consecutive days. Tacrolimus was switched with monthly cyclophosfamid. After two weeks, it was observed rapid improvement in her skin lesions and proteinuria was disappeared. After the remaining necrotic tissues were debrided by plastic surgery, flap transfer techniques was performed. Kidney and liver functions tests were normal. She was discharged home on oral methylprednisolone (15 mg/day), hydroxychloroquine, monthly cyclophosfamid (six months), anticoagulation therapy, amlodipin and vitamin D supplements. After 3 months in follow up, it was continued rapid improvement in her skin lesions and her anticardiolipin antibodies were still positive (Lupus anticoagulant and anti-β2 glycoprotein antibodies were not studied in our hospital).
Leg ulceration is a rare skin finding in patients with SLE, and usually occurs in patients with APS and/or leucocytoclastic vasculitis. It reported in adult studies of cases of SLE that it had noted leg ulcers in 5%-8% of them. In a study in Turkey, although mucocutaneous lesions were 97%, 8% in children with SLE, the skin ulcers were not reported [4,6]. PG is associated with a systemic disease, most commonly inflammatory bowel disease, arthritis, and hematologic malignancy, in at least half of the cases [1]. PG may precede, coexist, or follow the different systemic diseases. The association of PG with SLE, however, is rarely encountered [7]. In most of these reports, PG had developed years after the diagnosis of SLE. PG had developed two years after the diagnosis of SLE in our patient [8]. Most of the SLE patients associated with PG had antiphospholipid antibody positivity as in our patient. Though in some reports, ulcers observed in antiphospholipid antibody positive SLE patients are designated as PG like ulcers, it is not clear yet whether this discrimination is appropriate or not, as they show only minor differences in comparison to the real PG lesions [9]. Diagnosis of PG is based mainly on the morphology of the lesions, clinical course, exclusion of other conditions, and response to certain medication including steroids and calcineurin inhibitor [10,11]. Therefore, we initially administered steroid and tacrolimus to our patient. However, there was no response to these treatments. There are no specific immunohistopathologic findings and pathognomonic laboratory tests for PG. Histopathological examination of the ulcer may help to differentiate PG from some of its mimickers. When her skin biopsy findings is reassessed in keeping with the anticardiolipin antibody results, it was noticed by pathologist that her biopsy findings were suggestive a small vessel vasculopathy rather than vasculitis [12]. The exact pathomechanisms of the association of between SLE and PG are not thoroughly explained. However, the presence of antiphospholipid antibody suggests that mechanisms predisposition to vascular thromboses or infammation may also have contributed [13].
The therapeutic strategy in the general context of neutrophilic dermatosis as PG consists in modulating activation, maturation or migration of neutrophils (with colchicine, corticosteroids and immunosuppressive). Interestingly, drugs able to modulate connective tissue diseases activity like SLE (such as hydroxychloroquine, methotrexate, mycophenolate mofetil) were reported effective on neutrophilic skin lesions in the literature. However, the need for anticoagulation therapy and unresponsiveness to immunosuppressive only therapy confirmed also the diagnosis of SLE secondary development APS in our patient [14,15].
APS is an autoimmune disorder characterized by recurrent venous thrombosis or arterial occlusive events associated with elevated levels of antiphospholipid antibodies. APS may occur in association with SLE in childhood as well as in adults. The prevalence of antiphospholipid antibodies found to be higher in healthy children (5%–20%) [16]. However, it is difficult to predict the actual prevalence of APS in the pediatric population due to the absence of validated criteria for its diagnosis or classification [17]. The cutaneous manifestations of APS vary from livedo reticularis to cutaneous necrosis. Ulcers resembling PG have been described seldom in APS and may cause confusion in diagnosis. In addition, less than one third of patients with APS show histologic evidence of coagulopathy [18,19]. However, it can be said that APS is a highly problematic simulator of PG because of the low specificity of histologic findings in patients with APS and its frequent response to anticoagulation therapy [20]. It is important to have these information’s about APS to prevent misdiagnosis and delayed diagnosis.
SLE and APS are systemic autoimmune conditions which can present with a wide range of clinical manifestations related both to disease itself and to comorbidities. Based in clinical, pathological and response to proposed treatment, we can state that leg ulcers in patients with APS could be considered as a secondary form of PG. we recommend that testing for antiphospholipid antibodies should be performed in the initial evaluation of patients with SLE, and should be re-evaluated if new risk factors for thromboembolic events emerge.
Consent for publication was obtained from the patient’s mother.
None of the authors have any conflicts of interest or competing interests to declare for this specific project.
Citation: Gürgöze MK, Kara A, Sarı MY, Çalık I, Akarsu S (2021) Pyoderma Gangrenosum Mimicking Antiphospholipid Antibody Syndrome in a Child with Juvenile Systemic Lupus Erythematosus. Lupus: Open Access. 6:177.
Received: 17-Aug-2021 Accepted: 31-Aug-2021 Published: 07-Sep-2021 , DOI: 10.37421/2684-1630.2021.6.177
Copyright: © 2021 Gürgöze MK, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.