Research Article - (2017) Volume 3, Issue 2
Isolation and Identification of Bioactive Molecules Produced by Entomopathogenic bacteria, Acinetobacter calcoaceticus
*Corresponding Author: S. Remya Reghunath, Division of Crop Protection, Central Tuber Crops Research Institute, Thiruvananthapuram, Kerala, India, Tel: +918891068910 Email:
Abstract
Objective: To isolate and identify the bioactive metabolites produced by Acinetobacter calcoaceticus associated with entomopathogenic nematode, Steinernema sp.
Methods: In order to get maximum growth of bacteria and thereby maximum yield of organic extract optimization of basal media, temperature, pH and agitation speed was done. Effect of additional carbon source was also studied by doing fermentation using selected basal media at optimized conditions. Cell free culture obtained after fermentation is separated using ethyl acetate and the organic part is concentrated. Organic compounds were purified by column chromatography and identified using spectral techniques like HPLC, NMR and FTIR. Antimicrobial activity of the cell free extracts and organic compounds were tested by well diffusion technique.
Results: Cyclo (Pro-Tyr) and cyclo (Pro-Leu) were the compounds identified from organic extracts produced by Acinetobacter calcoaceticus, associated bacteria of entomopathogenic nematode, Steinernema sp. Cyclo (Pro-Tyr) showed antifungal activity against Candida albicans with 10 mm ZOI.
Conclusion: From the results it is possible to conclude that the entomopathogenic nematode and the associated bacteria could be promising source of bioactive compounds, and warrant further study. Significance and interest of study: The information obtained can be useful for commercial utilization.
Keywords: Entomopathogenic nematodes; EPN; Acinetobacter calcoaceticus; Antifungal; Steinernema
Introduction
Entomopathogenic nematodes are insect pathogenic nematodes and are the organisms considered for use in biological control. They have certain advantages over chemicals as control agents. Nematodes are non-polluting and thus environmentally safe and acceptable. Research into the use of entomopathogenic nematode as biocontrol agents has focussed mainly on the investigation of the families’ steinernematidae and heterorhabditidae. Entomopathogenic nematodes are highly virulent due to its symbiotically associated bacteria. The bacterial symbiont is Photorhabdus in nematodes of the genus Heterorhabditis and Xenorhabdus in nematodes of the genus Steinernema. In addition to Xenorhabdus and Photorhabdus sp. a novel entomopathogenic bacteria is isolated from a new entomopathogenic nematode, Rhabditis (Oschieus) sp., of the family Rhabditidae [1,2]. Entomopathogenic bacteria are potential source of antibacterial, antifungal, antiulcer, anti cancerous, insecticidal and nematicidal compounds, which might become promising pharmaceutical antibiotics or bio pesticides. EPB produce bioactive molecules which inhibit the growth of a wide range of bacteria, fungi and other microbes [3].
Entomopathogenic nematodes on entering the host insect, pathogenic bacteria are released by the nematode, and resulting in bacterial infection which causes the insect’s death [4,5]. The bacterial symbionts must perform three separate tasks to allow successful proliferation of the nematodes within the insect host, These are 1) to overcome insect immune defences and cause septicaemia and death, 2) to break down the tissues of the dead insects to release nutrients for the nematodes to proliferate and 3) to successfully re colonise the infective juvenile nematodes which will then be released [6].
Entomopathogenic bacteria encode a wide range of toxins that are exported by various secretion systems [7]. These toxins are involved in defense mutualism. A large number of novel genes are involved in the pathogenic and symbiosis of these organisms [8]. It was found that about 6% of genome is involved in secondary metabolite production [9] which is more than that of Streptomyces. Entomopathogenic nematodes are known for their antibacterial activity [10,11], antifungal activity [12-14], nematicidal activity [15,16], insecticidal activity [17] and cytotoxicity against various cell lines [18].
This study mainly aims at isolation and identification of bioactive molecules produced by entomopathogenic bacteria associated with a nematode strain NL among the collection of 65 entomopathogenic nematode isolates maintained at the CTCRI laboratory and its activity against fungi and bacteria.
Materials and Methods
Microorganisms and culture maintenance
EPN culture: The nematode isolate, NL (Accession No: CTCRI/ EPN/38) collected from the soil of Namakkal district, Tamil Nadu was selected for this study. It was taken from the nematode culture collection maintained in CTCRI laboratory.
Test pathogens
Test fungal pathogens were procured from the Microbial Type Culture Collection (MTCC), Institute of Microbial Technology (IMTECH), Chandigarh. The fungal strains are Fusarium oxysporum MTCC 284, Candida albicans MTCC 183, Rhizoctonium solani MTCC 4634, Aspergillus flavus MTCC 277 and Penicillium expansum MTCC 2006. Bacteria were maintained by sub culturing on nutrient agar and fungal strains were maintained on potato dextrose agar once in a month. Galleria mellonella larvae were obtained from the Department of Nematology, College of Agriculture, Thiruvananthapuram and were reared in CTCRI laboratory.
Culturing of EPN
Soil samples were processed with insect baiting method [19]. 250g soil sample was taken in a plastic bottle and baited with Galleria mellonella larvae. After two days dead larvae were placed in White Traps [20] and when there was emergence of infective juveniles (IJ’s), they were harvested. The IJs were collected in a beaker and rinsed 3 times with sterile distilled water and stored at room temperature. Nematode isolate was passed through G. mellonella every 6 months for sub culturing [21]. G. mellonella larvae were reared [22] by giving artificial diet.
Isolation of EPB
The entomopathogenic bacteria used in this study were isolated [23] from EPN belonging to the genus Steinernema, recovered from soil collected from Namakkal district (Tamil Nadu) and maintained at CTCRI. Bacterial isolate used in this study were taken from culture collection maintained in CTCRI laboratory.
Production of Bacterial Metabolites
Optimisation of media and cultural conditions
Optimisation of different factors like media, incubation period, temperature, agitation, pH and carbon source was done by one at a time method [24]. Growth characteristics were studied in three basal media namely Tryptic Soy Broth (TSB), Luria broth (LB) and Nutrient broth (NB) for various time intervals starting from 24 h to 120 h, at different temperatures (25, 30, 35, 40 and 45oC), at different agitation speed (50, 100 and 150 rpm) and at a range of pH from 5 to 9. To determine the effect of carbon sources on yield, different carbon sources such as glucose, fructose, maltose and sucrose were added to the preferred basal medium to give the total carbon concentration equal to 1%.
Fermentation
Seed culture preparation: A loop full fresh 24 h old culture from a newly sub cultured nutrient agar plate was inoculated into 100 ml broth in 250 ml flask which is incubated at 30°C at 150 rpm for 24 h. This was used to start fermentation.
Mass multiplication: Seed culture having an optical density of 1.5 at 600 nm was used to inoculate 400 ml fermentation broth in 1 L flask which is incubated in a shaking incubator at 30°C at 150 rpm for 24 h. Fermented culture was centrifuged in a cooling centrifuge at 10,000 rpm for 12 mts at 4ºC to collect the cell free culture filtrate.
Bacterial growth conditions like time, media, pH, aeration and temperature were optimised before starting fermentation. Influence of carbon source in fermentation was also studied by applying the same to preferred fermentation media.
Extraction of organic fraction: 500 ml cell free extract was transferred to a separation funnel to which equal volume of ethyl acetate was added. The funnel was agitated and kept for half an hour without disturbance for the separation of the organic and aqueous phases. Then organic phase was collected and stored at 4ºC. This process was repeated twice with the aqueous part. The organic fraction was concentrated by rotary flash evaporator at 30ºC.
Purification of the bacterial metabolites
Chromatography
Silica gel column chromatography
14 g of organic residue obtained from 35 L fermented culture was subjected to column chromatography using silica gel (230-400 mesh, Merck) column (60 × 3 cm) previously equilibrated with hexane. Organic solvents of different hydrophobicities (non-polar to polar) are used to elute column. Initially 100 ml of Hexane, linear gradient of 100 ml of Hexane/Dichloromethane (v/v 95:5 - 5:95), linear gradient of 100 ml Dichloromethane/Ethyl acetate (v/v 99:1 - 1:99) and finally column was eluted with 100% methanol. 25 ml elutes collected were concentrated using rotary flash evaporator, dissolved in methanol and stored at -20oC till further purification.
Thin Layer Chromatoraphy (TLC)
TLC was carried out using TLC aluminium sheets silica gel 60 F254 (Merck 5554). Standard chromatograms of organic crude extracts of TSB, LB and NB and concentrated fractions collected in coloumn chromatography were prepared by applying the same using capillary tube to a silica gel TLC plate. All the plates were developed with different solvent systems under saturated conditions to obtain optimum separation.
High Performance Liquid Chromatography (HPLC)
Sample was dissolved in HPLC grade methanol to get 1% solution and filtered through 0.45 μm membrane filter (Millipore). 15 μl of filtered sample was subjected to HPLC (C18 (Octadecyl silyl) column (5 μm × 4.6 × 250 mm) on an LC-10AT liquid chromatograph (LC shimadzu, Singapore) and run at a flow rate of 1ml/min) with UV detection at 210 nm using a photo diode array detector (Dionex) using methanol.
Gas Chromatograhy/Mass Spectrophotometry (GC/MS)
GC-MS analysis was performed using a Jeol GC MATE II mass spectrometer and injector MS transfer line temperature of 220°C, fused silica capillary column HP- 5 MS, carrier gas high pure Helium at a flow rate of 1 ml/min was used. The oven temperature was programmed from 50ºC to 250ºC @ 10ºC and electron impact mode at an ionizing voltage 70eV, scan range 40-600 amu. 1 ml of sample mixed with methanol (80%) at a split rate of 10:1 was injected. The compound identification was performed by comparing the GC relative retention and mass spectra to those of known substances analysed under the same conditions, by their retention time and by comparison to reference compounds using the database of National Institute Standard and Technology (NIST).
Structure elucidation (Spectral analysis)
UV- Visible Spectrophotometer
UV–vis spectrum of the isolated bioactive compound was recorded using Perkin Elmer Lamda 40 with UV-VIS Lab version 2.80.03 software and Systronics double beam spectrophotometer 2201, India using 1.0 cm quartz cuvette. The region from 200 to 800 nm was employed for scanning.
Fourier Transform Infra Red Spectroscopy (FTIR)
The FTIR spectroscopy was carried out using a Perkin Elmer FT-IR Spectrometer (Spectrum RXI). It was interfaced with a specac Golden Gate Diamond ATR system at room temperature. A region from 400 to 4000 cm−1 was used for scanning. Spectrum v 3.01 software was used for analysis and reporting. 1 mg (approximate) compound, after drying in a dessicator, was mixed with 100 mg of dry, powdered spectral grade potassium bromide (Merck) and ground using a mortar and pestle. A Perkin Elmer Bench Press set at a pressure of 1 × 104 kg/cm2 was then used to press the powdered mixture into a thin transparent disc.
Nuclear Magnetic Resonance (NMR)
1 H (400 MHz) and 13C (100 MHz) nuclear magnetic resonance (NMR) spectra were recorded DPX 300 MHz NMR spectrometer using deuterated chloroform (CDCl3) as solvent (Merck, Mumbai, India). The freeze dried sample was dissolved in the solvent and degassed by ultrasonic treatment for some minutes. The sample was kept in a 15 cm sample tube which was free of particles.
Mass spectrum (MS)
Accurate mass measurements were performed on a JEOL-JMS – SX/SX102A four sector tandem mass MS (JEOL Ltd, Tokyo, Japan) with a fast atom bombardment ion source operated in positive-ion mode. Glycerol was used as a sample matrix and the solvent was deuteriated DMSO (DMSO-d6). A small amount of sample was coated on the tip of the probe with the solvent. The molecular mass and possible fragment of the compound was obtained by analysing the spectrum.
Antimicrobial study
Antibacterial test: Antibacterial activity was studied by agar well and disc diffusion method (CLSI 1998). The test bacteria were cultured in nutrient broth at 37oC for 18 h and its concentration was adjusted to get a turbidity of 0.5 McFarland standards (1.5 × 108 CFU/ml). This culture was spread on Muller Hinton agar plate and wells (6 mm diameter) were cut on the plate. Sample (50 μl) was loaded in the well and allowed to diffuse in the agar media. After 18-24 h incubation at 37oC diameter of zone of inhibition (ZOI) was measured. Standard antibiotic disc, Ciprofloxacin (1 μg/ml, Himedia), was used as reference standard to determine the sensitivity of the strains. This was done in triplicates.
Antifungal test: Test fungi were swabbed on the potato dextrose agar plate and wells (6 mm diameter) were made. After loading the samples (50 μl) plate was incubated for 3-4 days at 25-28°C, and diameter of ZOI was measured to assess the antifungal activity (CLSI 1998) Bavistin and amphotericin (100 μg/ml) was used as reference standard to determine the sensitivity of the strains. The test was carried out in triplicates.
Statistical Analysis
Statistical analysis software (SPSS/version17.0 software) was used to evaluate nematicidal activity. Overall differences among means were tested using one-way analyses of variance (ANOVA). Duncan’s test was used to test significant differences among individual means if significant overall treatment effects were found at P<0.05.
Results
Isolation of EPB
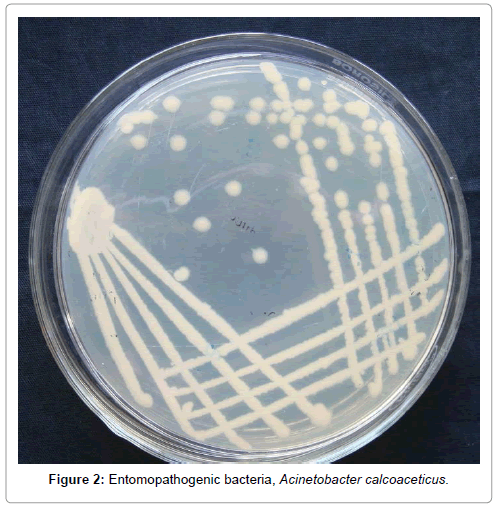
The entomopathogenic bacteria were isolated from the entomopathogenic nematode isolate collected from soil samples of Namakkal, Tamil Nadu (Figure 1). The bacteria were isolated on Nutrient agar medium and re isolated on NBTA medium. After incubation for 48 h on nutrient agar, single colonies were isolated which were appeared to be circular, slightly granular, irregular margin, opaque with 2-2.5 mm diameter (Figure 2). Only one type of bacterial colony found to absorb bromothymol blue from the NBTA plate and the colony appeared red in colour. The bacterial culture was maintained in nutrient agar by sub culturing. This bacterium was identified as Acinetobacter calcoaceticus (Accession no: NL JX470958) [25]. The nematode isolate NL was taken from the nematode culture collection maintained in CTCRI laboratory.
Extraction of organic crude
The results showed that all the three basal media, TSB, LB and NB showed maximum growth at 24 h fermentation. In order to find out the yield of organic crude extract at different fermentation time period organic fraction was separated and concentrated.
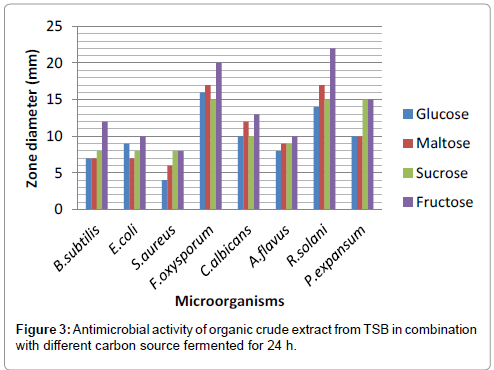
The cell free culture filtrates from TSB, LB and NB media at different fermentation time intervals were extracted with ethyl acetate as described in Materials and Methods. The ethyl acetate fraction was concentrated and weighed. Maximum yield was obtained with TSB (0.439 g) medium at 24 h followed by LB (0.382 g) and NB (0.315 g) per litre of the culture broth. Maximum yield was obtained with TSB in combination with fructose (0.479 g) per litre of the culture broth. These organic fractions were then assayed for antimicrobial activity.
Antimicrobial activity
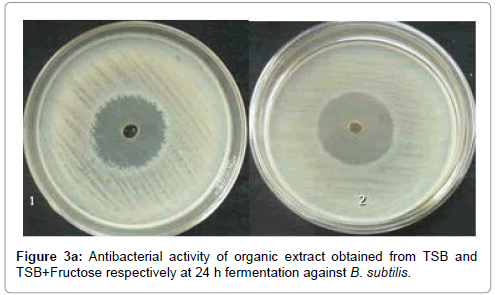
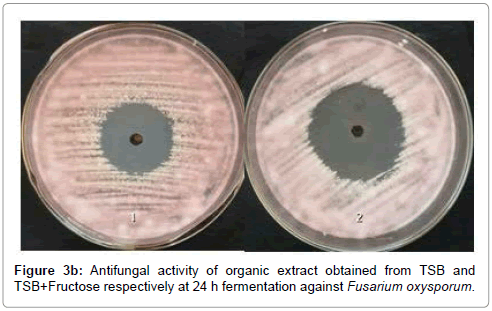
The concentrated organic extract of TSB showed maximum antibacterial activity against S. aureus, B. subtilis, E. coli and P. aerugenosa and antifungal activity against A. flavus, P. expansum, C. albicans, F. oxysporum and R. solani (Tables 1, 2 and Figures 3a-c).
| Incubation period (h) | Zone diameter (mm) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| B. subtilis | S. aureus | E. coli | P. aerugenosa | |||||||||
| TSB | LB | NB | TSB | LB | NB | TSB | LB | NB | TSB | LB | NB | |
| 24h | 10.93 ± 0.5a | 9.96 ± 0.5 b | 5.96 ± 0.15 c | 10.93 ± 0.11 a | 8.93 ± 0.05 b | 7 ± 0.05 c | 10.03 ± 0.05 a | 10 ± 0 b | 5.03 ± 0.15 c | 7 ± 0 a | 0.0 | 0.0 |
| 48h | 9.0 ± 0.2 a | 6.03 ± 0.05 b | 3.96 ± 0.15 c | 9.03 ± 0.05 a | 8 ± 0 b | 4 ± 0 c | 8.0 ± 0 a | 7 ± 0.10 b | 3.96 ± 0.15 c | 5 ± 0.10 a | 0.0 | 0.0 |
| 72h | 8.0 ± 0 a | 4.06 ± 0.05 b | 2.03 ± 0.05 c | 7.1 ± 0.10 a | 3 ± 0 b | 3.03 ± 0.05 c | 5.9 ± 0.10 a | 4 ± 0 b | 2.03 ± 0.05 c | 3 ± 0 a | 0.0 | 0.0 |
| 96h | 5.1 ± 0.1 a | 3 ± 0.20 b | 0.0 | 6 ± 0 a | 2 ± 0 b | 0.0 | 4.86 ± 0.11 a | 2.1 ± 0.10 b | 0.0 | 2.06 ± 0.05 a | 0.0 | 0.0 |
| 120h | 5.0 ± 0 a | 2.1 ± 0.10 b | 0.0 | 4.03 ± 0.05 a | 2.03 ± 0.05 b | 0.0 | 2.0 ± 0 a | 2.03 ± 0.15 b | 0.0 | 1 ± 0 a | 0.0 | 0.0 |
Table 1: Antibacterial activity of concentrated organic extract of basal media-TSB, LB and NB at different incubation period.
| Incubation period (h) | Zone diameter (mm) | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| F.oxysporum | C.albicans | A.flavus | R.solani | P.expansum | |||||||||||
| TSB | LB | NB | TSB | LB | NB | TSB | LB | NB | TSB | LB | NB | TSB | LB | NB | |
| 24h | 14.96 ± 0.15 a | 7.1 ± 0.1 b | 7 ± 0 c | 12.03 ± 0.5 a | 11.93 ± 0.11 b | 0.0 | 15.03 ± 0.05 a | 0.0 | 0.0 | 17.91 ± 0.1 a | 5.1 ± 0.1 b | 0.0 | 28.83 ± 0.15 a | 5.1 ± 0.10 b | 0.0 |
| 48h | 13 ± 0 a | 2 ± 0 b | 2.1 ± 0.10 c | 11.93 ± 0.11 a | 5 ± 0.1 b | 0.0 | 9 ± 0 a | 0.0 | 0.0 | 10.96 ± 0.5 a | 2 ± 0 b | 0.0 | 19.03 ± 0.05 a | 3 ± 0 b | 0.0 |
| 72h | 8.93 ± 0.11 a | 0.0 | 1.9 ± 0.15 c | 8.9 ± 0.10 a | 2 ± 0 b | 0.0 | 2.1 ± 0.10 a | 0.0 | 0.0 | 7.13 ± 0.15 a | 0.0 | 0.0 | 17.03 ± 0.15 a | 0.0 | 0.0 |
| 96h | 5.1 ± 0.10 a | 0.0 | 0.0 | 4.0 ± 0 a | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 5 ± 0 a | 0.0 | 0.0 |
| 120h | 5 ± 0 a | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 4.03 ± 0.05 a | 0.0 | |
Table 2: Antifungal activity of concentrated organic extract of basal media-TSB, LB and NB at different incubation period.
All assays were carried out in triplicate. Values represent the mean ± standard deviation. Values followed by the different letters in the same row are significantly different (p<0.05).
Purification of the Bacterial Metabolites
Column Chromatography
Since the ethyl acetate fraction of 24 h fermented TSB supplied with additional fructose showed higher antibacterial, antifungal and nematicidal activity than TSB medium, it was used for purification of bioactive compounds. Large scale fermentation from 30 L of culture broth yielded about 12 g of active oily organic crude extract which was loaded on to a silica gel column and chromatographed. The silica gel was packed into the column by mixing with the initial non-polar solvent Hexane. The column was washed in 100% pure hexane (100 ml), followed by 100 ml of linear gradient of hexane/DCM (v/v 95:5-5:95), 100% DCM, 100 ml of linear gradient Dichloromethane/Ethyl acetate (99:1-1:99), 100 ml 100% Ethyl acetate and finally 100% Methanol. 25 ml elutes from the coloumn were collected and concentrated in rotary vacuum evaporator at 28°C.
Thin Layer Chromatography (TLC)
Concentrated fractions were analysed by TLC (silica gel 60 F254). Solvent system for developing chromatogram has to be standardized for each coloumn fractions (Table 3). Chromatograms were observed under UV light (254 and 365 nm) and with iodine vapour in a saturated chamber. Fractions showing similar or same bands were combined, concentrated and stored at -20°C for further analysis like antimicrobial tests.
| Sl. No. | Fractions | Solvents system |
|---|---|---|
| 1 | Hexane | Benzene/Hexane |
| 2 | Dichloromethane/Hexane | Benzene/Hexane Acetone/Benzene |
| 3 | Dichloromethane | Acetone/Benzene |
| 4 | Ethylacetate/Dichloromethane | Acetone/Benzene |
| 5 | Methanol | Methanol/Chloroform |
Table 3: The solvent system standardized for TLC.
Antimicrobial assay of column fractions was done to find out active fractions and these fractions were further purified to identify the compound responsible for the activity (Table 4).
| Sl. No. | Active Fractions |
|---|---|
| 1 | 95% D/H |
| 2 | 10% E/D to 17% E/D |
| 3 | 18% E/D to 25% E/D |
| 4 | 28% E/D |
| 5 | 40% E/D |
| 6 | 50% E/D to 60% E/D |
| 7 | 61% E/D to 65% E/D |
| 8 | 70% E/D to 75% E/D |
Table 4: List of active fractions from coloumn chromatography.
Compound I
Solvent mixture of DCM-Hexane in the ratio 95% D/H yielded a yellowish powder (0.2 g). There were 6 bands in TLC of yellow powder. The yellowish powder thus obtained in 95% D/H was purified by washing in hexane followed by benzene. Then the powder became colour less and settled as pure crystals. The purified major compound’s Rf value was 0.4 (Figure 4).
TLC was developed on silica gel 60 F254 with 20% acetone/benzene as the solvent system.
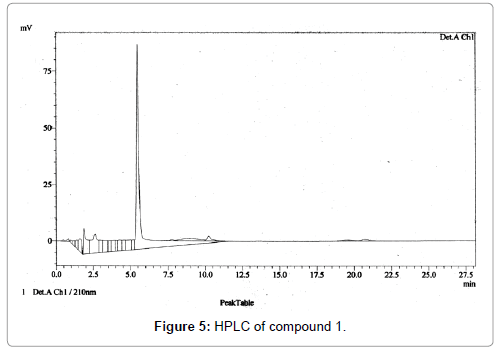
The HPLC was carried out in Silica gel C-18 reverse phase column with 100% methanol as the solvent at a flow rate of 1 ml/min with detection at 210 nm. The compound showed RT of 5.47 with 90.2% purity (Figure 5).
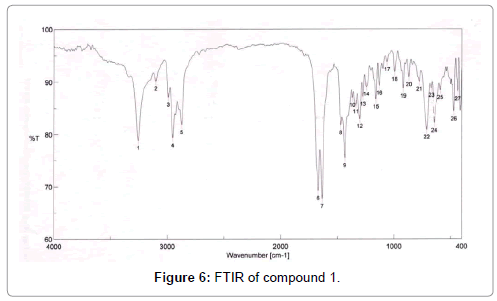
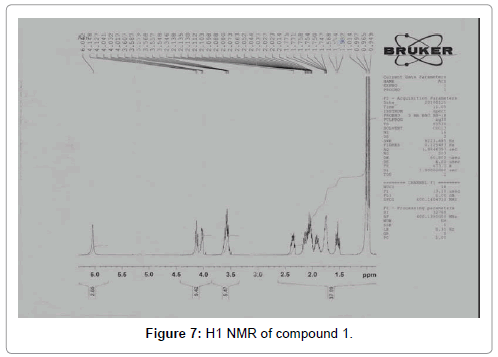
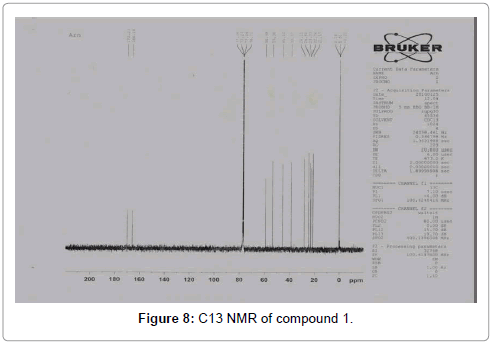
The pure compound was subjected to various spectral analysis like UV - visible spectrum, FTIR (Figure 6), NMR (Figures 7 and 8) and Mass spectrum and the structure was elucidated from spectral data [26].
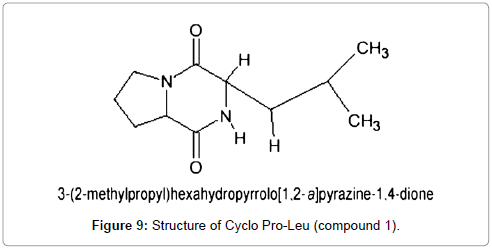
The absorbance of the sample was measured at wavelengths ranging from 200-800 nm, using a scanning uv-vis spectrophotometer. The maximum absorption was obtained at 210 nm. From the above results the compound was identified as Cyclo (Pro- Leu) (Figure 9).
Compound 1: Cyclo(Pro-Leu)-3-(2-methylpropyl) hexa hydropyrrolo [1,2-a] pyrazine-1,4-dione: A white amorphous powder; [a] D -139 (c, 0.02, MeOH); UV max : 210 nm (MeOH); 1HNMR (CDCl3, 500 MHz) δ: 0.965 (3H, d J=6.5 Hz Leu CH3); 1.014 (3H, d J =6.4 Hz Leu CH3); 1.568 (1H, m J=4.9 Hz/ 9.3 Hz/ -14.2 Hz Leu 3-Ha); 1.775 (1H, m, Leu 4-H CH); 1.91 (1H, m, Pro 4-Ha); 2.052 (1H, m, Pro 4-Hb); 2.073 (1H, m, Leu 3-Hb); 2.112 (1H, m, Pro 3-Ha); 2.346 (1H,m, Pro 3-Hb); 3.548- 3.607 (2H, m, Pro 5-Ha, b); 4.041 (1H, dd J=9.2 Hz/ 4.4 Hz Leu -H); 4.148 (1H, dt J=8 Hz/=1 Hz, Pro -H); 6.042 (1H, bs, NH). 13C NMR (CDCl3, 100 MHz) δ: 21.17, 22.76, 23.33, 24.68, 28.12, 38.57, 45.52, 53.36, 58.99, 166.18, 170.21.
Antimicrobial activity of compound 1: Even though 95%D/H fraction showed antibacterial and nematicidal activities the purified compound from the same fraction showed negligible biological activity.
Compound II: Crystals that settled down on storage of 40%E/D fraction at -20oC was purified by washing in hexane and benzene. This appeared as very pale yellow single spot on TLC developed against 70% acetone in benzene solvent system. Rf of this compound was 0.63 (Figure 10).
The absorbance of the sample was measured at wave lengths ranging from 200-800 nm, using a scanning uv-vis spectrophotometer. The maximum absorption was obtained at 275 nm.
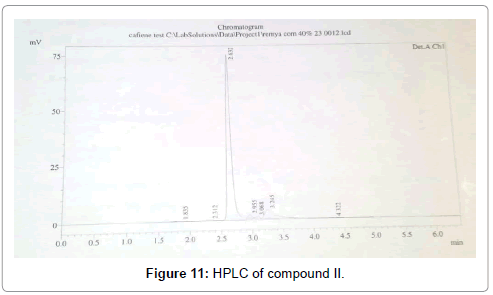
The HPLC was carried out in Silica gel C -18 reverse phase column with 100% methanol as the solvent at a flow rate of 1 ml/min with detection at 275 nm. The compound showed RT of 2.63 with 87.6% purity (Figure 11).
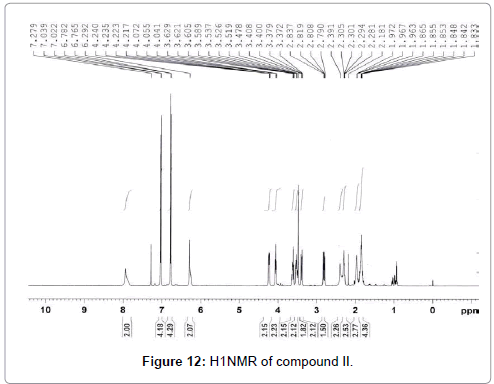
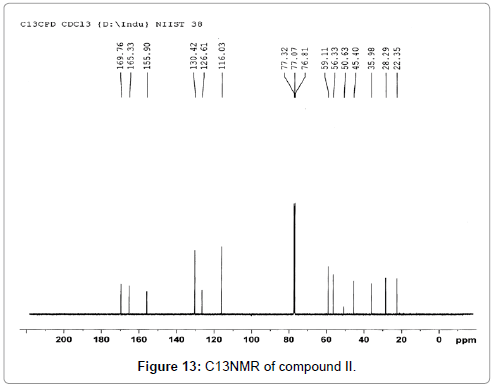
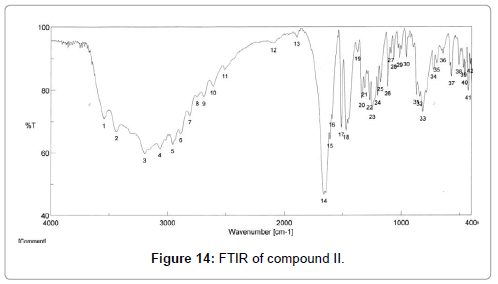
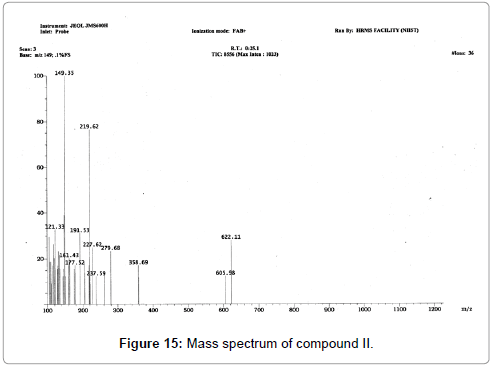
The pure compound was subjected to various spectral analysis like UV - visible spectrum, NMR (Figures 12 and 13), FTIR (Figure 14) and mass spectrum (Figure 15) and the structure was elucidated from spectral data.
Compound II
Cyclo-(Pro-Tyr), (3,8a) -3-(4-hydroxybenzyl) hexahydropyrrolo[1,2-a] pyrazine-1,4-dione: white amorphous powder; 1H NMR (CDCl3, 500 MHz) : δ 7.03 (2H, d, J =/8.5 Hz, H-12 and H-16), 6.78 (2H, d, J=8.5 Hz, H-13 and H-15), 4.21 (1H, m, H-3), 3.62 (1H, m, H-9a), 3.16 (1H, dd, J=/14.2 and 4.0 Hz, H- 10a), 3.13 (1H, m, H-9b), 2.81 (1H, dd, J=14.2 and 4.4 Hz, H-10b), 2.76 (1H, dd, J=6.1 and 2.1 Hz, H-6), 2.03 (1H, m, H-8a), 1.98 (1H, m, H-7a), 1.86 (2H, m, H-7b and H-8b) ; 13C NMR (CDCl3, 100 MHz) δ: 22.4, 28.3, 35.9, 40.4, 45.4, 56.3, 59.1, 116.1, 126.6, 130.4, 155.9, 165.3, 169.8. The molecular formula of the compound was determined to be C14H16N2O3 by FABMS (m/z 261.53 [M+H]).
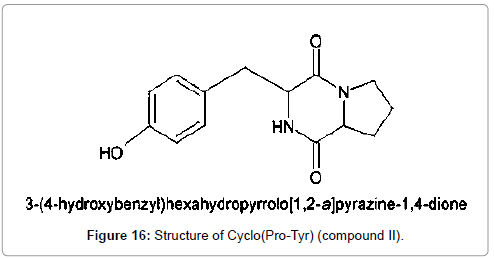
From the above results the compound is identified as Cyclo (Protyr) and the structure is given below (Figure 16).
Antimicrobial activity of compound II: This showed antibacterial activity with a zone diameter of >16 mm when tested against Mycobacterium tuberculosis H37Rv at Indian Institute of Integrative Medicine, Jammu and antifungal activity against C. albicans with a zone diameter of 10 mm at 500 μg/ml concentration.
Discussion
The entomopathogenic bacteria associated with the EPN of Steinernema spp., isolated from Namakkal district of Tamil Nadu was used in this study. The insecticidal activity of this nematode was proved in 24 h against Galleria mellonella larvae (in vitro). This bacterium was found to have 99% sequence similarity with Acinetobacter calcoaceticus (Accession no: NL (JX470958) [25]. This was done on the basis of morphological, biochemical and molecular characteristics. The 16s rDNA sequencing is the most trusted and widely used method in bacterial identification. It is demonstrated the occurrence of Acinetobacter sp and Enterococcus sp in Steinernema infected insect cadaver [24]. In that study it was also stated that Enterococcus sp. which originated in the intestine of insect got eliminated by the antimicrobials produced by Xenorhadus sp where as Acinetobacter sp came from the nematode survived along with Xenorhabdus. This type of dixenic association was also supported [27] for the presence of Photorhabdus and Providencia sp. in Heterorhabditis. Strong antimicrobial activity of crude organic extract was obtained from Acinetobacter sp. isolated from entomopathogenic nematode, Rhabditis Oscheius) [28].
It is reported diketopiperazine from Pseudomonas rhizosphaierae having antibacterial and antilarval activities [29]. Another study on diketopiperazines (DKPs) from a symbiotic bacteria associated with another Rhabditis (Oscheius) sp. of the CTCRI collection (NCBI Accession No: HQ200404) has been done [30]. The compounds are the diketopiperazines (DKPs) such as Cyclo(L-Pro-L-Leu), Cyclo(DPro- L-Leu), Cyclo(D-Pro-L-Tyr), Cyclo(L-Pro-L-Met), Cyclo(DPro- L-Phe), Cyclo(L-Pro-L-Phe), Cyclo(L-Pro-L-Tyr), Cyclo(L-Pro- D-Tyr), two stilbenes (3,4,5, trihydroxystilbene and 3,5, dihydroxy- 4-isopropyl stilbene), two depsides (atronorin and Usnic acid) and uracil. Among these, the diketopiperazines and stilbenes recorded significant antimicrobial activity and Cyclo (D-Pro-L-Tyr) inhibited the growth of A 375 (skin cancer), A 549 (lung cancer), MDAM B-231 (breast cancer) and HeLa (cervical) cell lines. In the present study cyclo (Pro-Tyr) showed anti-mycobacterial activity and very weak activity against Candida albicans while cyclo(Pro-Leu) exhibited negligible antimicrobial or nematicidal properties. Cyclo(Pro-Leu) obtained in this study may be inactive stereoisomers of the active cyclic dipeptides.
Acknowledgement
The authors are grateful to the directors of Central Tuber Crops Research Institute and Department of Science and Technology-Drugs & Pharmaceuticals Research programme for providing the facility and financial support respectively.
Conflict of Interest
From this study it was possible to identify a promising source of bioactive compounds. The information obtained can be useful for commercial utilization. There is no conflict of interest in this study.
References
- Mohandas C, Sheela MS, Mathews S, Naveen Raj DS (2004) Rhabditis (Oscheious) Spp (Nematoda:Rhabditidae) a new pathogenic nematode of crop pests. National Sympo. Green Pesticides for insect pest management 51-52.
- Deepa I, Mohandas C, Anupama PD, Siji JV, Benita Merlin I (2011) Diversity and identification of unique bacteria associated with novel entomopathogenic nematodes. Abstract volume of 23rd Kerala Science Congress 1-60.
- Khushbu Sharma, Suresh Walia, SudershanGanguli, AditiKundu (2016) Analytical characterization of secondary Metabolites from Indian Xenorhabdus Species the symbiotic bacteria of entomopatathogenic nematode (Steinernema spp.) as antifungal agent. National Academy Science Letters 1-6.
- Dillman AR, Sternberg PW (2012) Entomopathogenic nematodes. Current Biology 22: 430-431.
- Goodrich-Blair H, Clarke DJ (2007) Mutualism and pathogenesis in Xenorhabdus and Photorhabdus: two roads to the same destination. MolMicrobiol 64: 260-268.
- Zhang D, De Souza RF, Anantharaman V, Iyer LM, Aravind L (2012) Polymorphic toxin systems: Comprehensive characterization of trafficking modes, processing, mechanisms of action, immunity and ecology using comparative genomics. Biol Direct 7: 18.
- Ullah I, Jang EK, Kim MS, Shin JH, Park GS, et al. (2014) Identification and Characterization of the Insecticidal Toxin “Makes Caterpillars Floppy” in Photorhabdustemperata M1021 Using a Cosmid Library. Toxins (Basel) 6: 2024-2040.
- Duchaud E, Rusniok C, Frangeul L, Buchrieser C, Givaudan A, et al. (2003) The genome sequence of the entomopathogenic bacterium Photorhabdusluminescens. Nat Biotechnol 21: 1307-1313.
- Akhurst RJ (1982) Antibiotic activity of Xenorhabdus spp., bacteria symbiotically associated with insect pathogenic nematodes of the families 546Heterorhabditidae and Steinernematidae. J of Gen Microbiol 128: 3061-3065.
- ErzsébetBurgettinéBöszörményi, IstvánBarcs, GyulaDomján, KatalinBélafinéBakó, AndrásFodor, et al. (2015) Xenorhabdusbudapestensisentomopathogenic bacteria cell free conditioned medium and purified peptide fraction effect on some zoonotic bacteria. Akadémiai Journals 156: 1782-1786.
- Barbercheck ME, Kaya HK (1990) Interaction between Beauveriabassiana and the entomopathogenic nematodes, Steinernemafeltiae and Heterorhabditisheliothidis. J InvertebrPathol 55: 225-234.
- Chen G, Dunphy GB, Webster JM (1994) Antifungal activity of two Xenorhabdus species and Photorhabdusluminescens species of Heterorhabditismegidis. Biological Control 4: 157-162.
- Xiangling Fang, Manrang Zhang, Qian Tang, Yonghong Wang, Xing Zhang (2014) Inhibitory effect of Xenorhabdusnematophila TB on plant pathogens Phytophthoracapsici and Botrytis cinerea in vitro and in planta. Scientific Reports 4: 1-7.
- Han RC, Ehlers RU (1999) Trans-specific nematicidal activity of Photorhabdusluminescens. Nematology 1: 687-693.
- Hu K, Jianxiong L, Webster JM (1999) Nematicidal metabolites produced by Photorhabdusluminescens (Enterobacteriaceae), bacterial symbiont of entomopathogenic nematodes. Nematol 1: 457-469.
- Bowen D, Rocheleau TA, Blackburn M, Andreev O, Golubeva E, et al. (1998) Insecticidal toxins from the bacterium Photorhabdusluminescens. Science 280: 2129-2132.
- Webster J (2002) Bacterial metabolites. In Gaugler, R. (ed.) Entomopathogenic nematology. CABI Publishing, Wallingford, United Kingdom 99-114.
- Bedding RA, Akhurst RJ (1975) A simple technique for the detection of insect paristicrhabditid nematodes in soil. Nematologica 21: 109-110.
- White GF (1927) A method for obtaining infective nematode larvae from cultures. Science 66: 302-303.
- Rosa JS, Bonifassi E, Amaral J, Lacey LA, Simoes N, et al. (2000) Natural Occurrence of Entomopathogenic Nematodes (Rhabditida: Steinernema, Heterorhabditis) in the Azores. J Nematol 32: 215 -222.
- Woodring JL, Kaya HK (1988) Steinernematid and Heterorhabditid nematodes: Hand book of biology and techniques. South cooperative Ser. Bulletin Arkans. Agricultural ExpStn., Fayetteville 331: 1-30.
- Park SH, Yu SU (1999) Isolation and identification of a symbiotic bacterium from Steinernemacarpocapsae. Biotechnology and Bioprocess Engineering 4: 12-16.
- Walsh KT, Webster JM (2003) Interaction of microbial populations in Steinernema (Steinernematidae, Nematoda) infected Galleria mellonella larvae. J InvertebrPathol 83: 118-126.
- Anju KM (2015) Biological properties of metabolites of symbiotic bacteria associated with novel Rhabditidentomopathogenic nematode. PhD Thesis, University of Kerala 1-89.
- Jackson TJ, Wang H, Nugent MJ, Griffin CT, Burnell AM, et al. (1995) Isolation of insect pathogenic bacteria, Providenciarettgeri, from Heterorhabditis spp. J ApplBacteriol 78: 237-244.
- Deepa I, Nishanth Kumar S, Sreerag RS, Mohandas C (2014) Antimicrobial activity of crude extract obtained from an Acinetobacter sp. associated with an entomopathogenic nematode. IJPDA 2: 649-652.
- Qi SH, Xu Y, Jun GAO, Qian PY, Zhang S, et al. (2009) AntibacterialandantilarvalcompoundsfrommarinebacteriumPseudomonas rhizosphaerae. Annals of Microbiology 5: 229-233.
- Nishanth Kumar S, Mohandas C, Siji JV, Rajasekharan KN, Nambisan B (2012) Identification of antimicrobial compound, diketopiperazines, from a Bacillus sp. N strain associated with a rhabditidentomopathogenic nematode against major plant pathogenic fungi. J ApplMicrobiol 113: 914-24.
- CLSI (2008) Reference methods for broth dilution. Antifungal susceptibility tests for yeasts. CLSI documents M-27-S3.940 West Valley Road,Suite 1400, Wayne, Pennysylvania 19087-1998. USA.
Copyright: © 2017 Reghunath SR, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.