Journal of Hematology & Thromboembolic Diseases
Open Access
ISSN: 2329-8790
ISSN: 2329-8790
Case Report - (2021)Volume 9, Issue 7
Thrombotic thrombocytopenic purpura (TTP) is defined as thrombotic microangiopathy occurring in the context of severe ADAMTS13 deficiency (<10%) which is a normal enzyme in plasma that cleaves large forms of von Willebrand factor into smaller functional subunits. We report a case of a middle aged male with de novo iTTP ( idiopathic thrombotic thrombocytopenic purpura) after vaccination with ChAdOx1 nCov-19. He presented with severe abdominal pain, increased bruising, hematuria along with episodes of transient ischemic attacks, 5 days from the first dose of the antiCOVID-19 vaccine. Laboratory data revealed a very severe ADAMTS13 deficiency, however, autoantibody titer could not be measured due to the failure of refrigerating facility in our reference laboratory. Patient was successfully treated with serial plasma exchange and immunosuppression with steroids and rituximab.
Thrombotic Microangiopathy; Post vaccination; COVID-19; ADAMTS 13; Plsama exchange
Thrombotic microangiopathies characterized by microangiopathic hemolytic anemia, thrombocytopenia and microthrombi can be primary or secondary. TTP can be potentially left threatening if left untreated. Thrombotic thrombocytopenic purpura is defined as a thrombotic microangiopathy occurring in the context of severe ADAMTS13 deficiency (<10%) which is a normal enzyme in plasma that cleaves large forms of von Willebrand factor into smaller functional subunits [1]. It can result in the rare development of immune thrombotic thrombocytopenia mediated by platelet-activating antibodies against PF4, which clinically mimics autoimmune heparin-induced thrombocytopenia and should be considered in the workup of patients presenting with thrombotic microangiopathies in this Covid era [2].
A middle aged male presented with complaints with severe abdominal pain, increased bruising, hematuria along with episodes of transient ischemic attacks, starting 5 days from the first dose of the ChAdOx1 nCov-19. Routine investigations showed severe hrombocytopenia, anemia with raised reticulocyte count, raised LDH and hyperbilirubinemia along with elevated creatinine. Peripheral blood smear showed normochromic anemia with 1-2% schistocytes at admission which later increased to 4-5% (Figure 1). Neuro-imaging studies and EEG were within normal limits. In view progressive neurological worsening in the form of recurrent seizure episodes and altered sensorium along with worsening hemolytic anemia and renal dysfunction, patient was started on steroid pulse with methyl prednisolone for 5 days followed with oral steroids with daily plasma exchange sessions on clinical background of rapidly evolving thrombotic microangiopathy. More detailed work up showed severe ADAMTS13 deficiency with undetectable levels of ADAMTS13 enzyme activity confirming the clinical suspicion. Antibodies to platelet factor 4(PF4)-heparin were negative, using HIT testing which essentially ruled out vaccine – induced immune thrombocytopenia. Anti-SARS-Cov-2 polymerase chain reaction on nasopharyngeal swabs were negative at admission.
The patient was initiated on daily plasma exchange FFP; 1.5X total plasma volume] with methylprednisolone pulse (1000 mg) for 5 consecutive days followed by tapering steroids.
The patient had transient clinical improvement followed by clinical worsening in the form of status epilepticus which necessitated intubation and mechanical ventilation along with 3 anti-epileptic agents to control his seizure activity. The patient was also initiated on weekly rituximab (375 mg/m2) x 4 doses. This led to a better control of the disease activity and platelet counts were stable after 11 sessions of plasma exchange as described in the Figures 1-3. Patient was initiated on low molecular weight heparin and ecosprin following the recovery of the platelet count. The patient had developed swelling and pain over right forearm. Sonogram of right forearm showed possible multiple deep muscular hematomas in the fore-arm with surrounding edema. Antithrombotic and anticoagulation therapy was deferred and managed with conservative measures.
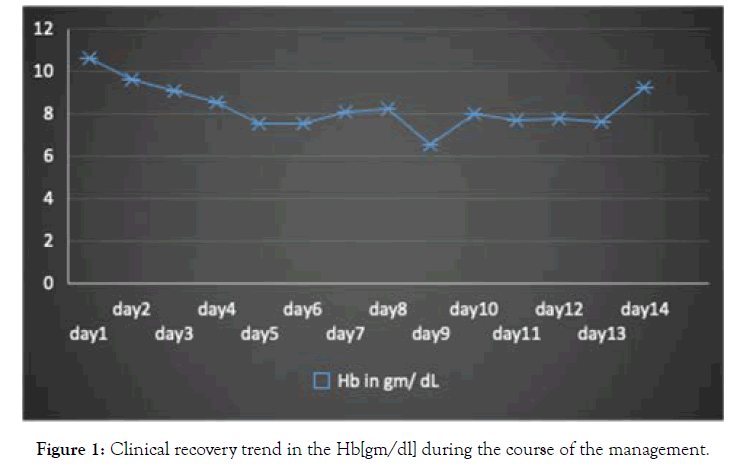
Figure 1. Clinical recovery trend in the Hb[gm/dl] during the course of the management.
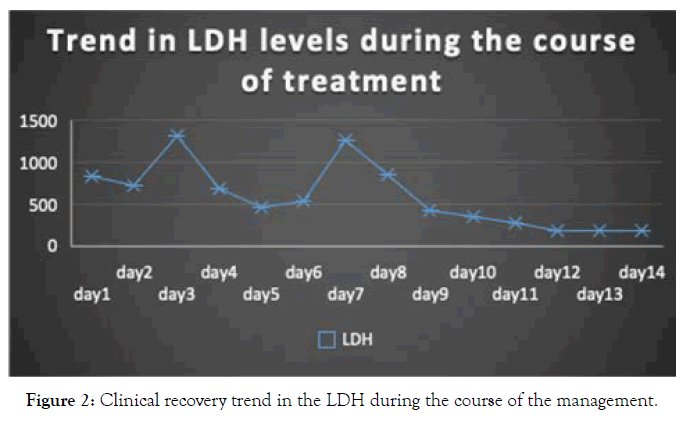
Figure 2. Clinical recovery trend in the LDH during the course of the management.
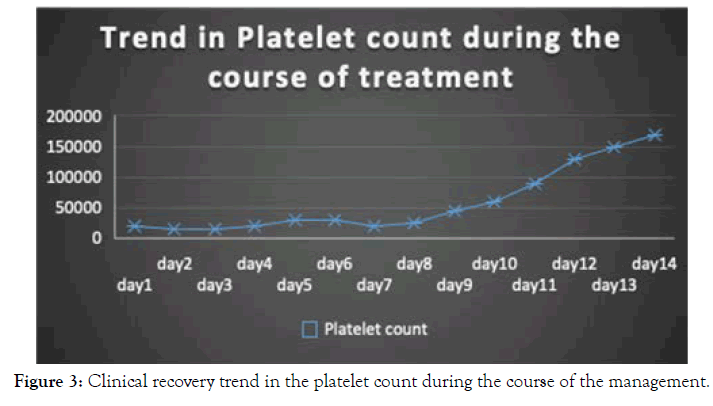
Figure 3. Clinical recovery trend in the platelet count during the course of the management.
Thrombotic microangiopathies are a group of rare life-threatening hematologic conditions that require urgent management [1]. Though presenting symptoms may be nonspecific, basic laboratory tests revealing thrombocytopenia accompanied by anemia with evidence of hemolysis and red blood cell fragmentation leads to the diagnosis. Microangiopathic hemolytic anemia, the hallmark of thrombotic microangiopathy is a process of red blood cell destruction within the microvasculature associated with thrombocytopenia due to platelet activation and consumption. Thrombotic thrombocytopenic purpura (TTP) and hemolytic uremic syndrome (HUS) are two distinct forms of thrombotic microangiopathies [3]. Thrombotic thrombocytopenic purpura is caused by deficiency of the enzyme ADAMTS13 (a disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13) which can occur either due to acquired autoantibodies or rarely due to genetic mutations [4]. Primary forms occur spontaneously with no associated underlying cause whereas secondary forms occur in the context of various precipitating factors like pregnancy, autoimmune disease and medications [5]. Once thrombotic microangiopathy is suspected, a thorough workup for secondary causes is important to help guide treatment.
There have been several reports of unusual thrombotic events and thrombocytopenia following vaccination with the recombinant adenoviral vector encoding the spike protein antigen of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) (ChAdOx1 nCov-19, AstraZeneca) [2]. Most of these cases were associated with high levels of antibodies to platelet factor 4 (PF4)– polyanion complexes similar to heparin-induced thrombocytopenia and is referred to as vaccine-induced immune thrombotic thrombocytopenia (VITT) [6].
There were no precipitating factors other than the history of vaccination in our case. Absence of anti-platelet antibodies raises the possibility of another pathogenetic mechanism for the development of thrombotic events post vaccination.
VITT is a well-established rare complication of ChAdOx1 nCov-19 vaccination. Further studies are required to consider the possibility of alternative thrombotic mechanisms post vaccination.
Authors have no conflict of interests.
Nil
MP, DV, SMP, LA, JV, JT contributed to study design. MP, LA contributed to data retrieval. MP and LA contributed to laboratory analysis. MP and LA contributed to analysis of data and interpretation of the results. MP and LA contributed to drafting of the manuscript. All authors contributed to writing and final approval of the manuscript.
Citation: Paul M, Abraham L, Sophy, Varghese D, Thomas J, et al. (2021) Idiopathic Thrombotic Microangiopathy with ChAdOx1 nCov-19 Vaccination in a Middle Aged Male: A Clinical Challenge in the Covid Era. J Hematol Thrombo Dis 9:431. DOI: 10.24105/2329-8790.2021.9.441
Received: 04-Jul-2021 Accepted: 19-Jul-2021 Published: 26-Jul-2021 , DOI: 10.35248/2329-8790.21.9.441
Copyright: © 2021 Paul M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.