Journal of Oceanography and Marine Research
Open Access
ISSN: 2572-3103
ISSN: 2572-3103
Research Article - (2013) Volume 1, Issue 3
Microalgae have the most significant contribution towards the global environment, oxygen production and carbon cycle consequently understanding marine microalgae is vital. Hence the factors that affect the growth should be considered and studied in advance before the culturing of these algae. Photosynthesis is most important in plant growth. Thus this study tries to find out the optimum light intensity (Lux) required for achieving maximum yield of Chaetoceros muelleri in a laboratory culture. The inoculation of the algae was done under controlled environment where the cells were counted and assumed to be the same for all. The pure strand of C. muelleri with f2 media (8 sets of 3 flasks) were placed in front of the light source at lux values of 0 lux, 500 lux, 1000 lux, 1500 lux, 2000 lux and 2500 lux. The cell count was the basic result that was collected. At every 24 hours cells were counted and readings were recorded. The experiment lasted 7 days. The result showed that there was significant growth that is the number of cells increased exponentially as the Light intensity (lux) increased till the culture was exposed to a 1000 lux where maximum cells were counted during the culture period (168 hours). From previous studies it was evident that the optimum light intensity for flask culture in laboratory was 1000 lux. However, results from the experiment show that the cell growth with 1500 lux is very close and similar to 1000 lux. Some recommendations after the experiment are; that there is very little literature on the above topic and that this area requires a lot more attention as Fiji’s Aquaculture Industries are blooming thus there is a need for further research and study around the same area.
<Keywords: Micro algae; Lux; Chaetoceros, Chaetoceros muelleri
Microalgae are defined as minute photosynthetic plants and can be found in both seawater and freshwater [1]. These have similar properties as land based plants but they have to be submerged in an aqueous environment where they have efficient access to water, carbon dioxide and other nutrients.
Nowadays, there are several marketable applications of microalgae some of which include; (i) used to enhance the nourishing value of food and animal feed owing to their chemical composition, (ii) they play a crucial role in aquaculture and (iii) they can be assimilated into cosmetics. Moreover, they are cultivated as a source of highly valuable molecules [2]. Microalgae is also used in the production of pharmaceuticals, diet supplements, pigments and biofuel [3] and represent the largest, yet one of the most poorly understood groups of microorganisms on earth [4]. A handful of microalgae species out of the many that have been found are mainly cultured for live aquaculture feed and Chaetoceros muelleri [5] is one of them.
The hypothesis for this experiment is to find whether light is required to culture Chaetoceros muelleri. Secondly to examine the amount of light required to give the maximum yield of the product. Also show the relationship between the amount of light required and the growth rate of Chaetoceros muelleri.
Microalgae culture has to be conducted under specialised conditions as explained by Coutteau [6] the most important parameters regulating algal growth are nutrient quantity and quality, light, pH, turbulence, salinity and temperature. Concentrations of cells in phytoplankton cultures are generally higher than those found in nature. Algal cultures must therefore be enriched with nutrients to make up for the deficiencies in the seawater. Macronutrients include nitrate, phosphate and silicate. Here the paper describes the General Set for culturing Micro algae: Temperature 16-27°C, Salinity 12–40 g/l and Light Intensity (lux) 1,000 – 10,000 lux depending on the volume of culture.
As with all plants, micro-algae photosynthesize, i.e. they assimilate inorganic carbon for transformation into carbon-based matter. Light is the source of energy which drives this reaction and in this regard intensity, spectral quality and photoperiod need to be considered. Light intensity plays an vital role, but the necessities fluctuate critically with the culture penetration and the density of the algal culture: at higher depths and cell concentrations the light intensity must be increased to penetrate through the culture 1,000 lux is suitable for Erlenmeyer flasks, 5,000-10,000 lux is required for larger volumes [6]. Light may be natural or supplied by fluorescent tubes. Too high light intensity (e.g. direct sun light, small container close to artificial light) may result in photo-inhibition. Also, overheating due to both natural and artificial illumination should be avoided. The duration of artificial illumination should be minimum 18 h of light per day, although cultivated phytoplankton develops normally under constant illumination [7].
Chaetoceros is probably the largest genus of marine planktonic diatoms with approximately 400 species described. Although a large number of these descriptions are no longer valid. It is often very difficult to distinguish between different Chaetoceros species. Several attempts have been made to restructure this large genus into subgenera and this work is still in progress. However, most of the effort to describe species have been focused in boreal areas, and the genus is cosmopolitan [7], so there are probably a large number of tropical species still undescribed.
Scientific Classification of Chaetoceros muelleri
Empire Eukaryota
Kingdom Chromista
Subkingdom Harosa
Infrakingdom Heterokonta
Phylum Ochrophyta
Subphylum Khakista
Class Bacillariophyceae
Subclass Coscinodiscophycidae
Superorder Chaetocerotanae
Order Chaetocerotales
Family Chaetocerotaceae
Genus Chaetoceros
In the past there have been very few studies done on the relationship between the lux and the growth rate of Chaetoceros muelleri. As stated by Blinn [8] respiration and photosynthesis are the two significant processes in microalgal growth that occur simultaneously in the light. To know the rates of both processes, at least one of them has to be measured. To be able to measure the rate of light respiration of Chlorella sorokiniana, the measurement of oxygen uptake must be fast, preferably in the order of minutes. This experiment looks deep into the process of Respiration where the amount of Oxygen uptake in the situ. The respiration rate is known to be related to the growth rate, and it is suggested that faster algal growth leads to a higher energy requirement, and as such, respiratory activity increases [8].
And as stated by Brown [9] equal inocula were added in the two bioreactors (1 and 3 cm light path); by the end of the second day, cell concentration was low in the 1 cm light path bioreactor, possibly indicating photo inhibition, since growth of the culture increased in the 3 cm light path bioreactor.. The optimal population density was obtained by a daily harvest of 5% of culture volume to be 2.39 g m (–2) day (–1) in 1 cm light path and 3.27 g m (–20) day (–1) in 3 cm light path. Light path length of 3 cm was found optimal in both with low and high initial cell densities [10].
As noted by the previous studies there has not been much research done on Chaetoceros muelleri in regards to the light and growth of the microalgae. Plus this research proposals will be one of the pioneer projects that will consider the relationship for the two variables in the labs as small experiment, from there more research can be carried out in order to use the investigate the intensity of light required for the maximum growth of Chaetoceros muelleri. Hence there is more attention required for the above pioneer project to be carried out successfully.
The experiment was carried out in the laboratory of the School of Marine Studies, University of the South Pacific (USP), Fiji, Suva.
A pure 20 ml sample of C. muelleri was purchased from Australian National Algae Culture Collection (ANACC), CSIRO in Tasmania and Australia with the assistance of the USP Laboratory Technician.
The glassware used in this experiment was cleaned and washed with detergent and autoclaved on programme A (121°C and 20 psi) as provided on the autoclave for 15 minutes.
Seawater was strained using 1µm (micro meter) cartridge filter and used for the preparation of the Media and Stock solution. After filtration the water was passed a Life Guard brand Ultra Violet Steriliser (Model no. – QL 40) that uses a 40 watt UV sterilizer bulb. All the above was done in the Aquaculture Laboratory (Wet Lab).
The procedures used to prepare the F/2 Media and also the Modification of F/2 media were followed from [11]. 75 g of NaNO3 and 5 g of NaH2PO4 H2O were dissolved in 1 litre of filtered sea water and this was labelled as Solution A. Trace Metals was labelled Solution B, Silicates was labelled solution C. 1 ml of solution B and C was used for every litre of media. Vitamins were labelled Solution D and 0.5 ml of this solution was used for every litre of media.
Inoculation
A sample of Pure C. muelleri is placed in a controlled environment for it to grow or reproduce. For this experiment the procedure used was adapted from Probert and Klaas [12] was followed. 100 ml of the f/2 media that was prepared earlier was filled into 250 ml Conical culture flasks which were covered using corks and aluminium foil. The prepared flasks were autoclaved under Programme A. After the Autoclave the flasks were removed and left overnight to cool in the algae room. All the flasks were inoculated with 10% of inoculum (C. muelleri) in the Laminar Flow cupboard. The cabinet was cleaned with 70% ethanol. The pippttes and micro pipettes were also autoclaved [13]. The Nitrile hand gloves used are to be sprayed with 70% ethanol and will be worn while inoculating. The mouth of the culture flask was sterilised by moving over a Bunsen burner flame.
After the inoculation the rubber corks were bored to make holes and the glass tubing were inserted for the inlet (3-4 mm) and exhaust (2-3 mm) were placed to seal the mouth. The above setup was prepared in the laminar flow cabinet to avoid contamination. Once the inlet and exhaust lines are fixed the flasks were ready for setup in the algae room. Some of the parameters that were kept constant were; temperature at 18°C, preferred pH of 7-9 with reference to FAO, Instrumental Paper, 1999, salinity was between 20-35 ppt, Aeration was controlled by opening or closing the switch valves linked to the inlet line and the cell starter culture the cells were calculated in terms of cells/ml. the inoculation required 10% of the starter culture. During the preparation of all the twelve flasks efforts were made to ensure that approximately the same number of cells is transferred.
The variable in the experiment is the lux that is a standardized unit of measurement of the light intensity (which can also be called “illuminance” or “illumination”) - as an example for reference purposes. One lux is equal to one lumen per square metre [14]
1 lx = 1 lm/m2 = 1 cd·sr·m−2
The Flasks were placed at 500 lux, 1000 lux and at 1500 lux from the light source. The Growth performance was measured in terms of cell density/ml.
Eighteen trails were carried out that is six treatments with three replicates of each. The six treatments were based on the different lux; 0 lux, 500 lux, 1000 lux, 1500 lux, 2000 lux and 2500 lux. These were represented as treatment A, B, C, D, E and F respectively.
Sampling
Hemocytometer was used to count the cells [2]. From the time the inoculation was done the counting was done at every 24 hour time interval until the culture reached a stationary stage. From every flask a sample of 1 ml was taken and average of 3 counts were done for better result analysis. The experiment was repeated two more times to confirm and reconfirm results that were obtained in the first set.
Statistical analysis
The statistical analysis that was adapted by this experiment was SPSS. This showed the relationship between the amount of flux and the growth rate at different lux rates. The comparison of the differences of the treatments with the control was done by the Duncan’s Test.
Over the period of the experiment the results were collected and tabulated as Replicate 1, Replicate 2 and Replicate 3 (Tables in Appendix) from this an average data was calculated as shown in Table 1. The table shows the average of all the three Replicates that was done. The values at the different lux (0 lux, 500 lux, 1000 lux, 1500 lux, 2000 lux and 2500 lux) were added and divided by 3.
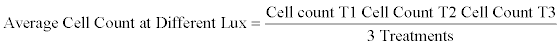
More over a Graph was constructed using the Table 1, in order to see the relationship between the Cell counts at different lux with the time of each count.
| AVERAGE CELL COUNT | ||||||
|---|---|---|---|---|---|---|
| Cell Count (x10 ^6) | ||||||
| Time (Hrs) | 0 LUX | 500LUX | 1000LUX | 1500 LUX | 2000Lux | 2500Lux |
| 0 | 0.2 | 0.296667 | 0.2 | 0.2 | 0.2 | 0.2 |
| 24 | 0.126667 | 0.723333 | 1.14 | 1.04 | 0.95 | 0.78 |
| 48 | 0.03 | 1.463333 | 2.28 | 1.6333 | 1.636667 | 1.42 |
| 72 | 0.005667 | 2.416667 | 3.41 | 2.8033 | 2.586667 | 1.973333 |
| 96 | 0 | 2.876667 | 3.776667 | 3.2433 | 2.873333 | 2.686667 |
| 120 | 0 | 3.196667 | 3.606667 | 3.0667 | 2.803333 | 2.973333 |
| 144 | 0 | 2.826667 | 3.596667 | 3.1133 | 2.823333 | 2.863333 |
| 168 | 0 | 2.483333 | 3.063333 | 2.9233 | 2.74 | 2.776667 |
Table 1: Shows the average cell count for the three treatments.
According to the average growth rate of the C. muelleri, the graph states that the highest cell count is at 1000 lux. From the 0hrs at stated lux the cell count increased gradually. The maximum growth is shown at 1000 lux around 96 hours. As indicated by the graph that a maximum growth is reached than the slows and starts to decrease. This graph indicates the phases exponential phase (Growth phase), Stationary Phase (Maximum point reached) than the Death Phase (No more growth). At all the different lux (0 lux, 500 lux, 1000 lux, 1500 lux, 2000 lux and 2500 lux) there is a similar trend seen. The control (0lux) showded little growth initially, than the cell count decreased to zero.
Moreover from the SPSS Statistics that was carried out indicated that There was a significant (df5, F298.54, P0.00) difference between 6 treatments (0 lux, 500 lux, 1500 lux, 2000 lux and 2500 lux). 1000 lux was significantly higher than the control (p 0.00), 2000 lux (p0.00) and 2500 lux (p0.00), however it was not significantly different from 500 lux (p0.899) and 1500 lux (p0.221). Thus it can be noticed that 1000 lux is more similar to 1500 lux.
The purpose if this experiment was to better understand the importance of light intensity in the growth of the C. muelleri as the time and the intensity was increased. The experiment was modelled in such a way that at different lux from a minimum to a maximum lux just to compare the cell count to observe the Cell Growth, the control was also set up to witness if there will be any growth at zero light intensity. The purpose of having the different lux setup for the experiment was to find out the best light intensity required for the Flask culture of C. muelleri in a controlled Laboratory.
The main result of the experiment showed that growth rate varied at different Light intensity (Lux). C. muelleri can be cultured in a laboratory at different light intensity but according to the results the best amount of light for the maximum yield is 1000 lux, 500 lux and also 1500 lux are also similar but lower growth rate seen than 1000 lux.
The experiment results is supported by Cotteau [6] where he stated that Light intensity played an important role, but the requirements varied greatly with the culture depth and the density of the algal culture: at higher depths and cell concentrations the light intensity must be increased to penetrate through the culture (e.g. 1,000 lux is suitable for erlenmeyer flasks, 5,000-10,000 is required for larger volumes). Too high light intensity (e.g. direct sun light, small container close to artificial light) may result in photo-inhibition. This may cause overheating due to both natural and artificial illumination [15]. Thus in this experiment states that the higher the lux the slower the growth rate at around 2000 lux and 2500 lux the growth fast initially that it slows; there might be few factors affecting this one could be the light intensity too high for the cells to tolerate, or the high population increase might decrease the nutrients thus slow growth rate. Hence this indicates that too much light is not suitable for the growth of C. muelleri. Comparing this to 500 lux; this also has slow growth rate; thus only conclusion that can be drawn is less light intensity induces slow growth rate, as the flask has enough nutrients, light was the only factor that was low.
Naturally not only Light is required for the microalgae growth, there are other factors that affect the growth rate of C. muelleri which include right salinity, right pH, right amount of nutrients and vitamins [16]. The term nutrient “limitation” has been widely used in the literature in a variety of contexts [17], often inter-changeably with the terms “deficiency” and “starvation.” There are two distinctively different types of limitation: one referring to limitation of the maximum population density achieved (Liebig limitation) and one referring to limitation of growth rates
From the earlier research’s carried out for the laboratory culture of C. muelleri, the light intensity required should be 1000 lux and the results for this experiment showed a similar outcome along with the other necessary factors that were kept constant. This experiment showed the effect of light on the Growth of C. muelleri which was only one factor. There are further studies required in the similar areas. Once there is a better understanding of the culture of microalgae at a small scale, this could be used in the mass culture and used in the developing aquaculture industry in Fiji and the Pacific Islands.
This study examined the amount of light intensity required to give the best yield of C. muelleri in a laboratory culture. From the experiment carried out and the data analysis it may be concluded that the 1000 lux is the optimum lux to produce the best yield. This is supported by the literature. It was noticed that as light intensity increased the growth of C. muelleri increased significantly to a limit at 500 lux, 2000 lux and 2500 lux where the growth rate slowed. This could be due to the either the decrease in the nutrient level in the flask or due to too less or over heat of the flask.
The relationship between light intensity required and the growth rate of Chaetoceros muelleri was also studied (Figure 1). Light is required for the growth of microalgae on the other hand too much light and too little light can slow the growth rate [17,18]. Thus the experiment indicated that 1000 lux to 1500 lux is the best light intensity in order to produce maximum yield.
The authors wish to thank Mr. Jone Lima and Mr. Ravikash Prasad for their assistance in getting the pure sample of C. mulleri from Australia and assisting in Statistical Analysis using SPSS software respectively. The authors are grateful to Pacific Islands Forum Secretariat for providing financial assistance in the project and research.