Journal of Antivirals & Antiretrovirals
Open Access
ISSN: 1948-5964
ISSN: 1948-5964
Research Article - (2013) Volume 5, Issue 4
Objectives: Securigera securidaca Degen & Dörfl (Fabaceae) is an annual herb occurring wild in West Asia, Europe and Africa. The seeds of this plant are used for the treatment of disorders such as hyperlipidemia, diabetes, and epilepsy in Iranian folk medicine.
Materials: This study was carried out to check antiherpetic substances of crude methanol seed extract of Securigera securidaca and its column chromatographic fractions. The antiherpetic activities of different concentrations (20, 2, 0.2 and 0.02 μg/ml) of crude methanol extract and sub-fractions were tested by use of plaque-forming unit (PFU) assay and the real-time polymerase chain reaction (PCR) assay.
Results: The most active fractions analyzed by column chromatography, containing kaempferol and kaempferol- 7-O-glucoside. The apparent effective concentration for 50% plaque reduction (EC50) of crude methanol extract, kaempferol, kaempferol-7-O-glucoside and acyclovir (ACV) were 2, 0.2, 0.1 and 0.1 μg/mL, respectively.
Conclusion: These findings demonstrate that kaempferol and kaempferol-7-O-glucoside isolated from S. securidaca may inhibit virus attachment to the cell membrane, virus entry into the cell and viral polymerase
Keywords: HSV1; Antiviral activity; Kaempferol; Real time PCR assay
Securigera securidaca Degen & Dörfl (Fabaceae) has been applied to a variety of ailments such as epilepsy, hypertension, malaria, gastric influx, and hyperlipidemia in traditional eastern medicine. It also shows natural promise for enhancing antidiabetic, positive chronotropic, diuretic, hypokalaemic and anti-hyperglycemic effects because of its antioxidants and flavonoids [1-3]. Previously, a number of steroidal, flavonoids, alkaloids, saponins and tannins have been isolated from S. securidaca seeds [4,5]. Since these compounds may have the potential to inhibit viral infection, here the anti-HSV activity of this plant has been studied.
HSV-related opportunistic infections cause a variety of malignancies. Since resistance of virus to conventional antiviral drugs has been reported, it is necessary to find new alternative antiviral compounds [6,7]. Currently, the only aspect of the herpes simplex life cycle for which antiviral therapy has been developed is the process of DNA replication, which is targeted by a small group of nucleoside analogues including acyclovir (ACV), valaciclovir, penciclovir, and famciclovir. ACV is applied in the primary steps of infection but induces the developed drug-resistant species mostly in immunosuppressed patients. Valacyclovir and famciclovir are prescribed for recurrent infections and foscarnet has been used for ACV-resistant cases. However, despite their high efficacy, they are expensive and cause severe side effects. Resistance is also observed clinically when foscarnet is used in patients with AIDS [8]. Therefore, in vitro assays to study the anti-HSV activities of a variety of herbal medicines used by world’s folk medical systems were performed. Then it was revealed that their mechanism of function is either through inhibiting viral replication or viral genome synthesis [6,9-11]. The extract of S. securidaca has been investigated for several biological activities [1], but the antiviral properties of crude methanol extract and subtractions against HSV- 1 have not been investigated yet. Therefore, we designed a Vero cell line study for assessing the ability of viral productivity and plaque formation in the presence of crude methanol extract, sub-fractions and pure compounds obtained from this species planted in Iran agricultural condition to reveal their probable antiherpetic activity.
Plant material
The seeds of S. securidaca were collected from Agriculture and Natural Recourses Research Center of Isfahan, Iran, in December 2009. The specimen was identified by Prof. Rahimnejad and deposited at the University of Isfahan herbarium, Iran (No. 13656). Plant seeds (2 kg) were carefully dried in a well-ventilated dark room and powdered. Finally, the dried seed powder (1 kg) was obtained.
Extraction and isolation of compounds
Methanol (98%) extracts of dried and powdered S. securidaca seeds (1000 g) were prepared. The extraction was done thrice at 40°C. Then, the resulting liquid was collected, filtered and reduced through evaporation under vacuum by a rotary evaporator (Stroglass, Italy) at 45°C and dried by use of a freeze dryer (Zirbus, Germany). Silica-gel column fractionation chromatography was carried out with the dried methanol extract (4 g) of S. securidaca, eluted with n-hexane: acetone (8:2 to 2:8, v/v) and then with 100% methanol. Several fractions 1 to 9 (0.47, 0.22, 0.24, 0.32, 0.35, 0.40, 0.21, 0.2, 0.3 g) were obtained. Fraction 6 was found to have antiherpetic activity by Plaque Forming Unit (PFU) analysis and re-chromatographed on silica gel column eluted with chloroform: methanol (30:1, 20:1, 10:1, 5:1, 3:1, 2:1, 1:1, v/v) to yield fractions 6a, 6b and 6c (0.20, 0.19, 0.1 mg). Fractions 6a and 6b were detected as anti-herpetic active kaempferol and kaempferol-7-O-glucoside by NMR analysis.
HPLC analysis
HPLC screening of methanol extract, fraction 6a and fraction 6b was carried out. HPLC was performed on a HITACHI Series HPLC system equipped with L-7100 pump and an L-7100 UV-Vis detector. Peaks were separated on a C18 column using the mobile phase methanol-water (1:1, v/v). The flow rate of the mobile phase was 1.5 ml min-1. The absorption of analytes was detected at 450 nm. Samples were injected to the HPLC bed manually with injection volume as 7 μl. T2000 software was used for peak integration and calculation.
Spectroscopic analysis
High resolution nuclear magnetic resonance (1H NMR) screening was used to detect trace compounds in each fraction. 1H NMR spectra were recorded on Bruker 400 MHz spectrometer by use of CH3OD as residual solvent with chemical shifts expressed in parts per million (ppm). Fourier Transform Infrared Spectroscopy (FTIR) analysis was recorded on Jasco FT/IR- 6300 (400-4000 cm-1), Japan, (νmax, KBr).
Cells and viruses
African green monkey kidney (Vero) cells (ATCC No. CCL- 81) obtained from University of Isfahan, Iran were grown in Eagle’s minimum essential medium (MEM) supplemented with 10% (v/v) Fetal Calf Serum (FCS; Gibco-BRL, Grand Island, NY, USA), 100 U/ml penicillin (Gibco), 100 mg/ml streptomycin (Gibco), 2 mM glutamine (Gibco) and 1 mM Na-pyruvate (Gibco). A virus stock of herpes simplex virus type 1 strain KOS provided by University of Isfahan, Iran. It was propagated in Vero cells as follows: Vero cells were infected with a low multiplicity of virus and incubated for 4 days. Then, supernatant of viral stock was harvested every day over a period of 4 days postinfection. Virus titers were determined by plaque assay in Vero cells and expressed as plaque forming units per ml (PFU/ml). The viruses were stored at -70°C until use.
Cytotoxicity assay
Cellular toxicity of S. securidaca extract, kaempferol and kaempferol-7-O-glycoside was tested in vitro to obtain their noncytotoxic concentrations for the antiviral test experiments by dissolving the extracts in sterilized double distilled water and if necessary, Dimethyl Sulfoxide (DMSO) was added to the test medium at sub-toxic concentration. To avoid toxicity or interference by the solvent, the maximum concentration of DMSO in the test medium was 0.019%. To assess the proliferative effect of methanol extract and pure compounds of S. securidaca on uninfected Vero cells, extracts at concentrations of 32, 60, 120, 250, and 500 μg/ml were added to Vero cell monolayers in the maintenance medium by use of a 96-multiwell micro plate with 4.0×104 cells per well. After 72 h of incubation at 37°C, cytotoxicity was determined through a MTT proliferation assay kit (Roche, Germany) then 50% cell cytotoxic concentration (CC50) of S. securidaca was calculated according to Weislow et al. [12]. All assays were carried out in triplicate (Figure 1).
Antiviral activity
Anti-HSV activities of methanol extract, kaempferol and kaempferol-7-O-glycoside were investigated via plaque reducing assay with confluent 24 h old monolayer of Vero cells grown in microtitre tissue culture plates. The cell monolayers were infected with 25 PFU of HSV-1 and incubated at 37°C for 2 h, and then infected cells were washed and overlaid with medium supplemented with 2.5% methylcellulose and different concentrations of extract (20, 2, 0.2, and 0.02 μg/ml). 0.1% DMSO and different concentrations of Acyclovir (20, 2, 0.2, 0.02 μg/ml) were used as negative and positive controls. After 3-4 days of incubation, the overlay medium was removed, the cell monolayer was fixed with 3.7% formalin for 5 min, and visible plaques were counted after staining with 1% crystal violet. The antiviral activity was determined by the following formula:
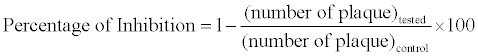
The minimal concentrations of extracts required to repress the development of virus plaque number by 50% (EC50) were calculated by regression analysis of the dose-response curves generated from data according to Cheng et al. [13] (Figure 2).
Figure 2: Effect of Securigera securidaca methanol extract, Kaempferol, kaempferol 7-O-glycoside , and ACV on HSV-1 replication in Vero cells. The 50% inhibitory concentration (EC50) of each extract was calculated using regression line. Each bar represents the mean ± SD of three independent experiments. (*) There is a significant difference between test extract and solvent control (p < 0.05). EC50 values of crude methanol extract, kaempferol, kaempferol-7-Oglucoside and acyclovir (ACV) were 2 ± 0.12, 0.2 ± 0.01, 0.1 ± 0.01 and 0.1 ± 0.01 μg/mL, respectively. The calculated selective index (SI) for methanol extract and kaempferol and kaempferol-7-O-glycoside was 125, 300 and 5000 respectively.
Time of addition study
The time-of-addition effect of kaempferol and kaempferol-7-O-glycoside were examined as described by Behbahani et al. [14] Vero cells, 2×105 per well, were seeded into 24-well culture plates (Nunc; Nalge Nunc International, Rochester, NY, USA) and incubated for 24 h. Then, confluent monolayer was treated by 5 μg/ml of kaempferol and kaempferol-7-O-glycoside added into the wells either concurrent with HSV-1 (0 h) or at intervals of 2, 4, and 6 h pre-infection and also postinfection. The remained procedures were similar to “plaque forming assay” section, except that cells were washed thrice by PBS to eliminate extract prior to the inoculation of virus for “pre-infection” (-6, -4 and -2 h) group, and the extracts were added at different times for both “during HSV” (0 h) group and “post-infection” (2, 4 and 6h) group. After 2 h of infection, the infected cells were washed and overlaid with medium supplemented with 5% FBS. After incubation at 37°C for 72 h, the percentage inhibition was calculated as the reduction in the virus titer obtained by real-time polymerase chain reaction (PCR) assay of infection cultures containing the extracts was compared with that of the control cultures for each treatment (Figure 3).
Figure 3: Time-of-addition effect of crude kaempferol (A) and kaempferol-7-OGlycoside (B) obtained from Securigera securidaca on HSV-1 replication in Vero cells. 5 μg/ml of each extract was added either before (−2 ,-4 and −6 h), during (0) and after (2, 4 and 6 h) virus infection. The extracts added before virus infection was rinsed off prior to the virus exposure. Each value is the result of mean ± SD of three independent experiments.
Quantitative real-time polymerase chain reaction assay for HSV-1
For real-time PCR, 200 ml of supernatant of each treated-infected, or untreated-infected (virus control) wells were collected. DNA was purified from 200 μl of each specimen by QIAamp DNA Mini Kit (Qiagen) according to the standard protocol. Purified DNA was resuspended in 200 μl of elution buffer, and 5 μl aliquots were used as PCR templates. As previously described forward primer, HSV pol F (5′ GCTCGAGTGCGAAAAAACGTTC 3′) and the reverse primer, HSV pol A (5′ GCGGTTGATAAACGCGCAGT 3′) [15] were used to amplify a 140 bp product. A pair of fluorescence labeled probes; HSV- 1 FLU (5′ GCGCACCAGATCCACGCCCTTGATGAGC-FLUOR) and HSV-1 LCR (5′ LC-Red 640-CTTGCCCCCGCAGATGACGCCphos) were used for real-time detection. The real time PCR master mix contained the following: Taq DNA polymerase (Molecular Diagnostics), a dNTP mix (containing dUTP instead of dTTP), 3 mM MgCl2, 0.5 μM of each primer, 0.2 μM HSV-1 FLU and 0.4 μM HSV-1 LCR. Cycling conditions were as follows: initial denaturation/ FastStart Taq DNA polymerase activation at 95°C/10 min, 45 cycles of denaturation at 95°C/5 sec, annealing at 58°C/10 sec (with fluorescence acquisition at the end of each annealing stage) and extension at 72°C/12 sec [16].
Statistical analysis
Data from three independent experiments were presented as mean ± SD. The EC50 and CC50 values were calculated by Microsoft Excel 2000. A selectivity index (SI) was calculated for each viral strain by the ratio of CC50 to EC50 value. The Student’s unpaired t-test was used to assess significance between the test sample and the solvent control. P-value<0.05 was considered to be statistically significant.
HPLC analysis
Figures 4A and 4B show the HPLC chromatograms of fractions 6a and fraction 6b respectively. After purification only one peak were obtained separately for both fractions 6a and 6b. Fraction 6a and 6b peaks were also observed at retention times 18.5 and 16.5 min. These peaks were observed with high intensity and were later identified by NMR analysis.
Spectroscopic analysis
The most active fraction obtained from the first column was fraction 6, which were further analyzed by NMR experiment. Then, it was fractionated by use of normal phase chromatography to get two active compounds 6a and 6b and analyzed using 1H NMR and FT-IR.
Compound 6a: 1H NMR (DMSO, 400 MHz): δ 11.89 (s, 1H, 5-OH), 9.82 (s, 1H, 3-OH), 7.85 (d, J=8.8 Hz, 2H), 7.20 (d, J=8.8 Hz, 2H), 6.80 (d, J=1.8 Hz, 1H), 6.72 (d, J=1.8 Hz, 1H), 5.92 (s, 2H, 7, 4’-OH).
FT-IR (νmax, KBr): 3335, 2928, 1668, 1584, 1495, 1392, 1075.
Compound 6b: 1H NMR (DMSO, 400 MHz): δ 11.78 (s, 1H, 5-OH), 9.80 (s, 1H, 3-OH), 7.80 (d, J=8.8 Hz, 2H), 7.20 (d, J=8.8 Hz, 2H), 6.78 (d, J=1.8 Hz, 1H), 6.70 (d, J=1.8 Hz, 1H), 5.88 (s, 1H, 4’-OH); Glucose moiety: 4.87 (d, J=7.3 Hz, 1H), 4.79 (dd, J1=9.2, J2=8.3 Hz, 1H), 4.38 (t, J=7.5 Hz, 1H), 3.90 (m, 1H), 3.76 (dd J1=9.7 Hz, J2=2.1 Hz, 1H), 3.72 (d, J=11.8 Hz, 2H), 3.60 (s, 4H).
FT-IR (νmax, KBr): 3393, 3298, 2926, 2895, 1704, 1660, 1605, 1513, 1460, 1420, 1245, 1139, 1072, 998.
Spectroscopic analyses suggested that compounds 6a and 6b which showed anti-herpetic activity were identified as kaempferol and kaempferol-7-O-glucoside respectively.
Anti-HSV-1 activity
Antiviral activity of 20 μg/ml of methanol extract and twelve fractions of S. securidaca were evaluated by plaque reduction assay. Results showed that 20 μg/ml of methanol extract, fraction 6a (kaempferol) and fraction 6b (kaempferol-7-O-glucoside) inhibited HSV-1 multiplication with an inhibition rate of 98 ± 5.2%, 100 ± 2.9% and 100 ± 2.9% respectively. Other fractions showed inhibition rate lower than 10% (data not shown). The antiviral activity of these compounds was further examined by different concentrations (Figure 2). The concentrations of 2, 0.2, and 0.02 μg/ml yielded percentage of inhibition as 70, 50 and 31 in sub-fraction 6a and 78, 60 and 40 in subfraction 6b, respectively. EC50 values of methanol extract, kaempferol, kaempferol-7-O glucoside and ACV were 2 ± 0.12, 0.2 ± 0.01, 0.1 ± 0.01 and 0.1 ± 0.01 μg/mL, respectively. Also, the calculated selective index (SI) for methanol extract, kaempferol and kaempferol-7-O-glycoside were 250, 300 and 2500 respectively.
Cytotoxic effects on viability of Vero cells
The concentrations of methanol extract from S. securidaca not affecting the viability of Vero cells were investigated by MTT assay. Results showed that methanol extract, kaempferol-7-O-glycoside and kaempferol had no cytotoxic effect up to the concentrations of 15, 15 and 5 μg/ml respectively. Therefore, the mechanism of anti-HSV activity of these compounds was assayed at low concentration (5 μg/ml). The CC50 values of methanol extract, kaempferol and kaempferol-7-O-glycoside were 500 ± 6.2, 60.0 ± 5.0 and 250 ± 1.7 μg/ml respectively.
Time of addition study
A time-of-addition experiment was performed by measuring the viral DNA yields in infected culture supernatants, by means of a real-time PCR assay. The plant extract, kaempferol and kaempferol- 7-O-glycoside were added before, during and after virus absorption presented in the culture medium until the end of the experiment. The curves generated during PCR reactions showed a prominent drop in HSV-1 DNA amounts in treated cultures compared to those of virus controls (Figure 3). The strongest HSV proliferation inhibition was achieved when the extract was added during the initial stages of infection and 2 h after infection. The result showed that viral DNA during and 2 h after infection was 103-folds lower than the viral DNA present in the untreated virus control. No viral DNA was detected in non-infected cells. kaempferol-7-O-glycoside produced a pattern similar to that observed in cultures treated with Kaempferol. The strongest HSV proliferation inhibition was also achieved during the initial stages of infection and 2 h after infection. The result showed that antiviral activity of kaempferol-7-O-glycoside was significantly better than kaempferol.
Several herbal remedies and natural products have been shown to exert antiherpetic effects. Some of them appear to pose the broadspectrum antiviral activity without extensive toxic effects as ideal candidates in antiviral therapy [17]. Two extracted preparations from the medicinal plant S. securidaca were investigated for their herpes virus inhibitory effect by two separate assay methods. The tested crude methanol extract and sub-fractions 6a and 6b showed moderate and excellent antiherpetic activity, respectively. Sub-fractions 6a and 6b were characterized as kaempferol and kaempferol-7-O-glucoside. According to literature kaempferol and its 3-O-glycoside derivatives are natural flavonols that have been identified in various plants, vegetables, fruits, and beverages [18,19]. These flavonols have been reported as antitumor, anti-oxidative, anti-allergic and antivirus activities [20,21]. Yarmoinsky [21] reported that kaempferol 3-O-rutinoside and kaempferol 3-O-robinobioside possess strong anti-HSV1 activity. Anti-viral activity of kaempferol against several virus strains such as HIV has been reported before [22-24]. Many in vitro and in vivo studies have been published about anti-herpetic properties of other flavonoids [25].
The naturally occurring flavonoids are believed to possess the ideal chemical structure for scavenging free radicals [26]. Flavonols appeared to be more active than flavones in anti-herpetic effects and the direct inactivation of HSV by some flavonoids such as quercetin, procyanidin, pelargonidin, catechin and hesperitin has been verified [27]. Debiaggi et al. [28] investigated propolis-derived flavonoids and reported a concentration-dependent reduction of HSV replication for quercetin, when high drug concentrations were used.
The calculated selective index (SI) for methanol extract, kaempferol and kaempferol-7-O-glycoside were 250, 300 and 2500 respectively. This important criterion determines that the SI ≥ 4 should be considered suitable for a very potent and very safe antiviral agent [28] suggesting that the remarkable anti-HSV-1 activity could be attributed to methanol extract and kaempferol and kaempferol-7-O-glucoside.
The antiviral activities in kaempferol and kaempferol-7-O-glucoside-treated Vero cells were similar during and 2 h after challenge by HSV-1. Pre-incubation of the Vero cells with extracts of S. securidaca and then washing the extracts out did not protect the cells from HSV- 1 infection (Figure 3). These observations indicate that kaempferol and kaempferol-7-O-glucoside isolated from S. securidaca may inhibit virus attachment to the cell membrane, virus entry into the cell and viral polymerase. Nevertheless, further studies are needed to verify the underlying mechanism of active compound from S. securidaca extracts in inhibiting HSV-1 infection. The results of identification on the bioactive compounds indicated that kaempferol and kaempferol- 7-O-glucoside compounds constituted the most active compounds in the crude methanolic extract from S. securidaca exhibited an effective inhibitory mechanism on the early stage of HSV-1 infection.
The authors have no competing interests in connection with this manuscript.
This work was supported by the grant from Faculty of Advanced Sciences and Technologies, University of Isfahan, Iran. We thank Faculty staff for their research efforts and comments on the study design