Journal of Sleep Disorders & Therapy
Open Access
ISSN: 2167-0277
ISSN: 2167-0277
Review Article - (2019)Volume 8, Issue 2
Erectile dysfunction (ED) and obstructive sleep apnoea (OSA) are increasingly common conditions seen in primary and secondary care. With shared risk factors and associated comorbidities men often suffer with both. The aim of this study is to review the literature to consider why there is a correlation, and clinically how assessment and management of such patients could be adjusted. A structured review of current literature was performed using search terms ‘erectile dysfunction' or 'erectile function' or 'Erection' or 'Sexual dysfunction' AND 'Sleep apnoea/apnea' or 'sleep disorder'. Descriptive analysis categorized results into: The association between OSA and ED; the pathophysiological hypotheses; the role of OSA treatment; and the role of ED treatment. Higher prevalence of ED in patients with OSA is demonstrated, particularly in men with more severe OSA disease, reaching 80% in some studies. However, the pathophysiological explanation remains unclear with a number of hypotheses researched. CPAP is the mainstay of treatment for OSA, and short and long-term benefits to sexual function are demonstrated in men with severe OSA from it’s use. Further improvement in ED and quality of life can be achieved by additional PDE5 inhibitor therapy. With the increasing evidence base showing higher rates of ED in OSA patients there is certainly a role for clinicians in both OSA and ED clinics to enquire about each condition. A referral to their counterpart for consideration of combination treatment with CPAP and a PDE5 inhibitor for both of these problematic, life changing diseases is recommended-particularly in those with severe disease. Further investigation is needed to try and establish any pathophysiological link, and the impact of treatment in less severe cases.
Erectile dysfunction; Sexual function; Obstructive sleep apnoea; Therapy
Obstructive sleep apnoea (OSA) is chronic disorder characterized by snoring, episodes of hypoxia, fragmented sleep and daytime somnolence due to upper airway obstruction [1]. It is estimated that the worldwide prevalence is between 9 and 38% [2], however the prevalence in the UK is thought to be just 4% [3]. Patients with OSA have reported increased fatigue, poor quality of life, deterioration of personal relationships, and reduced libido [4-6].
Risk factors for developing OSA include increasing age, male sex and obesity. This often co-exists with hypertension and cardiovascular disease. In fact, OSA has been suggested as a factor in development and accelerating progression of cardiovascular disease. Increased rates of systemic and pulmonary hypertension, atrial fibrillation and heart failure are thought to be due to nocturnal surges of sympathetic tone caused by episodes of OSA [7].
Erectile dysfunction (ED) is a common condition defined as the “persistent inability to attain and/or maintain a penile erection sufficient for sexual performance”. The British Society of Sexual Medicine Guidelines [8] reports a prevalence ranging from 10 to 52% from several large studies, with a steep age-related increase. ED shares many associated physical conditions with OSA and both have also been demonstrated to affect psychosocial health [4-6,8-11].
The associations between ED and OSA have been suggested and investigated in multiple clinical and epidemiological studies. Guilleminault et al. were the first to report an increased prevalence of ED within a series of 25 patients diagnosed with OSA [12]. Subsequent studies have reported similar findings with prevalences ranging between 41 and 80% [13-15]. Chen et al. reported the incidence of ED to be 9 times higher in patients with OSA [16]. Further studies have demonstrated an improvement in both ED and OSA with continuous positive airway pressure (CPAP) devices [17]. However, Schiavi et al. and Bozorgmehri et al. report conflicting results finding no association between the two conditions in their groups of systemically healthy men [18,19].
The aim of this descriptive review is to further assess the association that may link OSA and ED and give recommendations for the practitioner who is making a diagnosis of either condition.
A structured literature search was conducted independently by the lead author and then by a member of the University Hospitals Birmingham (UHB) Library and Knowledge Service. Within CINAHL, EMBASE and MEDLINE, the following search terms were used 'erectile dysfunction' or 'erectile function' or 'Erection' or 'Sexual dysfunction' AND 'Sleep apnoea/apnea' or 'sleep disorder' or OSA (obstructive sleep apnea). Databases were searched from January 2009 to January 2019.
Inclusion criteria from original searches were: Published January 2009–January 2019; English language; validated measures of erectile function and OSA; and clear relevance to the topic. Exclusion criteria were: Female patients only; other language than English; Commentaries, letters to the editor, interviews and updates.
Titles and abstracts were reviewed for relevancy and to confirm suitability for inclusion. Full texts were then studied to confirm meeting eligibility criteria. Citations of identified articles were then hand-searched to identify potentially relevant studies including those written prior to the search limits with no restriction on date published. Papers relating to the physiology of OSA and ED, their treatments and associations and outcomes were included. Robust critical appraisal of each article was performed to assess relevance and quality. Descriptive analysis allowed categorization into areas of interest.
95 articles were identified. Following review of the abstracts 34 were excluded for a variety of reasons including female study population, not clearly relevant to topic, or written in other language than English. On reviewing these articles a further 12 studies predating 2009 were selected from citation searches. 73 articles were selected for further analysis (Figure 1).
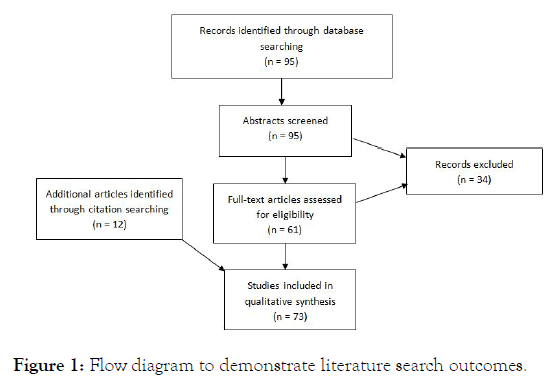
Figure 1: Flow diagram to demonstrate literature search outcomes.
Descriptive analysis was performed with categorizations into: The association between OSA and ED; The pathophysiological hypotheses; the role of OSA treatment; and the role of ED treatment.
Since Guilleminault et al. [12] suggested a relationship between sleep disturbances and ED, numerous international studies have published higher prevalences of ED in men with OSA ranging from 44-80% [13,14,20-24]. A recent meta-analysis of 14 studies by Kellesarian et al. also reported prevalence up to 80% and a pooled odds ratio of 0.45 with a 95% confidence interval of 0.18-0.71 [15]. In studies comparing patients matched for comorbidities and age, men with OSA had poorer sexual function when compared with those without OSA [22,24]. Given the variety of methods used to diagnose erectile dysfunction including nocturnal penile tumescence (NPT) studies, self-reporting or validated questionnaires interpreting a pooled prevalence may be misleading; however a clear increased prevalence of ED is noted in patients with OSA.
Fewer recent studies report prevalence of OSA in men presenting with ED. Seftel et al. report just 28% prevalence based on depression inventories and sleep habit questionnaires [25]. However, meta-analysis of 4 additional historic papers gave a range of 28 to 79% [15].
Growing evidence of positive correlations between the two conditions suggests OSA as an independent risk factor for ED [26,27]. It remains unclear if the severity of OSA has an impact on the erectile function. There are suggestions that only moderate or severe OSA is associated with increased ED. However, simple snoring, without clarification on confirmed diagnosis of OSA, was also associated with ED [25]. More recent studies have recommended direct questioning of patients presenting with ED about sleep disturbances and snoring or vice versa [20,28].
Pathophysiological relationship
The pathophysiological association between ED and OSA remains controversial. Early conclusions that erectile and sexual function was impaired due to associated depression and daytime somnolence from sleepiness have been dismissed and a more complex relationship is thought to be present [11]. Treatment of such patients with antidepressant therapy alone showed no improvement in erectile function despite depression score improvements [26].
Alternative theories range from tissue oxygenation, vascular endothelial or hormonal changes as effects caused by an underlying disturbance of sleep.
Luboshitsky et al. described a decrease in pituitary-gonadol secretion in men with altered sleeping patterns secondary to OSA [29]. These changes may lead to decreased testosterone release with an associated reduction in libido [30]. Higher degrees of hypoxaemia during sleep are related to lower testosterone levels. This effect in OSA patients is independent of age and obesity [31].
A further theory focuses on the role of physiological erections during Rapid Eye Movement (REM) sleep. These erections may encourage cavernosal oxygenation to maintain erectile function. The fragmentation of sleep associated with OSA reduces these physiological erections and subsequently has an adverse effect on erectile function when awake [23,24].
Similarly transient hypoxia, present in OSA, has been demonstrated to affect erectile function even in healthy individuals based on nocturnal tumescence studies. Alternative studies demonstrated significant impairment of pudendal nerve function and bulbocavernosus reflex secondary to hypoxia [32,33], suggesting a possible causation for ED in patients with OSA.
Both biochemical animal studies and pathophysiological patient studies have hypothesised that the link between ED and OSA is due to vascular aetiology. Intermittent hypoxia in OSA causes an inflammatory state [34] with subsequent impaired endothelial related vasodilatation mechanisms [35,36]. The rise in proinflammatory markers has been found to reduce nitric oxide, with levels of nitric oxide synthase lower in hypoxic environments [37]. In support of this, CPAP therapy increases nitric oxide levels in these patients [38].
In addition, Wilson’s animal study recently concluded that oxidative stress from chronic intermittent hypoxia mimics the effects of ageing on testosterone and subsequently male sexual behavior [39].
The mainstay of treatment for all degrees of OSA is through continuous positive airway pressure (CPAP) which splints the upper airway
The impact of CPAP treatment for OSA on erectile dysfunction is difficult to quantify because studies have used variable outcome measures. However, overall quality of life, sexual quality of life and erectile dysfunction have all been show to improve with CPAP.
Improvement of erectile function was first reported with nocturnal penile tumescence (NPT) studies in OSA patient pre and post CPAP treatment. Karacan and Karatas showed CPAP improved ED parameters in a third of patients [40]. The improvement in penile rigidity was greater in those with severe OSA and younger patients. This cohort study demonstrated an initial association but without clinical assessment or subjective improvement results must be approached with caution.
Melehan’s recent randomised trial of 61 men showed CPAP alone increased sleep-related erections and sexual satisfaction, but not erectile function on the International Index of Erectile Function (IIEF) score. An improved IIEF score was only seen in the group treated with CPAP and vardenafil [41]. However, Schulz reported significant improvement in erectile function with CPAP treatment in men with moderate or severe ED based on IIEF. This evaluation of 94 patients with OSA prospectively used IIEF before and after 6-12 months of CPAP treatment [42]. Several other studies have come to similar conclusions.
In a randomised study, Taskin et al. demonstrated an increase of mean IIEF-5 score from 15.71 to 19.06 after at least one month of CPAP [26]. This was statistically significant, and would move a man from mild-moderate ED to the mild ED category. Zhang also reported a significant mean improvement in IIEF-5 score following 3 months CPAP in 108 men with severe OSA [43]. However, numerically this improvement was from 18.21 to 19.21. Meaning patients remain in the mild ED category. It seems unlikely such a change would translate to significant clinical improvement. A small study of 21 men used the full IIEF questionnaire and showed a significant improvement in scores was demonstrated with 12 weeks of nasal CPAP [44].
Alternative assessments of erectile dysfunction have concluded similar positive results. In interviews, 75% of OSA patients found one month of CPAP therapy improved their sexual function, and in those with improved erectile function quality of life also improved [45]. Cruz et al. utilised responses to the Sleep Disorders Questionnaire in a cohort of patients with moderate and severe OSA treated with nasal CPAP for a minimum 6 months. The prevalence of ED in this group dropped from 25% to 18% following treatment but was not found to be significant [46]. The initial prevalence of 25% is lower than that found in most other studies, which may suggest that not using a validated tool such as IIEF to assess ED may cause underestimations of prevalence.
Two systematic reviews concluded that CPAP for OSA patients with ED achieved significant improvement in sexual parameters [47], specifically erectile function and orgasmic function [6]. However, since these publications, Jara’s randomised control trial found that the treatment association of CPAP and improvement of sexual quality of life was only found in women, not in the cohort of 115 men with severe OSA [48].
Two studies have demonstrated that long-term CPAP therapy maintains an improvement in sexual function. Margel et al. found 20% patients showed an improvement in IIEF results with 12 months CPAP therapy. Perhaps more importantly, the 18% with worsening of IIEF results were less compliant and less satisfied with CPAP therapy overall [49]. With over 3 years follow up Budweiser et al.’s observational study concluded that long-term CPAP can improve or preserve sexual function in men with OSA and moderate to severe ED. This was based on returned IIEF-15 scores from 21 CPAP users and 18 non-CPAP users with ED, and a demonstrably significant decline in erectile function of the group of 91 responders overall. This was a postal questionnaire with a response rate of 23% so utility of such data must be questioned [50].
It has also been demonstrated that OSA treatment with surgery has benefits to erectile function. Historical papers with small patient numbers report surgery to create unobstructed inspiration caused almost complete resolution of erection and ejaculation problems [51]. In addition there was normalization of previously suppressed testosterone, sexual function and libido [52]. Although no validated assessment tools were used, interviews were conducted with patient and their spouse. In support of these findings in 2012, Khafagy and Khafagy showed that men with poor CPAP compliance who underwent surgical procedures for OSA had statistically significant improvement in both respiratory and erectile function tests (p<0.05). This paper lacks a comparison between outcomes from surgical correction and CPAP so again conclusions of relative value are difficult to make [53]. Shin et al. compared CPAP with surgical management [52]. They found erectile function was only significantly improved in the surgical group (p=0.039). This paper had small numbers in each group, in which patients were not randomised [52] (Table 1).
| Author (year) (n=male patients) | OSA treatment | ED treatment | ED measures | Length of OSA treatment | Outcomes (Potential weakness of study) |
|---|---|---|---|---|---|
| Karacan and Karatas (1995) [n=15] | CPAP | - | NPT | 1 night | 33% patients improved ED–higher in severe OSA (1 night study, NPT may not correlate with patient perception of EF) |
| Melehan (2018) [n=61] | CPAP | 10 mg vardenafil | NPT, IIEF, Sexual function questionnaire | 3 months | Improved IIEF, sexual function, nocturnal erections with CPAP Significant IIEF improvement with combined therapy (Improvement in IIEF may not mean fully treated ED) |
| Schulz (2018) [n=94] | CPAP | - | IIEF-5 | 6-12 months | Significant improvement in ED for men with moderate or severe ED. (Improvement in IIEF may not mean fully treated ED) |
| Taskin (2010) (n=40) | CPAP vs antidepressant | - | IIEF-5 | 1 month | Significant improvement in mean IIEF-5 compared to non-CPAP group. (15.71 to 19.06 points–still diagnostic of ED; only severe OSA patients). |
| Zhang (2016) (n=108) | CPAP | - | IIEF-5 | 3 months | Significant improvement in mean IIEF-5. (18.21 to 19.21 – still diagnostic of ED) |
| Acar (2016) (n=21) | Nasal CPAP | - | IIEF | 3 months | Significant improvement in mean IIEF (50.28 – 65.42) |
| Goncalves (2005) (n=17) | CPAP | - | Sexologist interview, QOL SF-36 | 1 month | 76.5% improved from ED to non-ED group. (Interview bias, small numbers, short term study, no tool used specifically for ED) |
| Cruz (2012) (n=98) | Nasal CPAP | - | Sleep disorders questionnaire (SDQ) | 6 months | Non-significant fall in ED prevalence (25% to 18%) (No tool used specifically for ED) |
| Jara (2018) (n=115) | CPAP | - | SNORE-25 (Sexual domains) | 12 months | No improvement in sexual QOL in men. (No tool used specifically for ED) |
| Margel (2005) (n=60) | CPAP | - | IIEF-5 | 12 months | 20% men significantly improved. 18% who significantly worse had poor compliance and satisfaction with CPAP. (58% response rate to postal questionnaires) |
| Budweiser (2013) (n=91) | CPAP | - | IIEF-15 | 3 years | Overall worsening of ED in all men. Preserved function in CPAP group with moderate or severe OSA. (23% follow up response rate, no control for comorbidities) |
| Shin (2013) (n=56) | CPAP | - | IIEF-5 | 4-15 months | Significant improvement in IIEF scores in UPPP group, not in others. (Not randomized, small samples, no adjustment for OSA severity or age) |
| MAD | |||||
| UPPP | |||||
| Khafagy and Khafagy (2012) (n=80) | CPAP | - | IIEF-5, NPT | 3 months | Significantly improved EF in men who had surgical procedures due to poor CPAP compliance. (No comparison of CPAP vs surgery; no adjustment for OSA severity). |
| UPPP |
Note: OSA: Obstructive Sleep Apnoea; ED: Erectile Dysfunction; CPAP: Continuous Positive Airway Pressure; NPT: Nocturnal Penile Tumescence; IIEF: International Index of Erectile Function; MAD: Mandibular Advancement Devices; UPPP: Uvulopalatopharyngoplasty.
Table 1: The effect of CPAP and surgical treatment for OSA on erectile function.
The role of phosphodiesterase type 5 inhibitors (PDE5i) in addition to CPAP therapy has also been analysed.
Perimenis et al. compared IIEF results in men with mild to moderate OSA treated with CPAP and on demand sildenafil 100 mg against CPAP alone. On analysis of patient selfresponses they found significantly more (61.1 vs 24.7%) successful intercourse episodes reported in the combined therapy arm [54].
In an attempt to clarify the role of each treatment the same group conducted randomised trials comparing the benefits of each in OSA cohorts. After 3 months of either treatment an improvement from baseline was found. However, the men treated with sildenafil self-reported significantly higher numbers of sexual intercourse attempts, successful intercourse and IIEF scores [55], therefore more likely to be effective. A similar RCT of 82 men with severe OSA supported these findings [56].
Interestingly, both studies demonstrate overall satisfaction with either treatment is suboptimal. However, satisfaction is considerably higher in PDE5I groups (53-68% vs 20-29%), as confirmed by the systematic review by Campos-Juanatey et al. [47]. As commented in this review, these trials do not document respiratory outcome, and the concern would be those that are untreated for OSA may experience further decline in health, erectile function and quality of life.
A meta-analysis by Li et al. concluded significantly higher percentages of successful intercourse attempts and erectile function scores in those treated with PDE5i (p=0.02). These men also reported higher, but not-significant, satisfaction in regard to treatment for ED [57].
Minimal research has been conducted into the effect of PDE5 inhibitors on respiratory function of OSA patients. Two studies including a total of 13 patients reported worsening in oxygen saturations and cardiac effects when comparing Sildenafil 50 mg with placebo [58,59]. The authors acknowledged that validation of their conclusions was lacking due to the limited numbers in their study. Further randomized controlled trials should help enable conclusions to be drawn on whether PDE5 inhibitors have an impact on pulmonary function in all severities of OSA.
This search also demonstrated a paucity of high quality research into other treatments for ED including intracavernosal or intraurethral alprostadil, vacuum devices and penile prosthesis insertion (Table 2).
| Author (year) (n= male patients) | OSA treatment | ED treatment | ED measures | Length of OSA treatment | Outcomes (Potential weakness of study) |
|---|---|---|---|---|---|
| Perimenis (2004) (n=30) |
CPAP | Sildenafil 100 mg | Patient reported successful intercourse attempts, IIEF, Overall satisfaction | 3 months | Significant increase in successful intercourse attempts. IIEF improves in both, significantly higher in PDE5i group. (No treatment for OSA in Sildenafil arm; Patient self-reporting “successful” intercourse) |
| Perimenis (2007) (n=40) |
CPAP | Sildenafil 100 mg | Patient reported successful intercourse attempts, IIEF, Overall satisfaction | 3 months | 70% overall satisfaction, higher in PDE5i group. IIEF significantly higher in PDE5i group vs CPAP. (No placebo group; mean OSA severity=mild to moderate; Patient self-reporting “successful” intercourse) |
| Pastore (2014) (n=82) |
CPAP | Sildenafil 100 mg | Patient reported successful intercourse attempts, IIEF | 3 months | Significant increase in successful intercourse attempts (30.4% to 58.2%). Significantly more satisfied and higher IIEF scores in PDE5i group. (No treatment for OSA in Sildenafil arm; Patient self-reporting “successful” intercourse) |
| Li (2010) (n=70 from 2 RCTs) |
CPAP | Sildenafil 100 mg | Patient reported successful intercourse attempts, IIEF, Satisfaction |
3 months | PDE5i group significant advantage in successful intercourse attempts, IIEF scores and satisfaction of the patients and their partners. (Patient self-reporting “successful” intercourse) |
Note: OSA: Obstructive Sleep Apnoea; ED: Erectile Dysfunction; CPAP: Continuous Positive Airway pressure; PDE5i: Phosphodiesterase-5 inhibitor; IIEF: International Index of Erectile Function.
Table 2: The effect of PDE5 inhibitors on erectile function in patients with OSA.
With a wide crossover of comorbidities associated with both ED and OSA it is little surprise that associations between the two have been demonstrated widely throughout the literature. The range in prevalence of ED in the OSA population is understandably wide when considering the various tools used to measure symptoms, severity of disease, and potential confounding factors. Studies that matched for such confounders demonstrated higher prevalence rates. Similarly, many studies reported higher rates of ED in more severe forms of OSA only, leading to controversy regarding the impact or otherwise of a less severe OSA diagnosis. The current evidence demonstrates that OSA is certainly associated with higher risk of ED. However, there is a need for further trials and prospective studies to assess the impact of less severe disease so that a more tailored approach to management can be made.
Given the association between these two common diseases, it is disappointing that the physiological link remains unclear. A number of hypotheses have been explored and evidence supports erectile function being impaired due to repeated episodes of hypoxia in OSA. Theories of hormonal irregularities, vascular endothelial inflammatory responses, and interrupted REM sleep have being explored. Although uncertainty remains, these studies have somewhat overtaken the original thought that ED was solely related to the mood and sleepiness of a man with OSA. Research may demonstrate that these alternatives theories and the subsequent impact on erectile function should not be dismissed.
CPAP is the mainstay of treatment for OSA. Improvements in sexual function through CPAP therapy are demonstrated in the short and long-term. However, improvements ranged considerably; outcome measures were inconsistent, and seemingly the best outcomes were for those with severe ED or OSA. Of note, many papers reported improvements in ED based on minor changes in IIEF score, but in reality this is unlikely to equate to a noticeable symptomatic improvement. Clearly CPAP has an important role in the management of OSA, and it seems a byproduct of persistent uses is that some men have improved sexual function-particularly those men with severe OSA.
The improvement in ED and subsequent quality of life can be increased further by the addition of a PDE5 inhibitor. This is the agreed conclusion from a meta-analysis, systematic review, and small randomized trials. Higher satisfaction levels reaching 68% were demonstrated in the group receiving on demand Sildenafil 100 mg with CPAP therapy, compared to men receiving each individual therapy. The role of PDE5 inhibitors in conjunction with CPAP has been further verified by Melehan’s randomised trial demonstrating low dose daily vardenafil improves quality of life and ED in these men.
Erectile function outcomes from surgical management of OSA seem promising with better efficacy than CPAP. However, these studies have small numbers of patients, lack widespread utility, and the outcome measures used are questionable. Before wider recommendation of such surgical techniques can be considered, substantial additional evidence is required.
The recent clinical recommendations of enquiring about OSA or utilization of an OSA screening tool in men with erectile dysfunction should be encouraged. It has been demonstrated as feasible, and the evidence of treatment benefits for these men is widely presented [20,28]. However, this is likely to lead to detection of men with lower severity OSA that may not find the same improvements in ED with treatment. Perhaps the more important role is in the clinicians in sleep or respiratory clinics enquiring about erectile dysfunction. This would allow early referral to a specialist; or even prompt commencement of a PDE5 inhibitor with the intention of improving quality of life and overall satisfaction whether CPAP is required or not. Clearly this will require some additional time in busy clinics. Another concern would be a lack of patient education regarding ED and the association with OSA may affect outcomes. For those attending a respiratory clinic may find it unusual or distressing to be asked sexual function questions as a matter of routine, and then offered a tablet for ED at the end of the consultation.
This study is an update descriptive review of the literature regarding OSA and ED. Limitations include the search strategy with one clinician and one library employee was also limited by date. Search terms could have been more specific for each topic within the generic review title. Study quality of reviewed articles was not formally evaluated using available tools however each paper underwent robust critical appraisal. The papers were reviewed by one author and analyzed descriptively. Statistical analysis was not performed. The heterogeneity of the approaches used in the articles reviewed would make any such analysis challenging. The citation reviews were a rich source of additional studies in this area published prior to our date limit of 2009. This method of searching has high risk of missing important research, although utilizing citation lists and study quality assessment tables from similar earlier reviews will have reduced this.
With the increasing evidence base showing higher rates of ED in OSA patients there is certainly a role for clinicians in both OSA and ED clinics to enquire about each condition. A referral to their counterpart for consideration of combination treatment with CPAP and a PDE5 inhibitor for both of these problematic, life changing diseases is recommended-particular in those with severe disease. Further investigation is needed to try and establish any pathophysiological link, and the impact of treatment in less severe cases.
Citation: Polson PJ, Ralph DJ, Kalejaiye O (2019) Erectile Dysfunction and Obstructive Sleep Apnoea-What is the link and what can we do about it? J Sleep Disord Ther 8:304.
Received: 30-Jul-2019 Accepted: 23-Sep-2019 Published: 30-Sep-2019 , DOI: 10.35248/2167-0277.19.8.304
Copyright: 2019 Polson PJ, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.