Endocrinology & Metabolic Syndrome
Open Access
ISSN: 2161-1017
ISSN: 2161-1017
Research Article - (2019)Volume 8, Issue 2
Alafia barteri, Apocynaceae has been valued for its efficiency in traditional medicine system in Nigeria and other African countries, as an anti-inflammatory and fever remedy. This study was carried out to know the effect of Alafia barteri leaf extract on the histo-architecture of the pancreas and stomach in Alloxan induced diabetic wistar rats.
A total of Twenty (20) Wistar rats were used for this study, animals were randomly selected into Four (4) groups of Five (5) animals and were acclimatized for period of Two (2) weeks Group 1 the Control Group (CG) received 2mls of distilled water, Group 2 Normoglycemic Treated (NT) with 400 mg/kg of Alafia barteri aqueous leaf extract, Group 3 Induced Hyperglycaemic Untreated (IU), and Group 4 Induced Hyperglycaemic Treated (IT) with 400 mg/kg of Alafia barteri aqueous leaf extract. Hyperglycaemic was induced in Groups 3 and 4 by a single dose of intraperitoneal injection of Alloxan at 120 mg/kg. Administration was done for period of nine (9) weeks of experiment; animals were euthanized by cervical dislocation. Pancreas and stomach were excised following abdominal incision and fixed in 10% formol saline for histological studies using H/E and Masson Trichrome stains.
Aqueous leaf extract of Alafia barteri caused significant reduction (p-value<0.05) in blood glucose among Induced Hyperglycaemic Treated (IT) animals. Pancreatic islet demonstrated complete loss of cells and abnormal widening of the spaces between the pancreatic acini cell among the hyperglycaemic untreated group when compared to the normal control group. Lesion of the epithelia lining of the mucosa layer in the stomach was observed as associated complication among hyperglycaemic untreated group. However, in aqueous leaf extract of Alafia barteri treated hyperglycaemic group histological integrity of the islet and the mucosal lining were preserved in the pancreas and the stomach.
In conclusion, the leaf extract of Alafia barteri causes reduction in blood sugar, and maintained the histology integrity of the pancreas and stomach in Alloxan induced diabetic rats.
Intraperitoneal; Alloxan; Alafia barteri; Diabetes mellitus; Pancreas
Diabetes is a chronic disease characterized by high blood glucose levels and abnormal metabolism of carbohydrates, proteins, and fat associated with a relative or absolute insufficiency of insulin secretion and with various degrees of insulin resistance. Diabetes mellitus is ranked seventh among the leading causes of death and third when fatal complications are considered. Diabetes mellitus is characterized by a dysfunction of the pancreas, often in combination with reduced insulin sensitivity [1].
Hyperglycaemia and defective metabolism of glucose and lipids lead to an elevation of blood glucose due to relative or absolute deficiency of insulin. Complications in diabetes result from hyperglycaemia, causing oxidative stress in tissue and are usually accelerated by the depletion of anti-oxidants, as reported by Sicree et al. [2] and Adeeyo et al. [3] who noted reduction in glucose level and improved the health status in hyperglycaemic conditions.
Diabetes mellitus is a metabolic disease characterized by hyperglycaemia resulting from defects in insulin secretion, insulin action, or both [3,4]. The chronic hyperglycaemia of diabetes is associated with long-term damage, dysfunction, and failure of various organs, especially the eyes, kidneys, pancreas, nerves, heart, and blood vessels. Several pathogenic processes are involved in the development of diabetes range from autoimmune destruction of the β-cells of the pancreas with consequent insulin deficiency to abnormalities that result in resistance to insulin action. Treatment of diabetes ranges from insulin for type1 and insulin and antihypoglycaemic drugs like metformin and daonil for type 2, however, emphasis have placed on the use of botanicals that have been proven to have some anti-hyperglycaemic effects. Alafia barteri is valued for its efficacy in the traditional medicine system in Nigeria and other African countries, as an anti-inflammatory and fever remedy. The infusion of the leaves and twining stem are used for the treatment of inflammation and fever [5]. The extracts of the leaves were found to have antibacterial and antifungal activities [6,7]. The aqueous leaf extract was reported to display potent antiplasmodial activity [8]. There is lot of information on the anti-inflammatory, antifungi, and anti-bacteria activities of Alafia barteri leaf extracts, but there is inadequate information on the effect this botanical has on Hyperglycemia and histology of the pancreas and stomach, hence the need for this study.
The aim of this study is to elucidate to some extent the effect of Alafia barteri on the pancreas and the stomach of hyperglycaemic animals.
Experimental animals
Twenty Wister rats weighing between 75-120 g were used for the experiments and were acclimatized for 2 weeks at the ambient temperature of 25 ± 20ºC and 45%-55% relative humidity with a 12 hr light/dark circle. They were fed with standard pellets and water ad libitum.
Collection of plant
Fresh leaves of Alafia barteri where collected at soun’s palace Ogbomoso. Identification and authentication of the leaves were carried out at the Department of Pure and Applied Biology. Ladoke Akintola University of technology, with voucher number: LHO 407.
Extraction of plant
Extraction of Alafia barteri was done at the Chemistry Laboratory, University of Ilorin, Kwara State. The fresh leaves collected from the host tree were washed, air dried in the laboratory at room temperature (29-30ºC) and milled to a course powder using a grinder. Two hundred grams (200 g) of fine powder form of Alafia barteri was weighed into a clean glass ware containing 2000 mL of distilled water and was vigorously shaken in order to have a proper mixing and extraction of the solvent. The mixture was left for about 24 hrs, after which it was centrifuged and filtered to obtain the aqueous filtrate. The filtrate was then steam- dried and weighed 200 g. This was preserved in refrigerator for use through the period of the study.
Experimental design
A total of Twenty (20) Wister rats were taken to the Faculty of Basic Medical Sciences, College of Health Science for acclimatization for two weeks. The animals were given food and water freely. At the end of acclimatization, the animals were grouped in four, containing five animals per group. Group1 was made of five (5) normoglycemic rats that receive 2 mL of water, Group 2 constituted five (5) normoglycemic rats that received 400 mg/kg of Alafia barteri, Group 3 were diabetic (hyperglycaemic) rats that received 2 mL of distilled water, Group 4 were diabetic rats that received 400 mg/kg Alafia barteri. Animals were treated daily at 9:00 am for 42 days. Animals were weighed at alternate days using the weighing balance. The blood glucose of animals was measured every week using glucometer.
Induction of hyperglycaemia
Hyperglycaemia was induced in twenty-seven overnight-fasted randomly selected rats by a single intraperitoneal administration of Streptozotocin at 70 mg/kg bw. Streptozotocin (STZ) was dissolved in citrate buffer (0.1M, pH 4.5) just prior to injection. Hyperglycemia was allowed to develop for 72 hrs. Animals with Fasting Blood Glucose ≥ 250 mg/dL were considered hyperglycemic and were included in this study. Control animals (n=9) received a single intraperitoneal injection of 0.1M citrate buffer (1 mL/kg bw; pH 4.5)
Administration
The route of administration was orally using oral cannula, administration of Alafia barteri was done for 42 days (6 weeks). At the end of the 42 days of treatment, the animals were sacrificed by cervical dislocation; the pancreas and the stomach were removed, weighed and fixed in 10% formol saline.
Measurement of blood glucose
Blood glucose was estimated in overnight fasted rats at 9:00- 10:00 hrs using Glucose oxidase method of one touch ultra 2 glucometers. Blood was obtained from the median caudal vein of the tail by snipping the tip of the tail. The blood glucose was monitored weekly from two weeks (acclimatisation period) before the induction of hyperglycaemia and for six weeks of treatment.
Measurement of body weight
Body weight of the rats was recorded two weeks (acclimatization period) prior to induction of hyperglycemia and on a daily basis during treatment for six weeks. Weight was taken with a weighing scale.
Measurement of relative pancreatic weight
The relative pancreatic weight was recorded as the ratio of the pancreatic weight to that of the experimental rat using sensitive weighing balance (Sony F3G brand).
Measurement of relative stomach weight
The relative weight of the stomach was recorded as the ratio of the stomach weight to that of the experimental rat using sensitive weighing balance (Sony F3G brand).
Termination of treatment
After six (6) weeks the animals were sacrificed by euthanasia. Laparotomy was performed; Pancreas and Stomach were harvested, rinsed in phosphate buffered solution and fixed in 10% formol saline for histological procedures namely; Haematoxylin and Eosin stain, Masson’s Trichrome staining to demonstrate degree of fibrosis and morphological changes.
Histological technique
Tissue processing for light microscopy: Paraffin Wax embedding of the pancreas and stomach were done. At euthanasia, each organ was rinsed in PBS, trimmed free of adipose tissue, cut into smaller pieces (3x3 mm) and fixed in 10% formosaline, for a maximium of 48 hrs. Dehydration of the sample was done in graded alcohol, clearing of the sample in two (2) changes of Xylene I and II, Wax Infiltration of the sample and Embedding (Inclusion) of the sample was done in paraffin wax. 5 μm thick sections of the pancreas and stomach were cut on a Reichert-Jung 2050 rotary microtome (Cambridge Instruments, Germany). Sections were floated on the water bath at 50ºC, and mounted on pre-washed, sterilized, 25.4x76.2 mm glass slides (Pearls, China). Slides were washed with detergent, rinsed with deionised water, and sterilized with 70% ethanol. Mounted Paraffin sections of the pancreas were stained in Haematoxylin and Eosin, Masson’s Trichrome Staining and Gomori Staining to demonstrate the type of connective in pancreas while stomach was stained using H/E stain.
Photomicrography: Photomicrography of histological sections of the pancreas were taken with a Leica Microscope (Tokyo, Japan) coupled with desktop and CPU taken at the Histopathology Department, University of Ilorin Teaching Hospital, Ilorin.
Statistical analysis: Descriptive and inferential statistics were carried out as follows: Data were analysed using Excel 2007 (Microsoft Corporation, USA). Data were expressed as mean ± standard error of the mean (mean ± SEM). Mean values were compared using student t-test. P values less than 0.05 (p<0.05) were taken to be statistically significant. All graphs were drawn with Excel 2007 and graph pad prism 5 (Microsoft Corporation USA).
Weight distribution of animals
Normoglycemic group (Group 1) was observed to have increased body weight throughout the treatment period except for the 4th and 6th week. Normoglycemic (Group 2) rats that received 400 mg/ kg of Alafia barteri showed increased body weight throughout the treatment period except for the 6th week. Group 3 (hyperglycaemic) rats treated with 2 mL of distilled water exhibited increased body weight, which decreases relatively in 6th week. Group 4 (Diabetic rats) that received 400 mg/kg Alafia barteri increase in body weight throughout the treatment period (Figure 1).
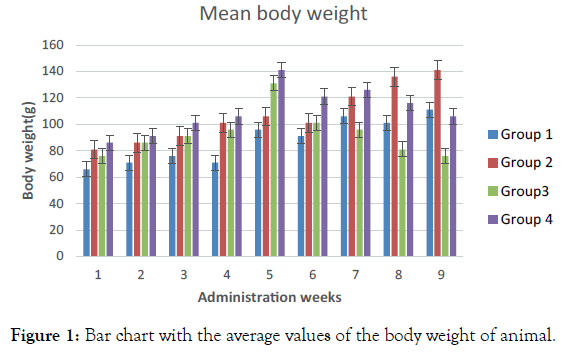
Figure 1. Bar chart with the average values of the body weight of animal.
Administration of Alafia barteri aqueous extract significantly caused increase in the body weight of the animals relative to the control animals that received only distil water. Induction of hyperglycaemic in rats resultantly decreased the body weight as observed among the hyperglycaemic untreated animals in Group 3 shown in Figure 1 above. However, hyperglycaemic rats treated with Alafia barteri aqueous extract increased body weight relative to the untreated hyperglycaemic animals.
A sustained increased in the blood glucose level among the hyperglycemic untreated animals’ Group 3, throughout the administration periods as revealed (Figure 2). Significant reduction in the glucose level among the hyperglycemic animals treated with Alafia barteri aqueous extract was observed from the 3rd week through the administrative period. However, blood glucose level among the control animals and those treated with Alafia barteri aqueous extract was maintained as shown above.
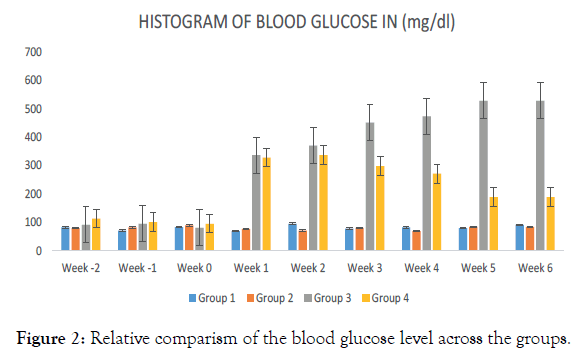
Figure 2. Relative comparism of the blood glucose level across the groups.
The aqueous leaf extract was reported by Lasisi et al. [8] to display potent antiplasmodial activity, while, Hamid and Aiyelaagbe, 2011 showed that the extracts of the leaves have antibacterial and antifungal activities. This study revealed that as shown in Figure 2 Alafia barteri aqueous leaf extract lowered the blood glucose levels of experimental rats. The control (Group 1) did not show any significant variation in blood glucose with Group 2 (normoglycaemic treated). However, hyperglycaemic induced (Group 3) animals which were given distilled water had their blood sugar elevated from week 4 (after induction) to the end of the experimental period (Figure 2). Administration of Alafia barteri aqueous extracts in hyperglycaemic treated with 400 mg/kg of aqueous extract had their blood glucose reducing from the 7th week and that reduction was maintained throughout the experimental period. This study is consistent with the findings of who documented that some plants like Moringa Olifera, Allvin cepa had some antihyperglycaemic effect.
Also traditionally it has been documented that Alafia barteri has some antihyperglycaemic effect [9]. The ability of Alafia barteri to reduce high blood sugar in this animals were supporting the traditional claim above.
In this study, weights of the animals in various groups were compared. The weight of the animals in Group 1, 2 and 4 significantly increased throughout the experimental period (Figure 1). It has been documented that diabetes state is associated with weight lost decrease in weight of the animals of Group 3 (untreated hyperglycaemic group) was in agreement with the above motion [10]. Although the decrease was not significant when compared with other groups.
Hyperglycaemia is usually associated with loss of β cells and fibrosis in the islet of Langerhans of pancreas, in line with this study, the animals in Group 3 had their islet disorganised as shown in plate 3,7 and 11 [11] (Figure 3). Loss of cells, scattered fibrosis which is in agreement with the above findings about the islet cells and hyperglycaemia. Plate 4,8, and 12 showed the islet of Group 4 animals which were hyperglycaemic and treated with Alafia barteri leaf extract there were loss of cells, distorted cytoarchitecture and some degree of fibrosis but not as evident as it was in Group 3 animals. The loss of islet cells in the pancreas of Group 3 animals must have been as a result of released of excessive oxygen radicals and superoxide that are usually associated with hyperglycaemic state. The rejuvenation of the cytoarchitecture of the islet of Langerhans in the Group 4 animals might be due to ability of Alafia barteri to reduce the blood sugar during the experimental period, thus reducing the amount of oxygen radicals and superoxide released. Also Alafia barteri might have some insulin like activity thus ability to maintain normal glucose level. Some plants like citrus medicus, mistletoe had been documented to have insulin like activities and were able to prevent hyperglycaemia in experimental animals. This histological alteration of the pancreas is associated with the lesion of the stomach mucosal lining and the vaculation of the submucosa layers as shown in plate 15 among the hyperglycaemic rats, however, administration of Alafia barteri maintained the stomach integrity; the lining of the mucosal and the presence of the mucus glands.
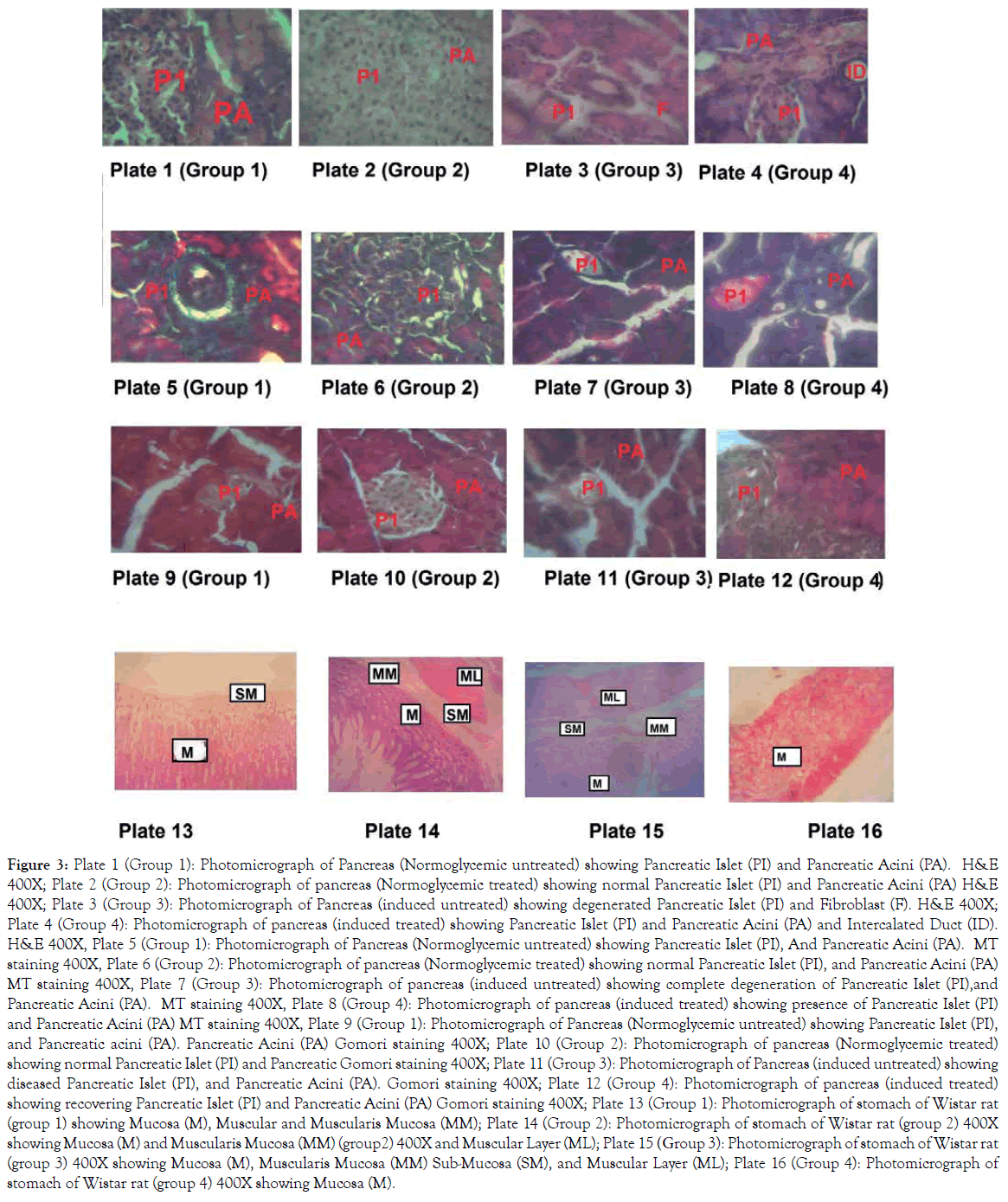
Figure 3. Plate 1 (Group 1): Photomicrograph of Pancreas (Normoglycemic untreated) showing Pancreatic Islet (PI) and Pancreatic Acini (PA). H&E 400X; Plate 2 (Group 2): Photomicrograph of pancreas (Normoglycemic treated) showing normal Pancreatic Islet (PI) and Pancreatic Acini (PA) H&E 400X; Plate 3 (Group 3): Photomicrograph of Pancreas (induced untreated) showing degenerated Pancreatic Islet (PI) and Fibroblast (F). H&E 400X; Plate 4 (Group 4): Photomicrograph of pancreas (induced treated) showing Pancreatic Islet (PI) and Pancreatic Acini (PA) and Intercalated Duct (ID). H&E 400X, Plate 5 (Group 1): Photomicrograph of Pancreas (Normoglycemic untreated) showing Pancreatic Islet (PI), And Pancreatic Acini (PA). MT staining 400X, Plate 6 (Group 2): Photomicrograph of pancreas (Normoglycemic treated) showing normal Pancreatic Islet (PI), and Pancreatic Acini (PA) MT staining 400X, Plate 7 (Group 3): Photomicrograph of pancreas (induced untreated) showing complete degeneration of Pancreatic Islet (PI),and Pancreatic Acini (PA). MT staining 400X, Plate 8 (Group 4): Photomicrograph of pancreas (induced treated) showing presence of Pancreatic Islet (PI) and Pancreatic Acini (PA) MT staining 400X, Plate 9 (Group 1): Photomicrograph of Pancreas (Normoglycemic untreated) showing Pancreatic Islet (PI), and Pancreatic acini (PA). Pancreatic Acini (PA) Gomori staining 400X; Plate 10 (Group 2): Photomicrograph of pancreas (Normoglycemic treated) showing normal Pancreatic Islet (PI) and Pancreatic Gomori staining 400X; Plate 11 (Group 3): Photomicrograph of Pancreas (induced untreated) showing diseased Pancreatic Islet (PI), and Pancreatic Acini (PA). Gomori staining 400X; Plate 12 (Group 4): Photomicrograph of pancreas (induced treated) showing recovering Pancreatic Islet (PI) and Pancreatic Acini (PA) Gomori staining 400X; Plate 13 (Group 1): Photomicrograph of stomach of Wistar rat (group 1) showing Mucosa (M), Muscular and Muscularis Mucosa (MM); Plate 14 (Group 2): Photomicrograph of stomach of Wistar rat (group 2) 400X showing Mucosa (M) and Muscularis Mucosa (MM) (group2) 400X and Muscular Layer (ML); Plate 15 (Group 3): Photomicrograph of stomach of Wistar rat (group 3) 400X showing Mucosa (M), Muscularis Mucosa (MM) Sub-Mucosa (SM), and Muscular Layer (ML); Plate 16 (Group 4): Photomicrograph of stomach of Wistar rat (group 4) 400X showing Mucosa (M).
This study confirms again the hyperglycaemic activities of Allosan and the complicated associated effects on the gastric mucosa and the mucus gland. However, Alafia barteri leaf extract has a therapeutic effect that alleviates hyperglycaemia and maintained the histological features of the pancreas and the gastric lining. The actual chemical compound that is responsible for this effect calls for further investigation.
Citation: Atilade AO, Ruth FA, Magret EO, Joseph DB, Adetunji OW, Segun EA, et al. (2019) Diabetes and Its Complication: Aqueous Leaf Extract of Alafia barteri Maintained the Pancreatic and Gastric Integrity in Allosan Induced Diabetic Rats (Rattus Novergicus). Endocrinol Metab Syndr 8:299.
Received: 13-Feb-2019 Accepted: 08-Mar-2019 Published: 15-Mar-2019
Copyright: © 2019 Atilade AO, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.