Andrology-Open Access
Open Access
ISSN: 2167-0250
ISSN: 2167-0250
Research Article - (2025)Volume 14, Issue 6
Background: Prostate Cancer (PCa) has a wide range of diversity and uncertainty in tumor progression. Despite the availability of multidisciplinary therapy, the mortality of patients with advanced PCa still remains high. In-depth research of the intercellular communications in the Tumor Microenvironment (TME) will improve our understanding of the tumorigenesis mechanism of PCa and bring up potential therapeutic targets to patients with PCa.
Methods: We integrate data including bulk RNA sequencing, single-cell RNA sequencing and spatial transcriptomics to investigate the intercellular communications between FAP+fibroblasts and tumor-associated macrophages in PCa. Immumohistochemical and immunofluorescence staining were performed for verification of the analysis.
Results: We analyzed single-cell sequencing data encompassing 23,519 cells across 23 prostate samples, including normal and tumor tissues. A remarkable increase in the number of FAP+fibroblasts within tumor tissues was observed. By analyzing spatial transcriptomics and bulk RNA sequencing data, we observed a notable correlation between FAP+fibroblasts and tumor- associated macrophages (particularly SPP1+macrophages). Compared to normal tissues, tumor tissues exhibited specific intercellular communication between FAP+fibroblasts and SPP1+ macrophages, including CSF1/CSF1R, CXCL/ACKR1 and other tumor-promoting signaling pathways, which could potentially reshape the TME in an unfavorable manner.
Conclusion: Our research identified a unique pattern of interactions between FAP+fibroblasts and SPP1 +macrophages in PCa, providing potential therapeutic targets for the treatment of patients with PCa.
Single cell RNA-sequencing, Spatial transcriptomics, Prostate cancer, Macrophages, Fibroblasts, FAP, SPP1
Prostate Cancer (PCa) constitutes one of most prevailing malignancies in the male urinary system which prominently contributes to high mortality rates in old men around the world. Nowadays, the treatment strategies of prostate cancer mainly include surgery, chemotherapy, radiotherapy, endocrine therapy, immunotherapy and so on. However, due to the concealed biological behavior of PCa and the absence of valid early screening methods, most PCa patients are diagnosed at advanced stage and more than 30% of patients experience biochemical recurrence after treatment, severely impacting the survival time and quality of life of PCa patients. Therefore, there is an increasing demand for biomarkers and therapeutic targets that can accurately predict the prognosis of PCa and guide treatment decisions, ultimately improving the prognosis of PCa [1].
Recent years, the development of novel nanomaterials and nanoparticles has provided a promising carrier for tumor drug delivery system. However, previous treatments targeting cancer cells have encountered various challenges like tumor metastasis, recurrence and medicine resistance. Therefore, concept of the Tumor Microenvironment (TME is introduced to improve treatment strategy of PCa. Currently, modulating the TME has become an alternative approach to cancer treatment and has already exhibited certain achievements.
The TME refers to the complex ecological system composed of cells besides tumor cells and molecules produced within the TME. The sustained interaction between tumor cells and TME contributes to the initiation, progression, metastasis and drug resistance of tumors. Cancer-Associated Fibroblasts (CAFs and Tumor-Associated Macrophages (TAMs make up the majority of TME. Previous studies have shown that there is extensive interaction between CAFs and TAMs, which plays a significant role in the progression of tumors. However, due to technical limitations, the complex communication between CAFs and TAMs have not been fully identified. With the rapid development and wide spread of single-cell RNA sequencing (scRNA-seq technique, we are able to explore the complex types of cells and their intricate interactions within the TME at singlecell resolution now. However, scRNA-seq loses the spatial information of cells and it is difficult to accurately reveal the complex cellular interactions within the TME solely relying on scRNA-seq [2].
The emergence of Spatial Transcriptomics (ST technology allows us to flexibly explore gene expression, cell differentiation and cell-cell communications among cells without losing their spatial information. Therefore, the integrational analysis of scRNA-seq and ST not only enables efficient exploration of the functions and interactions of different cellular subgroups within the TME but also allows for the investigation of spatial heterogeneity within tumor tissues, which greatly enhances our understanding of the TME.
The interactions between CAFs and TAMs have garnered extensive attention. However, due to the complexity of TME, the sub-classification of these cells and their specific interactions remain unclear, necessitating further exploration and research. Our study delves into the interactions between CAFs and TAMs in PCa by employing an integrated analysis of bulk RNA sequencing, scRNA-seq and ST [3]. We observed a significant increase of the number of FAP+fibroblasts in PCa tissues compared to normal tissues, with a strong correlation to TAMs, especially SPP1+macrophages. ST analysis revealed a colocalization of FAP+fibroblasts and SPP1+macrophages in PCa tissues, which could enhance the invasiveness of the tumor. Within the tumor tissue, the biological behavior of FAP+ fibroblasts is altered, acquiring characteristics that promote tumor growth. FAP+fibroblasts enhance the invasive properties of SPP1+macrophages via signaling pathways including FN1/ Integrins and CSF/CSF1R, while TAMs reciprocally amplify the pro-tumoral phenotype of FAP+fibroblasts through pathways such as CXCL/ACKR1. In summary, by integrating multi-omics data of PCa, we unveiled the intricate interactions between FAP +fibroblasts and TAMs (particularly SPP1+macrophages within the PCa TME, providing new strategic targets for PCa treatment.
Single cell RNA sequencing analysi
The scRNA-seq data processing was conducted using the Seurat package (v4.0.2) in R (v4.0.5). metadata row names were updated with sample information, followed by the merging of 23 selected samples into a single seurat object. This object then underwent filtration and normalization, retaining cells with over 800 genes expressed and less than 25% mitochondrial genome reads. Batch effects were mitigated using the harmony method (v0.1.0), facilitating the integration of seurat objects into a unified dataset. Analysis including UMAP and clustering was carried out on this dataset, focusing on the top 3000 variable genes and employing 1-40 principal components [4].
Differential-expression analysis and cell identification
We identified Differentially Expressed Genes (DEGs) in cell clusters from scRNA-seq data by the program of find all markers. Genes exhibiting positive expression in over 25% of cells within any given cluster were chosen. For functional analysis of cell clusters, cluster Profiler package (v3.18.1) was adopted for GO enrichment analysis, with a significance threshold set at p<0.05 and |avg_logFC|>1.5
Spatial transcriptomics data analysis
To process and visualize the ST data, seurat was utilized. The SCT method was employed for ST data normalization and the integration of ST data was achieved through functions such as select integration features, prep SCT integration, find integration anchors and integrate data. We then applied an unsupervised clustering approach to group similar ST spots. The annotation of cell populations was informed by Hematoxylin and Eosin (HE) stained slices and the highly variable genes across clusters. For calculating cell-specific signature scores from scRNA-seq, we employed five methods: AUCell, UCell, singscore, ssGSEA and AddModuleScore. Visualization of cell expression levels in ST data was conducted using spatial dim plot and spatial feature plot [5].
Charaterization of cell-type infiltration
To assess the infiltration level of cell types in ST sequencing data and the TCGA cohort PRAD, we incorporated the top 20 DEGs from scRNA-seq along with wildly acknowledged immune cell gene sets. The ssGSEA algorithm was employed to calculate scores for angiogenesis and cytokine-interaction signatures in GSVA package (v1.38.2). For mapping the distribution of FAP+ fibroblasts and SPP1+macrophages in ST sections, we utilized the top 20 DEGs from each cluster, applying methods including AUCell algorithm, UCell algorithm, singscore algorithm, ssGSEA algorithm and add module score function. Additionally, to examine the expression difference of FAP in tumors versus normal tissues across various cancer spicies, the online platform TIMER was employed [6].
Correlation analysis
To explore the relationships of the cell types we have allocated, correlation analysis and visualization were conducted using the R packages of ggstatsplot (v0.10.0 and corrplot (v0.92. Moreover, the online tool TIMER identified the correlations between immune infiltration and gene expression level within the PRAD cohorts. A p-value of less than 0.05 was defined statistically significant.
Survival analysis
To examine the impact of specific cell populations on clinical outcomes, survival analyses were performed on the TCGA cohort PRAD, along with GSE70768 and GSE70769 datasets, using the Survival package (v3.2-10 and survminer (v0.4.9. The infiltration levels of cell populations, based on the top 20 DEGs from scRNA-seq, were quantified with the ssGSEA algorithm. Patients were then stratified into groups with high or low levels of infiltration using the median value as the cutoff. Kaplan- Meier survival curve was generated with the survfit function, considering a p-value <0.05 as statistically significant.
Quantitatively analysis of cell-cell communications using cell chat the cell chat
package (v1.1.3 was employed to explore intercellular communications, particularly between cancer associated fibroblasts and tumor infiltrated myeloid cells, through the construction of a regulatory framework centered on ligandreceptor interactions. We excluded cell communications with less than 10 cells per specific subgroup. The net visual function facilitated the visualization of interaction patterns. For calculate and display the contribution of single pair to the whole activity of the signaling pathways, the net analysis contribution function was used. The expression levels of each pair of ligands and receptors in specific signaling pathways across cell clusters were illustrated by violin plots via the plot gene expression function. In determining the quantity of co-communication modes within the cell chat object, the non-Negative Matrix Factorization (NMF algorithm was applied, following the use of the identify communication patterns function to discern primary signals and communication patterns among cell groups. Furthermore, network centrality scores were computed using the net analysis centrality function. The net analysis signaling role network function was utilized for the graphical representation of network roles, aiding in the identification of predominant senders, receivers, mediators and influencers within the inferred networks [7].
Immunohistochemical technique
The immunohistochemical kit (Maixin biotechnology, China were used for the experiment. Firstly, human prostate tissue sections were dewaxed to water. The antigenic sites were exposed by microwave repair using sodium citrate antigenic repair solution. Subsequently, endogenous peroxidase blockers were applied to inactivate endogenous peroxidases. Following that, the slices were blocked in non-specific stain blockers for 30 min at 37â. After adding primary antibodies (FAP, Rabbit, JA56-11, HUABIO, 1:1000), they were stored for incubation overnight at 4°C. The following day, the sections were incubated with an HRP-conjugated secondary antibody for 20 min at room temperature. Diaminobenzidine (DAB, Maixin biotechnology, China) was used as the chromogen and nuclei were stained with hematoxylin. The expression of FAP was observed under microscope and the presence of brown-yellow or brown-yellow particles in the cells was the positive staining sign. The score was based on the percentage of positive stained cells and the intensity of the stain and the two scores were multiplied to obtain the total score. According to the total score, the samples were divided into low expression group and high expression group.
Immunofluorescence staining
Tissue was infiltrated with 0.5% triton X-100 for 10 minutes. A non-specific stain blocker (Maixin biotechnology, China) was added to the slices and incubated at room temperature for 10 minutes. FAP antibodies (rabbit, JA56-11, HUABIO, 1:500) and SPP1 (mouse, MH49021, Abmart, 1:300) were incubated overnight at 4°C. Anti-mouse fluorescent secondary antibody (SA00013-1, proteintech, 1:500) and anti-rabbit fluorescent secondary antibody (SA00013-4, proteintech, 1:300) were incubated at room temperature for 2 hours away from light. The cell nucleus was re-dyed with 4',6-Diamidino-2-Phenylindole (DAPI) reagent (E-IR-R103, elabscience, China) at room temperature, away from light, for 10 minutes. Finally, the sealing solution of anti-fluorescence quench agent was added to the slide and the cover glass was covered. Taking pictures under a confocal microscope (LSM880, Zeiss, Jena, Germany) [8].
Single-cell transcriptomics atlas of PCa and normal prostate tissues to characterize the diverse single-cell environments in PCa, we combined two single-cell datasets previously published, encompassing 19 samples from tumor cores and 4 from normal prostate tissue, totaling 23,519 cells for analysis. This dataset included 12,145 cells from tumor tissues and 11,374 from normal tissues.
Based on established gene markers, we categorized the cell populations into nine types: Epithelial cells (12,747), identified by KRT8 and EPCAM expression; endothelial cells (2,144), expressing VWF and PECAM1; fibroblasts (855), marked by DCN, COL1A1 and COL1A2; myeloid cells (2,123), defined by LYZ; T cells (4,054), with CD3D and CD3E; neutrophils (397), expressing S100A9; B cells (271), identified by MS4A1; smooth muscle cells (824), marked by ACTA2; and NK cells (134), characterized by KIT expression.
The distribution of these nine cell types varied across different tissues, with myeloid cells, B cells, neutrophils, NK cells and T cells showing higher infiltration in tumor regions than in normal tissues, highlighting the heterogeneous microenvironment of PCa compared to normal prostate tissue (Figure 1).
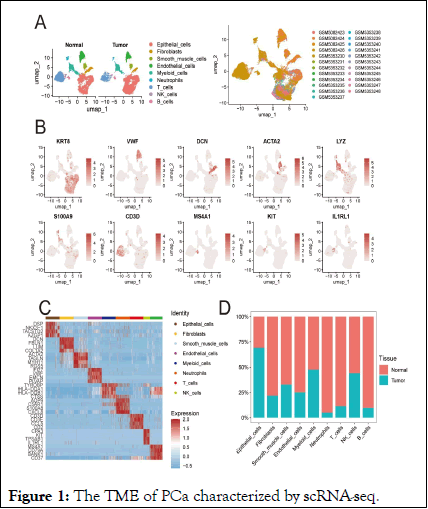
Figure 1: The TME of PCa characterized by scRNA-seq.
Note: (A) UMAP plots profiled the integrative analysis of scRNA-seq samples. (B) Feature plots showed cell markers in each cluster. (C) Heatmap visualizes the top 4 Differentially Expressed Genes (DEGs) of each cell type. (D) Stacked barplots showing the fractional composition of cell numbers for different clusters in normal and tumor tissues.
Tumor microenvironment contributes to the malignant characteristics of cancer-associated FAP+fibroblasts CAFs have been identified as the most abundant stromal cells in TME and are recently recognized as occupying the highest tier in the hierarchy of cellular interactions. However, due to the complex heterogeneity of TME, the specific identity of these cells remains a challenge. To better elucidate the subtypes of fibroblasts in prostate tissue, generally acknowledged fibroblast markers, highly variable features and functional enrichment analyses were integrated for subcluster annotation purposes (Figure 2A, B). Marker genes of FAP, MFAP5, CD74 and MCAM were used to identify the subclusters of fibroblasts. Accordingly, the fibroblasts were classified into FAP+ fibroblasts, MFAP5+ fibroblasts, CD74+fibroblasts, MCAM fibroblasts by the high expression degree of these marker genes [9].
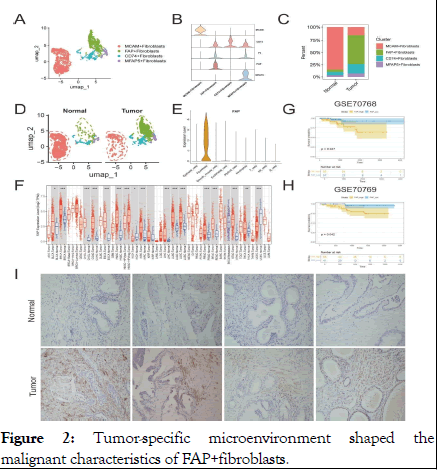
Figure 2: Tumor-specific microenvironment shaped the malignant characteristics of FAP+fibroblasts.
Note: (A) UMAP plot of scRNA-seq of fibroblasts in PCa. (B) Violin plot of fibroblast subclusters marker genes. (C) Bar plots show the percentage of each fibroblast subtypes in scRNA-seq. (D) UMAP plots of fibroblast subclusters faceted by tissue types. (E) Violin plot shows that the FAP is a fibroblast-specific signature in PCa. (F) Pan-cancer analysis comparing the FAP expression between cancer and para-cancerous tissues by TIMER tool. (G,H) Kaplan Meier analysis of high-FAP group and low- FAP group about OS in in two PCa corhorts (GSE70768 and GSE70769). (I) IHC analysis show the existence of FAP +fibroblasts in tumor and normal tissues.
Subsequently, we assessed the infiltration levels of each fibroblast subtype across different tissues, noting that FAP+ fibroblasts predominantly resided within tumor regions while MCAM+ fibroblasts were notably observed in normal region indicating their biological heterogeneity (Figure 2C, D). In addition, it is notable to find that FAP was specifically expressed in fibroblasts (Figure 2E). Next, we explore the pan-cancer differential expression of FAP comparing tumor tissues with normal prostate tissues using the online platform TIMER. Our findings indicated an upregulation of FAP in nearly all examined tumor types, with a notable increase in Prostate Adenocarcinoma (PRAD) (Figure 2F). Further analysis into the relationship between FAP and clinical prognosis revealed that elevated FAP expression related to poorer prognosis in PCa patients, which was also evidenced in two independent public cohorts (Figure 2G-H). In conclusion, we speculated that FAP+ fibroblasts may alter the biological properties of the TME through complex cell-to-cell interactions, thus resulting in the poor prognosis of PCa patients [10].
Intercellular communications between FAP+fibroblasts and TAMs revealed by multi- omics analyses.
Recent years, effect of the intercellular communications between CAFs and TAMs on tumor progression has gained more and more comprehensive attention. Therefore, we propose a hypothesis that the anomalous interactions between FAP+ fibroblasts and TAMs could significantly contribute to the aberrant biological activities of FAP+fibroblasts within the PCa TME. Herein, we utilized ST data to evaluate the spatial distribution of FAP+fibroblasts and immune cells within the PCa TME. By analyzing hematoxylin and eosin staining slices and differentially expressed genes of each cell population after clustering, we categorized the ST spots into four primary cell populations: Tumor cells, fibroblasts, epithelial cells and smooth muscle cells (Figure 3A). We used ssGSEA algorithm to evaluate the infiltration of FAP+fibroblasts and various kinds of immune cells with the genelist in ST data (Figure 3B). Coexistence of FAP+fibroblasts and macrophages were found in the area of fibroblasts while part of cytotoxic immune cells like CD8 T cells and NK cells were also enriched in that area. Further, we noticed that regions with high expression of FAP gene highly overlapped with regions with high expression of TAMs characteristic genes (CD68 and CD163), but there was no significant correlation with regions with high expression levels of T cells and B cells cell markers (Figure 3C). To further validate the association between FAP+fibroblasts and TAMs, we utilized bulk RNA sequencing datasets (TCGA cohorts PRAD). ssGSEA was employed to calculate the expression levels of FAP +fibroblasts and macrophages within the TCGA dataset following correlation analysis. We found that the expression of FAP+fibroblasts enhanced with the increased expression of macrophages (Figure 3D). Finally, by means of online tools TIMER, we investigated the relation between FAP expression and immune cell infiltration. We found that FAP had the strongest correlation with macrophages among all types of immune cell (Figure 3E) [11].
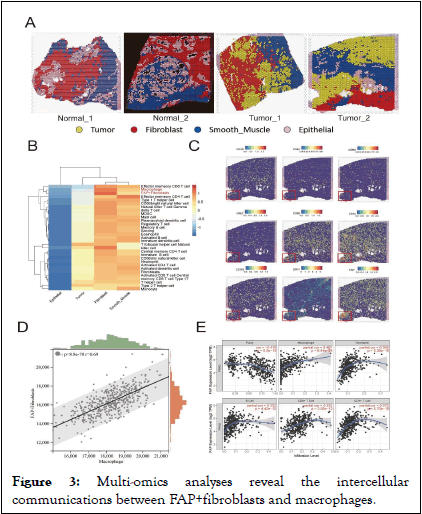
Figure 3: Multi-omics analyses reveal the intercellular communications between FAP+fibroblasts and macrophages.
Note: (A) Unbiased clustering of ST spots and define cell types of each cluster. (B) Heatmap of immune cells and fibroblasts infiltration level scored by ssGSEA algorithm. (C) Gene expression of CD3D, CD8A, GZMA, GNLY, CD68, CD163, CD14, SPP1 and FAP in spatial organization, red boxes mark the representative tumor region of PCa. (D) The positive correlation between FAP+fibroblasts and macrophages in PRAD cohorts visualized by scatter plot. (E) The correlation analysis calculated by TIMER tool shows the highest correlation between FAP and macrophages compared to other tumor infiltrating immune cells.
In conclusion, these findings emphasize the presence of extensive intercellular connection between FAP+ fibroblasts and TAMs, potentially having the function of promoting PCa progression within the TME. Cellular crosstalk between FAP+fibroblasts and SPP1+macrophages are related to poor outcomes in patients with PCa TAMs are predominant tumor-infiltrating immune cells, known for their immunesuppressive and tumor-promoting functions. As mentioned above, we have demonstrated the close distance between FAP + fibroblasts and TAMs in tumor region of PCa. However, it is unclear that which subtype of macrophages communicate with FAP+ fibroblasts. Thus, we further subjected TAMs to dimensional reduction and clustering analysis and divided them into four subgroups. According to our analysis, Dendritic Cells (DCs) were characterized for their high expression of CCR7. Similarly, macrophages featured by high levels of VCAN, SPP1 and C1QC were defined as VCAN+ macrophages SPP1+macrophages and C1QC+macrophages respectively (Figure 4D). The results are consistent with previous studies on TAMs [12].
Then, we obtained gene scoring signatures for different subpopulations of CAFs and TAMs through marker genes from scRNA-seq of PCa. Further, we scored the gene expression data in the TCGA dataset using the ssGSEA method. Subsequently, we determined the specific TAMs subtype interacting with FAP+ fibroblasts through correlation analysis. And the analysis revealed a significant positive correlation between FAP+ fibroblasts and SPP1+macrophages, with the highest correlation coefficient observed (Figure 4E). Additionally, ST data was also analyzed to validate our findings in the TCGA dataset. Scores for individual cell/spot were calculated using five different algorithms including AUCell, UCell, singscore, ssGSEA and AddModuleScore. The average score was calculated and visualized on ST sections. The findings suggested that FAP+ fibroblasts and SPP1+macrophages were coexisted in the fibroblast regions (Figures 4F). In contrast, in normal tissue, there were few SPP1+macrophages surrounding FAP+ fibroblasts, indicating that the interactivity between them primarily occurs within tumor tissues rather than in normal tissues (Figure 4G). These findings were also demonstrated by immunofluorescence of FAP and SPP1 in PCa (Figure 5). The protein encoded by FAP gene is a type of serine protease known as a membrane- associated gelatinase, which is specifically expressed in fibroblasts. The remodulation of the TME by FAP +fibroblasts through reshaping the extracellular matrix have already been reported and the overexpression of FAP+ fibroblasts indicates poor prognosis in various cancers. SPP1+ macrophages are one type of TAMs that may be induced by the hypoxic microenvironment of tumors with immunosuppressive property. The interactions between SPP1+macrophages and FAP + fibroblasts could be a prospective point for anti-tumor progression and metastasis. Clinical value of SPP1+ macrophages infiltration was then assessed in two independent PCa cohorts. It was noticed that low level of FAP+ fibroblasts and SPP1 +macrophages infiltration in patients tend to be with the best prognosis. In contrast, high infiltration level of FAP+ fibroblasts and SPP1+macrophages lead to the poorest outcome in the two cohorts (Figure 4I) [13].
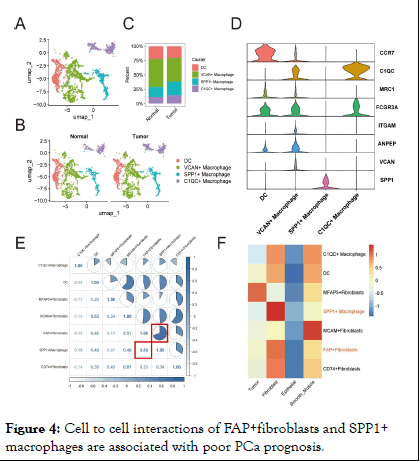
Figure 4: Cell to cell interactions of FAP+fibroblasts and SPP1+ macrophages are associated with poor PCa prognosis.
Note: (A) UMAP plot of tumor-infiltrating myeloid cells colored by cell types. (B) UMAP plots of myeloid cells subclusters grouped by tissue types. (C) Stacked bar plot of myeloid cell proportions in different tissue types. (D) Violin plot showing the expression of spcific genes of subclusters. (E) Complex heatmaps exhibit the correlation between tumor-associated fibroblasts and myeloid cells in PRAD cohort, the correlation between FAP +fibroblasts and SPP1+macrophages marked with red box. (F) Heatmap shows the infiltration level of fibroblasts and myeloid cells scored by ssGSEA algorithm in ST.
To account for confounding factors such as age and pathological staging, we employed the Cox regression method to analyze patients’ prognoses. The results indicated that lower infiltration level of FAP+fibroblasts and SPP1+macrophages represent better outcomes in patients with PCa. This finding supports our hypothesis that abnormal signaling interactions between FAP +fibroblasts and SPP1+macrophages lead to poor prognoses in PCa patients. Additionally, we conducted further analyses to explore the specific intercellular communication pathways between FAP+fibroblasts and SPP1+macrophages. Intercellular communication analysis reveals tumor-specific signaling pathways between CAFs and TAMs in PCa Previous studies have indicated that there are extensive reciprocal intercellular communications between CAFs and TAMs. These interactions make a great contribution to the biological behavior of tumor development, progression and metastasis (Figure 5). To investigate the specific signaling pathways through which the intercellular interactions among these cell clusters lead to a poorer prognosis in patients with PCa, we analyzed the intercellular communication networks between CAFs and TAMs in both normal and tumor tissues. We found that in tumor tissues, FAP+ and MFAP5+fibroblasts have most communication pathways with TAMs (Figure 6A), suggesting that FAP+ and MFAP5+fibroblasts play an important regulatory role within the TME. Compared to the intercellular interactions in normal tissues, we noted several tumor-promoting signaling pathways specificly found in PCa tissues, including ANGPTL, FN1, GDF, CSF, PTN and LAMININ signaling pathways (Figure 6B).
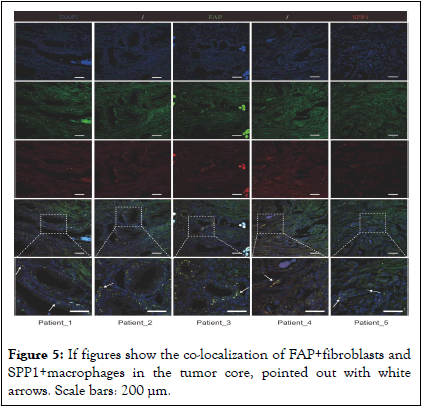
Figure 5: If figures show the co-localization of FAP+fibroblasts and SPP1+macrophages in the tumor core, pointed out with white arrows. Scale bars: 200 μm.
Analysis of the communication patterns of CAFs and TAMs also showed a coordinated tumor-promoting signaling communication model between FAP+ and MFAP5+fibroblasts. Previous literature has also confirmed that these two types of fibroblasts are important driver subpopulations of stromal cells in tumorigenesis. Hence, we infer that there is a coordinated cellular communication pathway between FAP+fibroblasts and MFAP5+fibroblasts, jointly driving the occurrence, development and metastasis of tumors (Figure 6C). Laminin, a crucial component of the extracellular matrix, was highly expressed by FAP+fibroblasts in tumor tissues compared to normal tissues. These fibroblasts release laminin (encoded by LAMB2, LAMC1 and LAMA4…) which interacts with integrin receptors and CD44 on TAMs (Figure 6D and E) [14]. This indicates that laminin released by FAP+fibroblasts may interact with TAMs, resulting in a remodeling of basement membrane, thus modulating tumor dissemination and metastasis. Additionally, FAP+fibroblasts were also observed to secrete FN1 and PTN acting on TAMs and promoting tumor progression (Figure 6D and F).
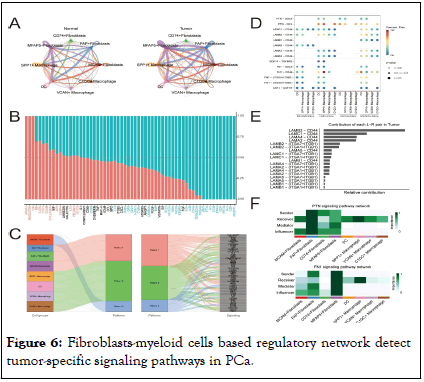
Figure 6: Fibroblasts-myeloid cells based regulatory network detect tumor-specific signaling pathways in PCa.
Note: (A) The circle plots show the overview intensity of cellular communication between fibroblasts and myeloid cells in normal and tumor tissues respectively, arrow width represents the communication intensity. (B) Bar plot of specific signaling pathways between fibroblasts and myeloid cells inferred by cell chat, with red indicating increased signals in normal tissue and blue indicating increased interactions in tumor tissue. (C) The outgoing signaling patterns of certain cells in PCa tissue visualized by alluvial plot. (D) Dot plot of Ligand-Receptor (L-R) pairs of several tumor-specific pathways between fibroblasts (sources) and myeloid cells (receptors). (E) Bar plot shows the relative contribution of each L-R pair to the overall LAMININ signaling network within the tumor tissue. (F) Heatmap shows dominant senders, receivers, mediators and influencers in PTN and FN1 signals of tumor inferred by network centrality score.
In conclusion, our research highlights the complex intercellular communications between FAP+fibroblasts and TAMs that exacerbate PCa malignancy. Targeting these tumor-specific intercellular communication pathways offers a promising therapeutic strategy for treating PCa. FAP+fibroblasts enhance the angiogenesis function of SPP1+macrophages by enhanced IL34/CSF1R and CSF/CSF1R signaling pathways TAMs are an important component of innate immune cells within the tumor microenvironment. Monocytes in the blood are recruited into tumor tissues, where they are induced to differentiate into TAMs. Specific cytokines and growth factors are indispensable for the proliferation and differentiation of TAMs, comprising of Colony Stimulating Factor-1 (CSF1), interleukins and so on. Through cell chat analysis, we found that the CSF pathway was one of the key points in the regulatory network of cell interactions between CAFs and TAMs in the TME [15]. We noticed that SPP1+macrophages primarily receive CSF signals secreted by MFAP5+fibroblasts in normal prostate tissues. However, in PCa, CSF signals from MFAP5+ and FAP+fibroblasts synergistically act on SPP1+macrophages and C1QC+macrophages, indicating a significant enhancement of the CSF signaling pathway in PCa (Figure 7A, C).
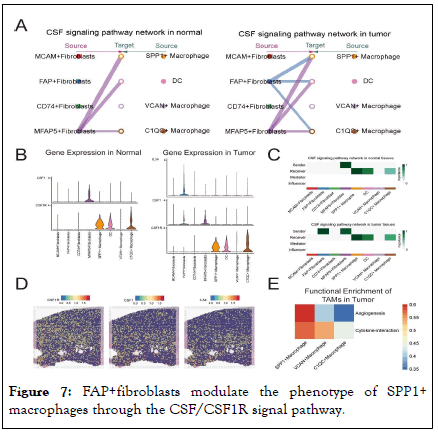
Figure 7: FAP+fibroblasts modulate the phenotype of SPP1+ macrophages through the CSF/CSF1R signal pathway.
Note: (A) The hierarchical plots of the intercellular communication network for CSF signaling. (B) Violin plot of CSF1, and CSF1R expression of tumor-specific fibroblasts and myeloid cells in distinct tissues. (C) Heatmap shows dominant senders, receivers, mediators and influencers in CSF signals of tumor inferred by network centrality score. (D) Spatial images localize the expression of CSF and CSF1R which suggest that the co-localization of CSF and CSF1R in PCa slices. (E) Heatmap showing angiogenesis and cytokine-interaction function scores of three identified tumor-associated macrophage subtypes.
Furthermore, when exploring the ligand and receptor expression of the CSF signaling pathways, it showed that in normal tissues only MFAP5+fibroblasts secrete CSF1, which acting on the CSF1R of SPP1+ and C1QC+macrophages. However, in PCa tissues, CSF factor and IL34 secreted by FAP+fibroblasts together act on the CSF1R of SPP1+ and C1QC macrophages (Figure 7B), suggesting that in PCa, the interaction between FAP + fibroblasts and SPP1+macrophages through the CSF pathway are significantly enhanced and altered. Subsequently, analysis of ST data verified the existence of the CSF1-CSF1R and IL34- CSF1R signaling axes within the PCa TME (Figure 7D). Our analysis (Figure 7E) indicates that SPP1+macrophages could promote angiogenesis in tumor tissues and increase the risk of tumor metastasis. What’s more, our research suggests that the enhancement of the CSF1- CSF1R and IL34-CSF1R signaling pathways might be an important reason for the enhanced angiogenic capabilities of SPP1+macrophages resulting in metastasis and poor prognosis in PCa patients with high infiltration levels of both FAP+fibroblasts and SPP1+ macrophages. Therefore, CSF1 blockade may be an important potential target for interrupting this process and improving the prognosis of patients with PCa.
Positive regulatory loop between FAP+fibroblasts and TAMs attributes to the progression of PCa SPP1+macrophages are reported to be pro-tumorigenic TAMs with the function of complement activation, antigen processing and presentation and so on. They are believed to be induced and regulated by the metabolites, cytokines and other bioactive substances within the TME. However, the specific signaling pathways regulating SPP1 +macrophages have not been fully classified. In our research, we found that FAP+fibroblasts and SPP1+macrophages communicate closely to each other [16].
We then explored whether FAP+ fibroblasts have the ability to regulate the immune responses of SPP1+macrophages. It was observed that FAP+fibroblasts were the unique source of complement C3, which interacts with the receptors ITGAM/ ITGB2, ITGAX/ITGB2 and C3AR1 on SPP1+macrophages within the TME, thereby activating the immune response (Figure 8 A and C). What’s more, we explored the spatial distribution of C3 in ST data of PCa. The results indicated that the area with high expression level of C3 overlapped with the area highly infiltrated by FAP+fibroblsts and SPP1+TAMs (Figure 8D). By analyzing the communication patterns of CAFs and TAMs, we found that FAP+ fibroblasts tend to send signals, while SPP1+macrophages tend to receive intercellular communication information (Figure 8E). Hence, FAP +fibroblasts might abnormally activate SPP1+macrophages through the complement pathway and attribute to the tumorigenesis, progress and metastasis of PCa. Besides the immune regulation functions of FAP+fibroblasts, we also explored the cellular interactions dominated by TAMs. We found that C-X-C Motif Chemokine Ligand (CXCL) signaling was remarkably upregulated in TAMs compared with macrophages resident in normal prostate tissues. CXCL chemokines (CXCL1, CXCL2, CXCL3 and CXCL8) secreted by TAMs bind to the ACKR1 receptor specificly expressed on FAP+ fibroblasts (Figure 8F-H).
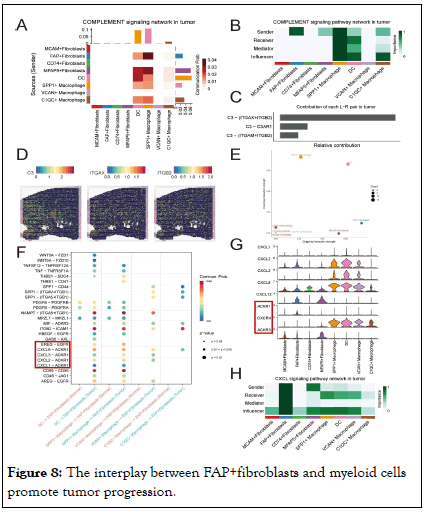
Figure 8: The interplay between FAP+fibroblasts and myeloid cells promote tumor progression.
Note: (A) Heatmap of complement signaling network suggests the FAP+fibroblasts play a crucial role in regulating the functions of SPP1+macrophages and other immune cells in tumor. (B) Heatmap shows dominant senders, receivers, mediators and influencers in complement signals of tumor inferred by network centrality score. (C) Bar plot shows the contribution of all L-R pairs in tumor complement pathway. (D) Spatial transcriptomics visualize the expression of C3, ITGAX and ITGB2. (E) Dot plot shows an increase in signal targeting SPP1+macrophages by FAP+fibroblasts. (F) Dot plot shows the expression of L-R pairs of CXCL and EGF signaling pathway between fibroblasts and myeloid cells in normal (red color) or tumor (green color) tissues. (G) Violin plot of various CXCL ligands expression of tumor-specific fibroblasts and myeloid cells in tumor tissues. (H) Heatmap shows dominant senders, receivers, mediators and influencers in CXCL signals of tumor inferred by network centrality score.
Overexpression of CXCL signal pathway has been found to be one of the hinges on tumor growth, angiogenesis, invasion and metastasis, as well as drug resistance. Thus, the aberrant activation of ACKR1 receptor and its downstream signaling pathway in FAP+fibroblasts could be a partial reason for their malignant action. In conclusion, FAP+fibroblasts could motivate the proliferation and tumor angiogenesis characteristics SPP1+macrophages. Conversely, SPP1+macrophages could also enhance the aggressive phenotypes of FAP+fibroblasts by secreting chemokines including CXCL1, CXCL2, CXCL3, CXCL8 and so on. These reciprocal intercellular communications form a positive regulatory loop attributing to the malignant TME in patients with PCa (Figure 9) [17].
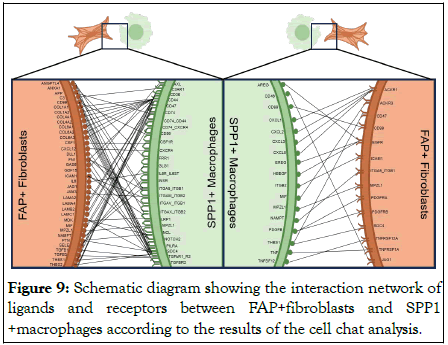
Figure 9: Schematic diagram showing the interaction network of ligands and receptors between FAP+fibroblasts and SPP1 +macrophages according to the results of the cell chat analysis.
PCa is one of the most common cancers of the male urinary system with high and long-term survival. Although carefully monitoring are enough for patients with low-risk and localized primary PCa, the mortality rate for patients with advanced PCa remains high, necessitating a multidisciplinary treatment including surgery, radiation therapy and hormone therapy. It was reported that approximately 15% of patients with prostate cancer are diagnosed with advanced PCa. However, the specific mechanisms driving immune escape and tumor progression in advanced PCa have not yet been elucidated.
Recent years, with the emergence and progress of single-cell RNA sequencing and spatial transcriptomics, the details of TME could be further detected and explored. Intercellular communications networks are an important part of the TME and have been reported to be associated with the malignancy property of cancers. CAFs and TAMs are the most two common types of non-tumor cells in solid cancers with extensive intercellular communications and transformation. It is believed that an in-depth identification of intercellular communications between CAFs and TAMs could help us to find new novel therapeutic targets for PCa. Therefore, we integrated bulk RNAseq, scRNA-seq and ST to explore the detailed intercellular communications between CAFs and TAMs with IF and IHC experiments to validate our findings (Figures 10 and 11) [18].
Figure 10: The interaction network between FAP+fibroblasts and TAMs in PCa TME.
Note: In our study, we demonstrated that FAP+fibroblasts have reciprocal communication with SPP1+macrophages and other macrophage subtypes. The ligands (include PTN, LAMININ, FN1, GDF and CSF) expressed by FAP+fibroblasts that modulate SPP1+macrophages. Mutually, myeloid cells could also release CXCL ligands to reshape the malignant behaviors of FAP +fibroblasts.
Figure 11: Graphic overview of this study design.
Note: Combining bioinformatic analysis and experimental validation, we explore the biological characteristics of FAP +fibroblast and with a particular emphasize on its interactions with tumor infiltrating SPP1+macrophages in PCa. Integrated scRNA-seq (n=23 pairs, normal and tumor tissues), ST (n=4, tumor tissues) and public cohort (PRAD: n=682, GSE70768 and GSE70769) to comprehensively analyze FAP+fibroblasts from multiple perspectives.
The diversity of CAFs significantly influences the proliferation, differentiation and function of tumor infiltrating immune cells. Recent studies have identified several novel fibroblast subclusters with different gene expression and functions such as ACTA2+ myofibroblasts and FAP+fibroblasts. These investigations have highlighted that CAFs are activated fibroblasts typically expressing markers like FAP, FSP and αSMA, which are important in promoting tumor growth and resulting in chemotherapy resistance. FAP+fibroblasts are involved in multiple biological processes including tissue repair, fibrosis and extracellular matrix degradation with the expression of collagenase and dipeptidyl peptidase. It is reported that both cancer and stromal cells undergo hypoxic necrosis rapidly in FAP depleted transgenic mice models of Lewis lung carcinoma or pancreatic ductal adenocarcinoma. However, up to now, there has not been research investigating the effect of FAP+ fibroblasts on the TME of PCa. Therefore, our research focused on the pivotal role of newly identified FAP+fibroblasts in PCa, investigating their biological characteristics and impact on the TME. It is found that FAP+fibroblasts predominantly reside in PCa tissues rather than normal PCa tissues. What’s more, patients with higher level of infiltration of FAP+fibroblasts have poorer prognoses compared to those with lower level of infiltration, indicating their pro-tumoric effects [19].
Increasing evidence indicates that fibroblasts are able to communicate with different immune cells by secreting cytokines, chemokines and other biomolecules, which attributes to the formation of an immunosuppressive microenvironment and facilitates tumor immune tolerance. Kumar V, et al. found that FAP+fibroblasts were associated with the infiltration of immune cells in gastric cancer. In our study, we explored the relationship between FAP+fibroblasts and multiple tumor infiltrating immune cell types in PCa. To ensure the validity of our findings, we integrated multi-omics data, including bulk RNA-seq, scRNA- seq and ST. For the first time in PCa, we identified a significant correlation between FAP+fibroblasts and TAMs.
Regarding the heterogeneity of TAMs, further research is needed to identify the specific subtype of TAMs that interact with FAP 10 +fibroblasts. Through analysis of multi-omics sequencing datasets and validation of IHC and IF experiments, we demonstrated a close interaction between FAP+ fibroblasts and SPP1+macrophages in PCa tissues. Previous research has revealed the immunosuppressive and angiogenesis properties of SPP1+macrophages, which could lead to poorer clinical outcomes in PCa patients. In our research, the survival analysis indicates that patients with high infiltration level of both FAP+ fibroblasts and SPP1+macrophages have the poorest prognosis compared to those with lower infiltration levels of either FAP +fibroblasts or SPP1+macrophages. IHC and IF experiments reveal that FAP+fibroblasts could attach to SPP1+macrophages in border stromal regions, prompting interactions with each other. These findings underscore that the intercellular communications between FAP+fibroblasts and SPP1 +macrophages are crucial in influencing the malignant phenotype and leading to poor prognosis in patients with PCa. Given the important role of FAP+fibroblasts in modulating protumoric signaling pathways within the PCa TME, we performed intercellular correlation analyses to uncover potential tumordriven mechanisms. Our findings identify several tumor-specific interactions between FAP+fibroblasts and TAMs, especially SPP1+macrophages. Our analysis shows that FAP+fibroblasts may activate SPP1+macrophages through the CSF/CSF1R and IL34/CSF1R axis and induce an immunosuppressive microenvironment via GDF15/TGFBR2 signaling.
High infiltration level of FAP+ fibroblasts infiltration also contributes to the formation of a desmoplastic microenvironment through laminin pathways and other intercellular signals creating conditions unfavorable for antitumor immunity. Moreover, coordinated signaling between FAP +fibroblasts and MFAP5+fibroblasts further exacerbate the invasive characteristics of the PCa microenvironment
Reciprocally, activated TAMs also help sustain the activation of FAP+ fibroblasts in a positive feedback loop via CXCL signaling pathway. These communication pathways provide potential new targets for future PCa therapies. There are still some limitations in our study. For example, the sample size we analyzed is relatively small and the biological characteristics of FAP+ fibroblasts are investigated only in PCa. Therefore, it still remains uncertain whether FAP+fibroblasts are a conserved cell type across various cancer types. Previous research has revealed high levels of FAP expression is associated with poor prognosis in patients with colon and bladder cancers. However, further investigations are still necessary to clarify the role and properties of FAP+fibroblasts across different kinds of cancers [20]
In this study, we utilized multi-omics datasets of bulk RNA-seq, scRNA-seq and ST to investigate the detailed landscape of the PCa microenvironment. Our findings provide an extensive insight into the function of FAP+fibroblasts, particularly focusing on their interactions with SPP1+macrophages and other TAMs. This could promote the development of targeted treatment strategies to improve outcomes for patients with PCa.
Xinyu Li: Conceptualization, methodology, formal analysis, investigation, writing-original draft.
Fei Zheng: Methodology, software, validation, formal analysis, investigation, visualization, data curation, funding acquisition.
Zepeng Dong: Methodology, data curation, formal analysis.
Hanchao Liu: Writing-review and editing, funding acquisition.
Tingting Wu: Conceptualization, Writing-review and editing, supervision, project administration, Funding acquisition.
All authors have read and agreed to the published version of the manuscript.
The authors declare that they have no conflict of interest.
All data generated or analyzed in this study are included in the article. Further inquiries can be directed to the corresponding author.
We sincerely thank Shenzhen Qianhai Taikang Hospital for their support of this project.
The authors declare no competing financial interests.
We express our gratitude to the dedicated staff and participants involved in this study for their valuable contributions.
The prostate cancer tissues and adjacent normal tissues used for IHC and IF experiments were obtained from previously collected paraffin-embedded surgical specimens of patients with prostate cancer who had provided informed consent. In order to protect patients’ privacy, their personal information was not collected. This study obtained approval of the ethics committee of the first affiliated hospital of Sun Yat-sen University (approval number: SYSU-IACUC- 2021-000188).
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
Citation: Li X, Zheng F, Dong Z, Liu H, Wu T (2025) Crosstalk between FAP+ Fibroblasts and Tumor-Associated Macrophages in Prostate Cancer Unveiled by Single-Cell Sequencing and Spatial Transcriptomics. Andrology. 14:366.
Received: 19-Sep-2024, Manuscript No. ano-24-34132; Editor assigned: 24-Sep-2024, Pre QC No. ano-24-34132 (PQ); Reviewed: 08-Oct-2024, QC No. ano-24-34132; Revised: 13-Oct-2025, Manuscript No. ano-24-34132 (R); Published: 20-Oct-2025 , DOI: 10.35248/2167-0250.25.14.362
Copyright: © 2025 Li X, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.