Forest Research: Open Access
Open Access
ISSN: 2168-9776
ISSN: 2168-9776
Special Issue - (2021)Volume 10, Issue 2
All living organisms are required to keep the ecosystem alive. One organism cannot exist without other organisms. Biodiversity is important for survival of the human and preserves the ecosystem. Hence biodiversity is called as ‘balance of nature’. It is a hidden treasure that enriches all our lives. Undoubtedly the humans and animals survivability is mainly relying on the ecosystem niche. Indeed, with life support, biodiversity is the diverse range of organisms inhabiting their lives equally in our green planet. Now-a-days due to rapid urbanization, industrialization, modernization and vague policy and planning is drastically affected the biodiversity system, which can accelerate negative impact on quality of life (QOL) of all living organisms. All educational Institutions in India is forefront of fixed biodiversity and vital habitats, due to horizontal and vertical expansion of vegetation area for the purpose of creation of human settlement, construction of roads and bonafide of Government land to some other purposes is inevitability shows habitat loss, which is the largest factor contributing to the current national and global extinction events. Asper the past literature, Institutional biodiversity is alarming stage at national as well as global level; everybody should extend their helping hands to conserve the existing plant and animal species for the benefit of human health concern. In broad perspective ecosystem restoration in small area (Institutional biodiversity) is uniquely valuable tool for environmentalist, policy makers and forest officials for implementation of new policy to improve biodiversity at selected sites of various educational Institutions (smaller area). As long as, the estimation of biodiversity at Institutional level is recursive factor to predict how much vegetation area would be affected by biotic and A-biotic factors. In this proximity, the present study aims to estimate the species richness and floristic diversity at selected sites of KVAFSU regional campus. An observational survey-based study was conducted in the year 2014- 2020 in accordance with standard operating procedure (SOP). Scientifically, the sample measuring was done in a 20 × 20 plot that was made and recorded as an individual species that are present in the demarcated boundary. The cluster sampling method was used to extrapolate the species composition and evenness. As per the research findings, a total 414 (13.66%) [Odds 0.987, p<0.05; 95% CI 10.25-26.38] tree species with 13 family, 8 genera; 10 herb species; 3 family 2 genera and 16 shrubs species besides with 4 family 3 genera was recorded during study period. Suboptimal biodiversity (unusable land) shield with geographical area of 356 acres of land. The human intervention or anthropogenic factor is greatly affected the biodiversity and it was found to be statistically significant (35.0%) [CI -95% 12.55-41.22; Odds 5.22 p=0.0022] followed by pollution (25%) [95% CI 10.68-28.79; Odd 6.31 p=0.0022]; drastic climatic changes (20%) [95% CI 8.63-25.18; odds 3.18 p=0.0016]; habitat degradations (8.0%) [95% CI: 3.16- 10.74 p=0.0008]; Odds 4.86; invasive species (10%) [95% CI: 6.32-12.55; Odds 5.02 p=0.0012]; disease outbreaks (1.0%) [95% CI: 0.32-1.5% Odd 0.96 p=0.4123] etc. The following rare bird species were enlisted during the study period viz Forest owlet-05 (11.11%), Great Indian Bustard-02 (4.44%), Siberian Crane-03 (6.66%), Indian Vulture-01 (2.22%), Bald eagle-03 (6.66%), Indian Rover-04 (8.88%), Spotted owl-05 (11.11%), Wood pecker - 03 (6.66%), Common myna-10 (22.22%), King fisher-02 (4.44%), pipit-01 (2.22%), flycatcher-02 (2.22%), laughing Thrush-03 (6.66%);Mammals; Indian Palm Squirrel-10 (66.66%), Northern Palm Squirrel-05 (33.33%); Reptiles; Western skink-15 (39.47%), common crow 20 (17.51%), Blue tail mole skink-06 (15.78%), Camelian -03 (7.89%),Common Kraits-02 (5.26%),Indian Cobra-10 (26.31%), Viper-02 (5.26%). An approximately 20-28% habitat loss was recorded in the study sites due to anthropogenic factors [odds 6.32; 95% CI 19-36], high level of radiation effect (mobile tower transmit microwave radiation in the range of 869-894 MHz and power density is in vicinity is about 4.7 W/m2 and in 1805-1880 MHz range is 9.2 W/m2, recently 4G network entered in to the city) and pollution (mean CO2 level shoot up to 450-490 ppm). An overall findings of the research, the present study concludes that, the competent authority should take necessary steps to prevent the habitat loss from anthropogenic factors (human intervention) and strictly adhere the National environmental forest policy and also encourage to establish green economy for restoring endangered and rare species at larger extent.
Biodiversity; Species richness; Evenness; Model; Small area estimation
Biodiversity is a total variety of life on earth; it includes all genes, species, ecosystem and the ecological processes of which they are part (Convention on Biodiversity, 1992). The richness in variety and variability of species of all living organisms in a given habitat consulates the restoration of ecosystem and ecological balance Logically, the biodiversity is a variety and differences among living organisms from all sources, including aquatic, terrestrial and agricultural ecosystems etc. The diversity intuition was found on earth around 5000 years ago; it consisted of millions of distinct biological species that were a product of nearly 3.50 billion years of evolution [1-3]. It can be expressed in four levels viz diversity of genes, species and ecosystem settlement. The concept of ecological study is appreciated by the numerous wealth on different biological categories and relative abundance (evenness) of new species. Moreover richness of species extensively helps us to extrapolate the variability between local level (alpha diversity), augmentation of biological habitats (Beta diversity) and variability of landscapes (Gamma diversity). In contrast, the ecological succession is mainly reliant on human and animal intervention; it includes all living things or biological resources and their essential components to maintain the existing or paradoxical changes of floristic diversity [4-7]. Now days due to industrialization, political and human intervention, small area ecosystems shows significant degradation, which can accelerate negative impact on Biological system and ecological balance (MoFR, 2019). There is now a growing realisation that we are able to conserve the bio diversity from hazardous risk through conservation of existing natural resources (plantation and inception of developmental programmes) [8-11]. Many literatures suggest that, the human intervention on existing ecosystem which is significantly associated with sustainability and livelihoods of living [1,12-18]. This means ecological restoration is primary concern for conservation of our native -mother land for restoring culture and beliefs on nature to manage self-sustainability at national and Global level [4].This ecological restoration in small area biodiversity is uniquely valuable tool for environmentalist, policy makers and forest officials for implementation of policy to improve the floristic diversity at selected sites (smaller area) and also which can provide employment and extended economic opportunity for local community. In this proximity, the present study aims to know the species richness and floristic diversity at Regional campus of Karnataka Veterinary Animal and Fisheries Sciences University, Bengaluru (small area biodiversity estimation).
The selected site KVAFSU (B) regional campus is located in Veterinary College Campus it was established on 25th July 1958 affiliated with University of Mysore The Veterinary College was started in a temporary building located in the Mysore Serum Institute by highness of Maharaja of Mysore presidency, now called as the Institute of Animal health and Veterinary Biologicals, Hebbal, Bengaluru. The Veterinary College became a constituent College of University of Agricultural Sciences, Bangalore in 1965. It became a part of the newly established Karnataka Veterinary Animal and Fisheries Sciences University Bidar from April 2005. The campus is spread around 356 acres of total land area allocated in different components; Agriculture and allied service (95 acres) Human settlement (60 acres) and vegetation covers (201acres). As per the record, the campus had rich biodiversity and it has enviable list of rare tree species Medicinal plants and shrubs It is a matter of pride that 40 acres of land is practiced as Agriculture and horticulture fruit yielding crops, which are a home to rare species of plants insects butterflies and snakes. As per the UAS research compendium, 1998 approximately 20-30 plants 86 bird species 10 types of butterflies and few species of reptiles are unique to the area. Meliaduba plantation was established during 2017 for fodder purpose, animals were allowed for open grazing system. Animal, sheep rearing is common and strictly used for experimental purpose. The city is positioned at 12.97°N; 77.56°E and covers an area of 2190 sq km with mean temperature in summer 30.16°C; winter 14-20°C. A landlocked Metropolitan city located in the heart of the Mysore Plateau at an average elevation of 920 meters. Vegetation in the city is primarily in the form of large deciduous canopy with mean sea level 839-962 meters. There are gentle slopes and valleys on either side of this ridge. The low-lying area is marked by a series of water tanks varying size from a small pond to those of considerable extent but all fairly shallow.
Study design
An observational purposive study was conducted at KVAFSU (B) regional campus, Hebbal, Bengaluru to formulate a floristic diversity and species richness in small area. The sample frame was made in accordance with standard operational research. Scientifically, the sample measuring was done in a 20 x 20 plot that was made and recorded as an individual species that are present in the demarcated boundary. The cluster sampling method was used to know the species composition and richness. Analytically, the species diversity index was determined based on the total composition (geographically distributed). Each individual species composition was enumerated separately and squared deviations were obtained from observed species and were simulated from the total number of species presented in the selected plots (derived from the total species composition). The variance component σ2 (i=1,2.. ith species) was derived with weighted score (normalcy have been assumed for determination of variance component i.e. all observations are normally distributed with mean (μ=n) and common variance σ2 independent and identical in nature (i.i.d).
The species new diversity index was mathematically formulated by the following eqn.
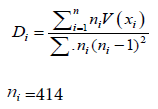 (1.1)
(1.1)
 (23.78% associated with species
richness)
(23.78% associated with species
richness)
D1, D2, D3…..Dk Squared deviation taken from the mean value of species index component
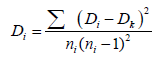 (1.2)
(1.2)
Biotic and A - biotic intervention of species regeneration in selected sites
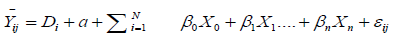 (1.3)
(1.3)
Yij =Observed impact of ith species at jth selected sites
Di =Diversity index at ith species
βo, β1,....βn Coefficients or slope of the independent random variables Xi s
X0=age of the tree
X1=Circumference of the tree
X1=Number of years of human intervention
Di=(Model index) + error Xij
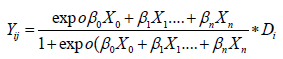 (1.4)
(1.4)
The distance of individual species score was calculated from
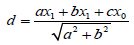 (1.5)
(1.5)
Weighted score of w0, w1, w2.wn is determined is as follows
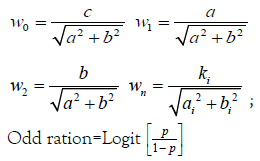 (1.6)
(1.6)
However, the policy frame we estimated in the range of optimal growth of tree species were modelled
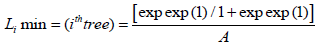 (1.7)
(1.7)
Where I=index of growth development measures of tree and overall environment; A=area so any tree could be permissible if Li ≥ Ll derived under Institutional biodiversity (small area estimation)
The above model was demonstrated by using primary data recorded from the study area; from an each individual plot area we have estimated the species richness and evenness. A total (13.66%) [odds 0.987, p<0.05;95% CI 10.25-26.38] of tree species covers with geographical area of 201 acres of land in the selected sites of regional campus of Karnataka Veterinary Animal and Fisheries Sciences University, Bengaluru. The richness of species is suboptimal and it was found to be insignificantly different (p>0.05) between the present or existing geographical area.
We assessed the floral species richness at each selected sites, and randomly positioned between the species of 10 non overlapping (20 × 20 m) quadratic plots (depending on site size). Within each plot we identified the colloquial names of all tree species, shrubs and ground layer plants with the help of experts and documented the frequency of occurrence of all plants rooted within each quadrate. Additionally we estimated canopy cover by taking a photograph of the quadrat canopy form a vertical height of 1.5-2.0 m above the ground with a camera held by enumerator (8 pixels). Collected data was analysed by R -studio statistical software. As per the analysis among ten quadratic sample across two three sites While there was found to be some variation between sites in terms of the number of new species regeneration [Marginal error 2.54], there was no significant difference between the interaction of ‘μ Girth−μ Cluster μ Girth−μ Cluster’. We identified, there is a significant difference between the richness and cluster, each cluster had some significant variation that was roused and uniformly distributed in different kind of species distribution with mean age of 10.52 years [IQR 3.55-25.88 years] (Figure 1 and Table 1).
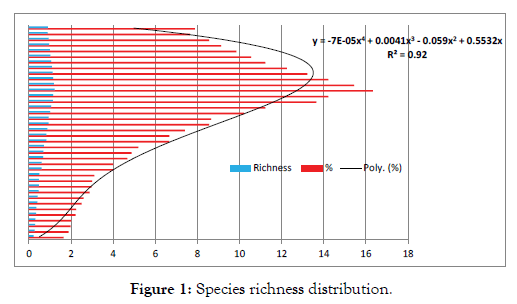
Figure 1: Species richness distribution.
| Species name | Species composition (%) |
|---|---|
| Artocarpus heterophyllus (Jack fruit) | 1.65 |
| Dubergia sissoo (Indian Rose wood) | 1.88 |
| Shorea Robusta (Sal tree) | 1.98 |
| Azardirachta indica (Neem) | 2.01 |
| Ficus religiosa (Sacred fig) | 2.22 |
| Syzygium cumini (Malbar Plum) | 2.24 |
| Ficus Benghalensis (Bangars) | 2.58 |
| Mangifera indica | 2.61 |
| Phyllanthus emblica (Indian Gooseberry) | 2.88 |
| Aeglemarmelos (Golden Apple) | 2.98 |
| Buteamanosperma (Muduka) | 3.00 |
| Bhunea perpuria L | 3.11 |
| Cassia fistula (Kakke) | 4.00 |
| Terminalia arjuntia | 4.00 |
| Lagerstroemia lanceolata | 4.68 |
| Vachellina nilotica | 4.88 |
| Saraca asoca (Asoka tree) | 5.21 |
| Pongamia Pinnata (honge) | 6.66 |
| Milletia pinnata | 6.67 |
| Bombax ceiba | 7.41 |
| Madhuca longifolia | 8.55 |
| Melia Azedaracta | 8.65 |
| Terminalia Billerica | 10.22 |
| Moringa Oleifera (Nugge Fodder) | 11.22 |
| Ficus  recemosa (Fodder purpose) | 13.65 |
| Albizia lebback (Bage mara) | 14.22 |
| Santalum album( Sandal wood) | 16.33 |
| Bauhinia variegata (Orchid tree) | 15.44 |
| Mimusops elangi | 14.22 |
| Switenia mahagani | 13.22 |
| Tamarinds indicia (Hunase) | 12.25 |
| Polyalthia lognifolia | 11.22 |
| Terminalia catappa (Almond) | 10.55 |
| Melia duba (Fodder purpose) | 9.85 |
| Toona celiata | 9.12 |
| Semecarpous anacardium | 8.55 |
| Lannea coromandelica | 7.65 |
| Bamboosa bamboo | 7.88 |
| Mean ± SD | 7.24 ± 4.52 |
Table 1: Notable tree species composition at Regional campus of KVAFSU (B).
The floristic diversity was measured by two main components viz species richness and (the number of species present) species evenness that is measured on how relatively abundant each of the species are. A basic computation was done during the study period for counting number of species enlisted in each selected sites. Total number of species known as species richness, apser the resulted findings the (13.78%) richness was significantly differ in regional campus Hebbal, Bengaluru diversity index eqn (1.1) P<0.05 as compared with small area estimation taking in to account to extrapolate the number of individuals as well as number of species varies from (0) for communities with only a single species to one (1) for sites with many species each few individuals. The change in species richness with spatial scale can be shown in Table 2 off which 10 plots the mean species composition (11.32 ± 2.11) per plots, where species richness is found to be statistically significant in selected Cluster [F=86.13 P=0.000] and also interaction between the richness and cluster was found to be statistically significant [F=9.63 P=0.0001] (Table 3). The results were interestingly defined on how species richness varies across selected sites, it can be more informative than comparison at a single sites. A single measurement in smaller area is more informative than comparison at multiple sites or larger area. The selected site of KVAFSU measurement of species richness deleteriously found to be insignificant at every point of measurement driven by the model eqn (1.2). we have noticed that all selected sites significantly covered Lantana camera and weeds and lower level of species composition it is relatively more adversely affected for restoring the ecological balance with respect to animal and plant interactions This is because the processes that determines richness is likely to change with geographical locations For example the slope may be influenced by increase in habitat diversity resulting from a large area (Figure 1). Moreover, sampling effect due to a larger area having more individuals and thus some clusters had greater proportion of the species pool is likely to depress diversity at very small area. The species individual increment changes were noticed, which assess species diversity by comparing rates of new species addition. The composition of the regional species pool depends on various factors such as rates of evolution and various environmental constraints (pollution, uprooting of plants for construction of building, grazing and agricultural practices). One of the biggest threats to the Institutional biodiversity is farming and leasing of land to other purposes (park and other avenue services). The Institutional biodiversity is in steep decline. The numbers of plant species and mammals, birds and insects have fallen an average of (35%) from 1998-2018 (UAS research compendium). Human are destroying nature at a rate never seen before, and the research findings shows uprooting of rare medicinal plant species from local community. Dispersal limitation may prevent some species in the regional pool from being in local species pool (derived from the during study intervention). For example if the local and University protected area is exploited by human or an isolated area, species in the regional pool in KVAFSU may be missing simply because they cannot get there. Dispersal limitation along with various biotic and abiotic inclusion with human intervention can affect the difference in species composition between the local and actual species pools and thus the potential species richness should be addressed by the competent authority example strong inter specific competition may serve as a varying parameters greatly influenced on the ecological succession in small area, because fragmentation of species, colonization of the species and non-uniformity of the species etc. (Table 4). One of the important constraints with assessing the species composition and pool concept to understand small scale species richness is that it can be a very difficult practice to determine what species should be included in the local, regional and actual species pools For example, it can be difficult to determine what proportion of the species in a region could potentially persist in any given sites. For the actual species pool it can be difficult and arbitrary in practice to assign the edge of a community, especially for selected sites (small area estimation) where communities gradually change across some high degree gradient and climatic condition elsewhere. As we noticed, the plant community has been desecrated by the human intervention (Tables 5-7).
| Cluster | Species Richness (%) | Girth (meter) | Age (Years) | No. of Species Regeneration | Human intervention | PP | 95% Confidence Interval |
|---|---|---|---|---|---|---|---|
| 1 | 23.62 | 2.38 | 9.0 | 25 | 1 | 0.7205 | (0.3393,0.9283) |
| 12.64 | 5.55 | 8.0 | 36 | 0 | 0.2565 | (0.0462,0.7109) | |
| 8.32 | 6.30 | 10 | 22 | 1 | 0.8760 | (0.5665,0.9745) | |
| 10.41 | 5.20 | 12 | 21 | 1 | 0.9038 | (0.6358,0.9806) | |
| 2 | 13.52 | 3.56 | 15 | 24 | 1 | 0.8554 | (0.6213,0.9552) |
| 16.61 | 2.80 | 9 | 27 | 1 | 0.6072 | (0.3212,0.8347) | |
| 13.96 | 2.40 | 8 | 28 | 0 | 0.4940 | (0.1997,0.7926) | |
| 41.85 | 2.60 | 7 | 25 | 1 | 0.7785 | (0.1037,0.9907) | |
| 3 | 9.96 | 3.69 | 4 | 26 | 1 | 0.5091 | (0.1601,0.8494) |
| 6.20 | 3.50 | 6 | 23 | 1 | 0.6732 | (0.2496,0.9273) | |
| 9.29 | 3.40 | 9 | 24 | 1 | 0.7187 | (0.4167,0.9014) | |
| 12.20 | 5.63 | 8 | 27 | 0 | 0.6723 | (0.4400,0.8427) | |
| 4 | 15.29 | 4.80 | 7. | 28 | 1 | 0.5837 | (0.3166,0.8093) |
| 11.32 | 5.60 | 6 | 26 | 0 | 0.6583 | (0.3491,0.8737) | |
| 4.88 | 12.02 | 9 | 23 | 1 | 0.9307 | (0.4550,0.9954) | |
| 6.88 | 13.22 | 8 | 24 | 1 | 0.9283 | (0.4103,0.9959) | |
| 5 | 6.32 | 14.74 | 18 | 28 | 1 | 0.9604 | (0.5201,0.9982) |
| 14.85 | 12.32 | 5 | 35 | 1 | 0.5228 | (0.1144,0.9028) | |
| 3.88 | 10.58 | 6 | 36 | 0 | 0.3516 | (0.0719,0.7915) | |
| 6.32 | 14.63 | 9 | 34 | 0 | 0.7194 | (0.2082,0.9615) | |
| 6 | 3.73 | 3.69 | 8 | 32 | 0 | 0.2975 | (0.0564,0.7499) |
| 5.88 | 5.63 | 17 | 31 | 1 | 0.6887 | (0.3263,0.9099) | |
| 6.44 | 6.33 | 15 | 32 | 1 | 0.6246 | (0.3026,0.8645 | |
| 7.93 | 5.84 | 16 | 28 | 1 | 0.7969 | (0.5439,0.9281) | |
| 7 | 13.96 | 7.42 | 13 | 29 | 1 | 0.7821 | (0.5547,0.9119) |
| 12.20 | 6.32 | 12 | 26 | 0 | 0.8184 | (0.6274,0.9235) | |
| 2.76 | 6.33 | 17 | 32 | 1 | 0.6545 | (0.2589,0.9113) | |
| 12.88 | 8.52 | 16 | 34 | 1 | 0.6918 | (0.2902,0.9249) | |
| 8 | 13.88 | 8.52 | 13 | 34 | 1 | 0.6199 | (0.2719,0.8769) |
| 8.64 | 6.32 | 13 | 22 | 1 | 0.9101 | (0.6511,0.9821) | |
| 5.73 | 3.33 | 14 | 25 | 0 | 0.7699 | (0.4650,0.9279) | |
| 9.69 | 3.48 | 18 | 26 | 1 | 0.8332 | (0.5191,0.9585) | |
| 9 | 3.88 | 3.63 | 15 | 23 | 1 | 0.8485 | (0.5165,0.9671) |
| 12.78 | 3.47 | 16 | 24 | 1 | 0.8648 | (0.6174,0.9621) | |
| 6.88 | 4.78 | 12 | 27 | 1 | 0.7066 | (0.4702,0.8672) | |
| 12.20 | 8.56 | 13 | 26 | 1 | 0.8842 | (0.6347,0.9711) | |
| 10 | 13.08 | 6.32 | 17 | 23 | 1 | 0.9371 | (0.7014,0.9895) |
| 12.64 | 3.33 | 18 | 25 | 1 | 0.8653 | (0.5714,0.9687) | |
| 20.82 | 2.85 | 16 | 28 | 0 | 0.7635 | (0.3606,0.9487) | |
| 18.55 | 3.63 | 18 | 23 | 0 | 0.9222 | (0.6352,0.9878) |
Table 2: Species richness and correlation between other parameters.
| Source | df | Sum of Squares | Mean Square | F | p-value |
|---|---|---|---|---|---|
| Cluster | 2 | 2796.2689 | 1398.1345 | 86.1361 | 0.000 |
| Richness | 1 | 270.0245 | 270.0245 | 16.6356 | 0.0001 |
| Richness*Cluster | 2 | 314.0107 | 157.0053 | 9.6728 | 0.0001 |
| Error | 234 | 3798.2176 | 16.2317 | ||
| Total | 239 | 7178.5217 |
Table 3: Significance of species richness as compared with cluster.
| Label | Comparison | Absolute Value of Sample Mean Difference | Margin of Error | 95% Tukey Interval | Difference |
|---|---|---|---|---|---|
| 1 | μ Richness-μ Clusterμ Richness-μ Cluster | 5.8217 | 2.5454 | (3.2763,8.3672) | Significant |
| 2 | μ Girth-μ Clusterμ Girth-μ Cluster | 0.5805 | 2.5454 | (-1.9649,3.1259) | Not Significant |
| 3 | μ Girth-μ Richnessμ Girth-μ Richness | 5.2412 | 2.5454 | (2.6958,7.7867) | Significant |
Table 4: Comparison of species richness and girth (Circumference of the tree).
| X | Sample mean No of species distributed | Human Intervention | Probability of impact | CI-% | -2 Log Likelihood |
|---|---|---|---|---|---|
| Cluster 1 | 13 | 0 | 0.060 | [0.025-0.647] | 13.46 |
| Cluster 2 | 15 | 0 | 0.219 | [0.038,0.671] | 12.76 |
| Cluster 3 | 13 | 0 | 0.060 | [0.002,0.647] | 11.70 |
| Cluster 4 | 14 | 0 | 0.118 | [0.009,0.647] | 10.68 |
| Cluster 5 | 15 | 1 | 0.219 | [0.036,0.673] | 9.96 |
| Cluster 6 | 16 | 0 | 0.369 | [0.102,0.749] | 9.57 |
| Cluster 7 | 17 | 1 | 0.550 | [0.184,0.868] | 9.41 |
| Cluster 8 | 18 | 0 | 0.718 | [0.231,0.955] | 9.37 |
| Cluster 9 | 19 | 1 | 0.841 | [0.245,0.988] | 9.37 |
| Cluster 10 | 19 | 1 | 0.841 | [0.245,0.988] | 9.39 |
Table 5: Significance of human intervention impact on species diversity.
| Variables | Co efficient | SE | PP-Value | Odd ratio | P-value | |
|---|---|---|---|---|---|---|
| Constant (Species abundance)-Clusters | -6.92 | 532.12 | 1.00 | - | ||
| Number of species regeneration status | 4.01 | 479.78 | 0.999 | 55.52 | =0.001 | |
| Age of the species (Years) | -2.23 | 625.7 | 0.997 | 0.107 | =0.001 | |
| Girth (cm) | -2.40 | 294.52 | 0.999 | 0.086 | =0.001 | |
| Human intervention | 0.109 | 266.06 | 0.987 | 1.11 | =0.001 | |
| No of species competing for main species | 0.704 | 311.39 | 0.985 | 2.02 | =0.001 | |
Table 6: Correlation between species abundance and other associated traits.
| Clusters | No. of species regeneration Mean ± SE | expected age (Years) Mean ± SE | Girth (m) (meters) Mean ± SE | Human Intervention Mean ± SE (Hours per day) | No. of species competing for main species Mean ± SE | Species abundance Mean ± SE | P-value |
|---|---|---|---|---|---|---|---|
| 1 | 12.22 ± 0.18 | 13.22 ± 0.0 | 4.68 ± 0.98 | 60 ± 2.25 | 19 ± 1.22 | 1 | 1.00 |
| 2 | 5.26 ± 0.22 | 23.25 ± 0.02 | 5.51 ± 0.85 | 65 ± 3.65 | 10 ± 1.36 | 0 | 0.000 |
| 3 | 13.15 ± 0.48 | 15.58 ± 0.00 | 3.69 ± 0.76 | 70 ± 4.44 | 15 ± 1.22 | 1 | 1.000 |
| 4 | 5.18 ± 0.25 | 20.13 ± 0.01 | 4.78 ± 0.55 | 125 ± 4.25 | 13 ± 1.28 | 0 | 0.000 |
| 5 | 4.96 ± 0.18 | 22.46 ± 0.02 | 3.87 ± 0.69 | 136 ± 3.55 | 20 ± 1.32 | 0 | 0.000 |
| 6 | 3.01 ± 0.01 | 33.22 ± 0.00 | 2.85 ± 0.71 | 240 ± 4.85 | 26 ± 1.29 | 0 | 0.000 |
| 7 | 13.02 ± 0.02 | 26.26 ± 0.02 | 2.69 ± 0.58 | 230 ± 4.69 | 21 ± 1.22 | 1 | 1.000 |
| 8 | 18.08 ± 0.56 | 28.15 ± 0.00 | 3.38 ± 0.62 | 185 ± 5.55 | 22 ± 1.27 | 1 | 1.000 |
| 9 | 5.22 ± 0.22 | 29.05 ± 0.01 | 3.47 ± 0.66 | 69.00 ± 6.52 | 26 ± 1.17 | 0 | 0.000 |
| 10 | 6.12 ± 0.23 | 2400 ± 0.00 | 3.47 ± 0.45 | 78.00 ± 6.33 | 23 ± 1.22 | 0 | 0.000 |
Table 7: Relation between anthropogenic factor and species abundance (Mean ± SE).
During the study period we observed that, significant relationship between species richness and that strong association between the important vital parameters like age [r=0.85, p<0.01], girth [r=0.76, p<0.01] biotic and abiotic [r=0.82, p<0.01] factors inclusion with biomass accumulation [r=0.81, p<0.01]. The present research work extrapolates any co axial changes or relationship between species and other vital parameters. The study found that there is a significant difference (p=0.003) between vital parameters and species richness in some of the selected clusters freeze with protected areas. Mixed effect model was used to test the hypothetical results the model clearly determines. The absolute value of interaction of species richness and vital parameters was found to be statistically significant [95% CI Tukey test p<0.01; 95% CI 3.27-8.36 2.69-7.78] with pooled marginal error (2.54) it indicates that, species richness of protected clusters were absolutely shows exponential distribution when compared to without protected areas (p=0.00163) The species richness peaking yield at strong level of association in a smaller area or selected site (KVAFSU Campus) where productivity is very high few species can economically fetch more values and positively correlated with good productivity (e.g. Lagerstroemia lanceolate, Dulbergia sisso, Santalum album and Tectona grandis) Importantly the KVAFSU competent authority established a fruit yielding orchards and Meliadooba plantation for fodder purpose approximately 250 animals and 25 types of birds reptiles and few mammals were leading their life and close approximate to the existing environmental condition. We notice that the mechanism for the strong correlation between productivity and well richness in protected area is directly related to change in the lower level of competition of good fertility gradient. A low levels of fertility compitation is asymmetrical distribution between the species richness and floristic diversity in non-protected area However, at high levels of fertility the larger individuals can overtop and pre-empt light from the smaller ones with the result that competition becomes a symmetric with the larger individual species capturing a disproportionate share of the available resources. All selected clusters facilitation occurs when the presence of one species helps the establishment and growth of other species thus increasing the richness of species For example the presence of a nitrogen fixing plant may allow other plants species to establish at a nitrogen limited sites or clusters Similarly nurse plants or young ones provide otherwise rare conditions such as shades and soil stability in drainage (soil erosion) conditions, which allow other plants to establish and survive Facilitating species may also create habitat that allows other species to persist in the local community on stratified manner. Environmental heterogeneity can also contribute to local species richness by increasing floristic diversity Heterogeneous environment persist broader array of conditions that may be optimal for a broader range of species than more homogeneous environments For example, in some locations soil texture and nutrients can vary considerably over a very small spatial distribution, producing significant deviation over in plant species richness through space. Similarly, topographic complexity creates considerable variation in temperature moisture and other vital parameters across a land scapes (excluding human settlement like staff quarters, Institutional building and unprotected areas), providing a wide variety of conditions for both plants and habitat therefore is an increasing trend in species richness across the selected clusters
Listed of notable endangered species
The bird survey was conducted during the study period (2014- 2020) continuing into the middle or late breeding season i.e. summer migrant had not yet left, and winter migrants had not yet arrived. In this period, many young birds leave the nest and participate in the foraging behaviour, so we considered this period to be good season for observing the birds focusing on guilds of diet and foraging site. Additionally, in the agriculture crop field, fruit orchard, plantation and animal grazing land. During the survey, 3-4 census at each study sites were conducted along a 1 km transect during the evening and morning times the rainy days were avoided The census transect was slowly walked and the appearance (direct observation of a bird) and voice (identified listening) of birds were recorded within 30 m of census line. The cumulative data sets were used for determine diversity. The following rare species was enlisted viz Birds; Forest owlet-05 (11.11%), Great Indian Bustard-02 (4.44%), Siberian Crane-03 (6.66%), Indian Vulture-01 (2.22%), Bald eagle-03 (6.66%), Indian Rover-04 (8.88%), Spotted owl-05 (11.11%), Wood pecker-03 (6.66%), Common myna-10 (22.22%), King fisher-02 (4.44%), Nilgiri pipit-01 (2.22%), Nilgiri flycatcher-02 (2.22%), Nilgiri laughing Thrush-03 (6.66%);Mammals; Indian Palm Squirrel-10 (66.66%), Northern Palm Squirrel-05 (33.33%); Reptiles; Western skink-15 (39.47%), Blue tail mole skink-06 (15.78%), Camelian -03 (7.89%), Common Kraits-02 (5.26%), Indian Cobra-10 (26.31%) Viper-02 (5.26%).
Total 414 tree and herb species were recorded in study site (Veterinary College Campus), of f which 114 (27.53%) were recorded economic valuable tree species, the following tree species were enlisted based on the different usage (Figures 2-4).

Figure 2: Species evenness in selected plot.

Figure 3: Agriculture crop practicing nearby experimental plot.

Figure 4: Spectacular view of study plot.
Fuel purposes
Casearia siamea, Pongamia Pinnata, Anogeisus latifolia, Casurina, Acacia pycnantha.
Fodder trees
Leucaena leucocephala (Subabul), Ficus benghalensis (Banyan), Ficus religious (Peepal tree), Azadirachta indica (Neem), Meliaduba (Hebbevu), Prosopsis juliflora (Jali), Delonix regia, Sesbania grandiflora, Tamarindus indica (Hunse), Caliyandra.
Green leaves manure trees and bio insecticide tree
Pongamia Pinnata, Glyricidia, Casearia siamea, Samanea saman (Malle mara), Commiphora caudate (Kondamavu), Indian tulip tree- Spathodia Campanulata (Hoovarisi).
Fruits yielding trees
Tamarindus indica, Artocarpus integrifolia (Halasu), Mangifera indica (mavu) Syzygium jambulna (Nerale), Limonia acidissima (Byla), Annona squamosa (Custard), Ficus recemosa (Anjur) Syzigium jambos, Anacardium occidentale.
Oil yielding and aromatic tress
Samanea saman (Male mara), Pongamia Pinnata, Jatropa, Azadirachta indica (Neem), Madhuca logifolia (Hippe), Simarouba glauca, Eucalyptus globules, Vitex negundo, Cymbopogon (Lemon grass), Canarium strictum, Garcinia indica.
Medicinal trees and herbs
Azadirachta indica tree, Syzygium cumini, Tinospora cardifolia (Amruthaballi), Terminalia chebula, Acacia catechu, Amla, Andrographis paniculata (Nelaberu), Aegle marmelos (Bilwapatre), Magnolia champaca (Sampige), Rauvolfia serpentina (Sarpaganda), Solanum Nigrum (Ganike), Ocimum sanctum (Tulasi), Amaranthus tricolor, Alternanthera sessilis (Hongone).
Agricultural implement preparation purpose
Bamboosa bamboo, Agave (Kathale), Pongamia Pinnata, Moringa oleifera (nugge), Salix tetrasperma (Niranji), Wrightia tinctoria (Beppale), Alangium savifolium (Ankole), Limonia acidissima (Bilwada).
Log or timber species
Teak, Diospyrousebenum, Acaciacatechu, Matti, Gmelinaasiatica (Shivani), Lagerstroemia indica, Calophylluminophyllum, Meliaduba, Syzigiumcumini (Jamoon), African Tulip, Acacia pycnantha,Mahagani, Ficusbengalensis, Albizziachinesis, Artocarpusintegrifolia, Mangiferaindica, Albizzia lebback.
Wind break protection trees species
Silver oak, Casurina indica, Spathodia Campanulata
Soil erosion protecting trees
Bamboosa bamboo, Agave, Pongamia, Caseariasiamea, Delonix regia
Economic valuable tree species
Santalumalbum, Dalbergiasisso, Dalbergialatifolia, Tectonagrandis (Saguwani), Calophylluminophyllum, Tamrindus indica.
Other tree species
Swetania mahangani, Albizzia amara, Moosimara, Nauniapurpura, Diosyprouse benum, Gmelina asiatica, Hardwickiabinata, Calophyllumino phyllum, Antuvala, Acaciaca techu, Prosopis cineraria (Bhanni), Holoptelia integrifolia, Hippe.
Factor affecting species richness or evenness
The present study evaluated various factors that affect or area affected by species richness and evenness importantly, species richness varies through time as well as t space. Overlong time, the composition of species eagerly determines on speciation and extinction of endangered species from radiation effect and pollution (Figure 5). Short time scale the species richness linked to geography and environmental factors such as temperature environmental gradient resource levels and disturbance of naive species etc One of the important factors is the human intervention and disturbance that greatly affect the species richness as can immigration following disturbance. The species richness increases over time as new species colonize the habitat and the neither remains stable or decline due to processes such as competition Further, in communities subject to repeat disturbances such as heavy rainfall soil erosion, flood accidental fire delineated the species richness, may follow a series pattern of rapid decline were seen due to disturbance then gradual increase for several years followed by another disturbance and subsequent decline. Unfortunately very few studies that measures small scale estimation on species richness at Institutional level.
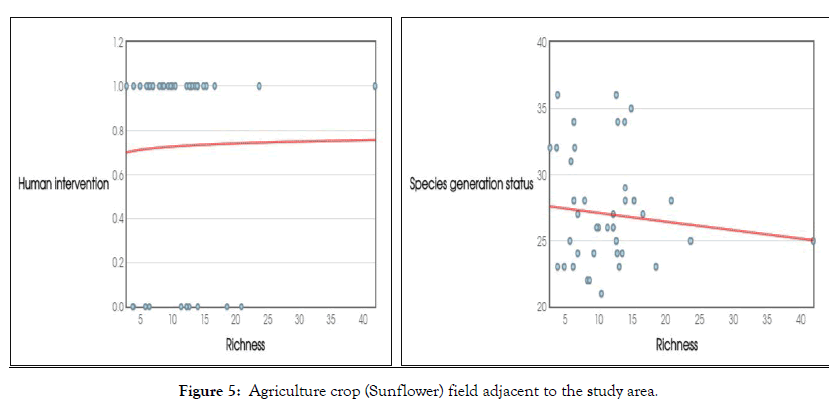
Figure 5: Agriculture crop (Sunflower) field adjacent to the study area.
Further floristic diversity is a measure that combines richness and evenness across species It is often measured because high biodiversity has perceived a synonymous with ecosystem health In general diverse communities are believed to have increased stability increased productivity and resistance to invasion and other disturbances. Human activities that affect biodiversity are here referred to as critical environmental issues the study designed to know the effect of intervention on biodiversity in selected sites. Predictive model was fitted to clarify the intervention of human on the biodiversity. As per the findings human intervention has negatively affected the biodiversity [r=- 0.85, t=- 6.32; p=0.7412] (Figure 6). These issues provide the focus for prioritizing research on the pattern processes and consequences of biodiversity In turn, intimate knowledge of how ecological imbalance changes which includes reduced populations decline biodiversity or extinction that affect biodiversity ultimately provides clearer insight regarding the symptoms of climatic change in the sites caused by human activities The biodiversity change is caused by a wide range of human induced factor called as driver A driver is any natural human induced factor that directly causes a change in the ecosystem The most important direct drives affecting biodiversity are habitat change, climate change invasive species overexploitation and pollution of distinct geographical area. In the aggregate and at a global level there are five indirect drivers of change in biodiversity and ecosystem services; demographic economic, socio-political cultural and religious and scientific technological changes Although, biodiversity and ecosystem services experience change due to natural causes current change is dominated by these anthropogenic indirect drivers. In particular, growing consumption of ecosystem services (as well as growing use of fossil fuels) when results from growing population and growing per capita consumption, leads to increased pressure on ecosystems and biodiversity. Global economy activity increased nearly tenfold between [1950-2020]. Many processes of globalization have amplified some driving force changes in ecosystem services and attenuated other forces over the post-independence there have been significant changes in socio-political drivers including a declining trend in centralized authoritarian governments and a rise in elected democracies which allow for a new forms of policy in particular adaptive management of environmental resources such as development of infrastructure, alteration of climatic resources through construction of Roads and escalation etc. Another most important direct driver of biodiversity is loss and change in ecosystem services are habitat change such as land use change for agriculture crops and physical modification etc.
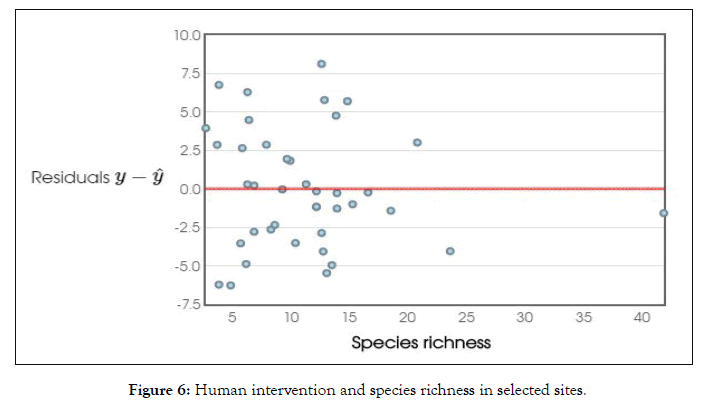
Figure 6: Human intervention and species richness in selected sites.
Significant merits of floristic diversity in Regional Campus of KVAFSU (B)
The study of floristic diversity has attained more importance or merits as the world biodiversity in its status has threatened in its existence. Plant diversity may be considered as the backbone of biodiversity Plant diversity or species composition is important from medicinal economical ecological and environmental point of view and also nurtures the scientific information pertaining to climatic changes in the selected geographical location. The intuition of diversity greatly helps the diversified habitat with variety of plants that can have benefits such as providing forage for a variety of insects and vertebrate species stability resulting from plants in the community that are able to survive drought insect plagues, and or disease outbreaks so that the site will have some soil protection forage etc in those years Since plant contacting a variety of genetic material that may be useful in long term survival and stability of the community where as community will be more benefits from a mixture of plants However plant containing a variety of genetic material that may be useful in long term survival and stability of the community where as community benefits from a mixture of all habitat and healthy diverse plant communities generally have all niches filled and are theoretically less likely to be invaded by noxious or opportunistic introduced species In another words very few demerits listed to high density or species richness (Figure 7). The diverse communities are often a sign of fragmented or somewhat degraded sites where much of the species richness is contributed by disturbance species. Since plant communities with high diversity can be more difficult to manage for grazing because different species of plants have different grazing tolerances and different rates of phenological development. Many plant communities are very stable with few species that are well adapted to the environment in the study area. Furthermore the global warming (temperature gradient) and the loss of biodiversity through human activities (e.g. land use change, pollution invasive species) are two most profound threats to the functional integrity of the institutional ecosystem. These factors are, however most frequently investigated separately ignoring the potential synergistic effects of biodiversity loss and environmental warming on processes, declined trend of biodiversity have more consequences for human well-being As per the research findings (Figures 8-11) human intervention or anthropogenic factors greatly affect the biodiversity and found to be statistically significant (35.0%) [CI -95% 12.55-41.22; Odds 5.22 p=0.0022] followed by pollution (25%) [95% CI 10.68-28.79; Odd 6.31 p=0.0022]; drastic climatic change (20%) [95% CI 8.63-25.18; odds 3.18 p=0.0016]; habitat degradation (8.0%) [95% CI: 3.16-10.74 p=0.0008]; Odds 4.86; invasive species (10%) [95% CI: 6.32-12.55; Odds 5.02 p=0.0012] disease (1.0%) [95% CI: 0.32-1.5% Odd 0.96 p=0.4123].

Figure 7: Brooding of common babbler (Argya caudate) Bird species (Local name Gubbachi).
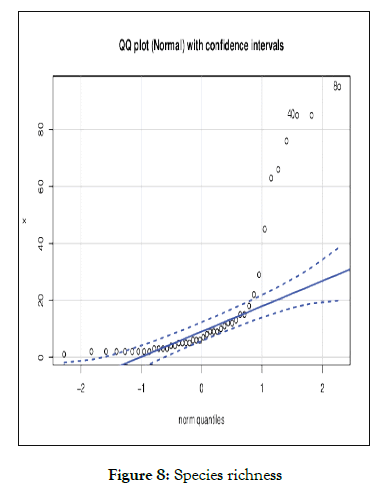
Figure 8: Species richness
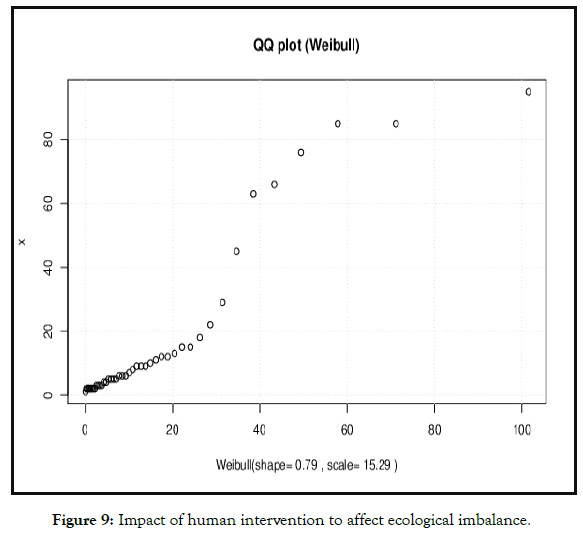
Figure 9: Impact of human intervention to affect ecological imbalance.
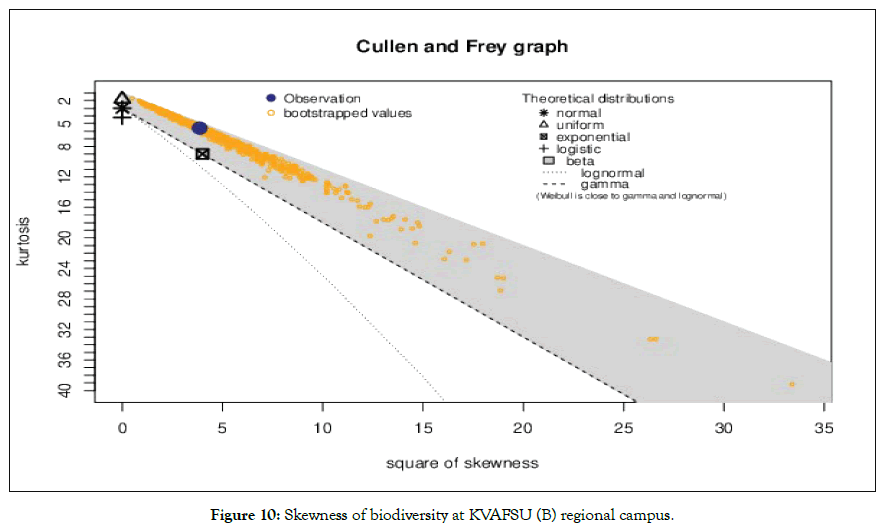
Figure 10: Skewness of biodiversity at KVAFSU (B) regional campus.
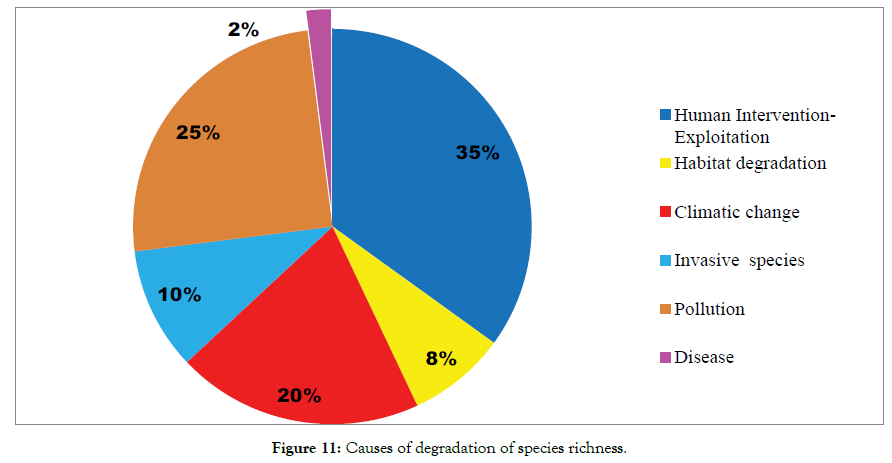
Figure 11: Causes of degradation of species richness.
Biodiversity is fundamental to human life on earth, and the evidence unequivocal it is being destroyed by us at a rate unprecedented in history of pre-Independence of India said the many literature published in reputed journals. Since the industrialization and urbanization human activities have increasingly destroyed and degraded forests grasslands wetlands and other important ecosystem threatening human well-being. As per the Kasturirangan Report, 2016 about (70-80%) earth’s free land surfaces has already been significantly altered, most of the protected and unprotected forestland are deforested and water has contaminated by poisonous substances than (80%) of the area of wetlands has been lost [22]. The living planet Index is one of the most comprehensive measures of global biodiversity [4]. An average decline of (63%) after post-independence The alarming situation was seen in India As per the India’s ecological footprint the figure is lower than 1.6 global hectares per person, which is the lowest bracket and is the smaller than that of many large countries. However, its high population levels make it likely for country face a widening ecological deficit even if current per capita levels of resources consumption remain the same. India is a highly bio diverse country, holding over 45000 species of plants in only (2.4%) of the world’s land area Over (12%) of the wild mammal species are threatened with extinction in the country. Larger animals, higher level plant species, particularly fresh water habitats, are in greater danger extinction [5]. These include river dolphins found in India Giant catfish in the Mekong otters and beavers among others. In India, (3%) of bird species face extinction (e.g. Vulture hornbill) with the number increasing every year As many as (19%) of amphibians are threatened or critically endangered. Bee colonies are also collapsing drastically across the country. In practical point of view Institutional Biodiversity is a foundation of entire ecosystem and services to which human wellbeing is intimately linked. No more feature of earth is more complex dynamic and varied than the layer of organisms that occupy its surfaces and its physical covers, and no feature is experiencing more dramatic change at the hands of humans than this extraordinary, singularly unique feature of earth. This layer of living organism the biosphere through the collective metabolic activities of its innumerable plants mammal’s reptiles and birds physically and chemically unites the atmosphere, geosphere and hydrosphere in to one environmental system within which millions of species including humans have thrived [7]. In case of small area biodiversity, it follows that large scale human intervention over this have tremendous impacts on ecological succession and biodiversity it also follows that the nature of these impacts good or bad is within the power of humans to influence. Further Biodiversity plays an important role in ecosystem function that provides supporting provisioning regulating and cultural services [14]. These services are essential for human wellbeing, since at present there are few studies that link changes in biodiversity with changes in ecosystem function to changes in biodiversity system [16]. A large proportion of small area biodiversity at the species level is well concentrated in rest of the system [23,24]. Mostly in the protected and reserved area the species richness and evenness is heavily concentrated (13.12%) similar study was reported [20]. The challenge of biodiversity upscaling, estimating the species richness of a large area from scattered local surveys within it, has attracted increasing interest in recent years, producing a wide range of competing approaches. Such methods, if successful, could have important applications to multi-scale biodiversity estimation [25-28]. Applied small area estimation model, the methods should greatly facilitate biodiversity estimation in poorly studied taxa and regions, and the monitoring of biodiversity change at multiple spatial scales [29-31]. Institutional and urban green area faces many threats worldwide [32]. Without robust methods for estimating the consequences for biodiversity planning decision run the risk of being uninformed or misinformed [4]. The grounds redevelopment programs provides an opportunity for systematic ecological surreys to quantify the effects of habitat creation, expansion reduction and disturbance in the future, adding useful knowledge about this culturally important urban green space at the same time as allowing improvement of biodiversity models to support planning decisions [33], over half of the world’s population lives in cities and for many urban green spaces are the only places where they encountered biodiversity [17]. This is a particular concern because there is growing evidence that human well-being is enhanced by exposure to nature However, the specific qualities of green spaces that offer the greatest benefits remain poorly undersood. The lack of knowledge of a consistent relationship between actual plant species and ecosystem. Nicholas et al. [33] species richness is a fundamental measurement of community and regional diversity, and it underlines many ecological models and conservation strategies [34]. In spite of its importance, ecologists have not always appreciated the effects of abundance and sampling effort on richness measures and comparison. These pitfalls can be largely avoided by using accumulation and rarefaction curves, which may be based on either individuals or sampled level. In the present study we have recorded more than 2/3rd of all species that ranges less than 2 km (small ecosystem functioning. In this propinquity, we recorded mean annual temperature of study area (data extracted from Meteorology unit Main Research Station UAS Bengaluru). We found that changes in temperature systematically altered the relationship between biodiversity and Institutional ecosystem As temperatures departed from ambient conditions, the exponent of the diversity functioning relationship increased meaning that more species were required to maintain ecosystem functioning under thermal stress In our study area the mean temperature was recorded in summer season was 33.56 ± 0.93°C (IQR 29.12-36.122); winter season (25.36 ± 0.12°C). We have noticed that, during summer season the temperature was strongly associated with destruction of certain kinds of tree species (r=0.83; odds 4.74 likelihood ratio was 23.17 p=0.0017).
Species richness and conservation and managing for ecosystem services present conservation challenges for planning and national policy [1]. Although university campus is a centre for production of new young talented buds who are serving nation in different fields they present a considerable opportunity in forwarding global sustainability and environmental goals For example universities are the forefront in planning climatic change adaptation and mitigation of knowledge hub for conservation of habitat and ecosystem besides with campus also revealing the potential for managing and large scale environmental changes over decadal relative comparison richness and species evenness was found to be statistically significant and moved to exponential growth (Figure 8). However, growing population and human intervention relatively (20%) habitat loss was seen with respect of human intervention (Figure 9). The predictive loss of biodiversity was determined by Cullen and Frey graph (Figures 10 and 11), the results substantially found to be (15-35%) estimated loss by 2025.
Nationally, academic Institution and university are rapidly increasing in area and in population Institutional area is projected to triple by end of 2036 [1]. Most urbanization is occurring in regions identified as biodiversity hotspots with profound effects on ecological patterns and processes including habitat destruction degradation and fragmentation changes to biological assemblages resulting in novel ecological communities increased level of pollution in soils air and water system and alteration of natural disturbance regimes and ecosystem processes, such as water and nutrient cycling (Figures 12 and 13). As a result, the density of flora and fauna is substantially reduced at Institutional level (small area) compared to that of a other unprotected area habitat [19-21]. As per the driven results land conservation for agriculture has caused (60%) lease of Institutional land for others (30%) and land utilized for recreation purpose (10%) of entire biodiversity loss and half of all tree cover loss from ground level and of the total predictable loss (60-70%) withdrawn from available resources. However, reduction in biodiversity decreases the capacity of ecosystem to capture essential resources produce biomass, and maintain ecological area). Various approaches were practiced in association with cultural beliefs and faith. Human intervention or anthropogenic factors is one of the key indicators that affect the existing ecosystem and ecological succession and species retrogression [35]. In contrast of this intervention local and regional species evenness would be drastically declined and tends to move negative skewness when compared to protected area. The regional diversity of species richness and interaction of the animals found to be significant differ for nesting their lives in open atmosphere because of their adaptation [36]. Direct loss of biodiversity loss includes use change practice of Agriculture system habitat loss overexploitation human intervention pollution and paradoxical changes of climatic condition found to be deleterious factor for reducing species evenness and richness in small area The continued decline of diversity in selected sites including loss or degradation of ecosystem is reducing the ability of biodiversity and ecosystem to provide essential life sustaining services and in many cases lead to negative outcome for health and wellbeing. Ecosystem degradation may lead to both biodiversity loss and increased risk from infectious diseases. In turn the indirect drivers of biodiversity loss are demographic changes and large scale social and economic processes. Social change and development trends (such as urbanization, encroachment of land for the infrastructure purpose) poverty and dishonesty of the individuals also influence these drivers of change New policies and structures at national level for small area that provide perverse incentives or fail to incorporate the values of biodiversity often compound the dual threat to existing biodiversity and human wellbeing. The expansion of agriculture infrastructure will cause habitat loss in the study area and lead to a continuing decline in the local and urban biodiversity The habitat loss between 1965 (UAS, Bangalore research compendium report, 1967)and till date, will lead to the extinction of approximately 20-30% of the species in the long term (depending on the existing scenario). Losses of habitat and plant populations will be fastest in warm mixed due to human intervention or anthropogenic factors admixture with expansion of habitat area for experimental and infrastructure purposes. More productive approaches to the environment will have more success in slowing forest habitat and biodiversity loss in the near future than reactive approaches The scenario most concerned with security protection has the highest rate of biodiversity loss. In pragmatic approach developing countries will experience an expansion of their agricultural lands and a reduction of their species richness [6]. Although the reverse is expected to happen in Industrial countries the result will be net loss of forest. Overall, biodiversity loss in the small area will be driven by land use change to greater extent than by climate change and excessive nutrient levels but the impact of these drivers will be different in different ecological niche. Other factors such as overharvesting of plant species invasive species, weeds and pollution will also speed up substantial loss of biodiversity in threat area (study area).Since biodiversity loss will lead to a deterioration of the benefits that humans obtain from our ecosystem It will increase the likelihood of ecological surprises such as rapid climatic change, pandemic disease outbreak drought low rainfall and some accidental causes (fire) [35]. The Vulnerability of human well-being to these adverse surprises is different in each scenario but is greatest order from strength Such changes will affect human well-being directly as well as indirectly for instance, because of conflicts due to scarcity of food and basic necessities [12]. The scenario indicates that many environmental and developmental long-term goals are independent [27]. Therefore, private partnership and Forest Department agreements are important and vary greatly from one climax to another. Major decision in the next 50 years will have to seriously address the trade-off issues between health indicators with respect to medicinal plants to mitigate the basic necessities for common ailments and also use of non-renewable resources [26]. For a given level of socioeconomic development policies that conserve more biodiversity will also promote higher aggregated human wellbeing through the preservation of regulating our own culture and beliefs (ancestral period). In this prospective of large unexpected shifts in ecosystem services can be addressed by adopting new forest policies that hedge for the development of biodiversity that is geographically distributed in urban small area and also adjusting the flexibility as advanced knowledge becomes available for a routed scientific temperament of altering our existing ecosystem. Study reported by Joshi [37], he studied floristics diversity in Agriculture College and Main Research Station Hebbal Campus, Bengaluru. As per the study species richness was 62.16% (small area estimation) with extensive good habitat Vegetation covers are uniformly distributed throughout the campus without any fragmentation and also he documented 1200 tree species with 50 families and 20 genera. Our study is comparable to above study, the species richness was just (13.78%) and shows insignificant vegetation covers with respect to existing geographical area such huge differences was due to anthropogenic effect, pollution fragmentation of land for human settlement and bonafide of government land to others Future work using device Li for different tree under different climatic conditions especially temperatures in several states of India may be explored, analysed and inferred [38-65].
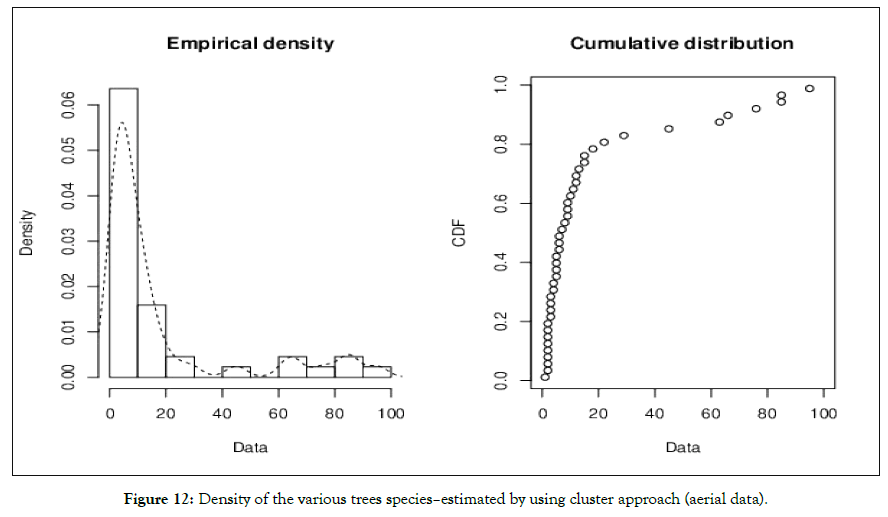
Figure 12: Density of the various trees species–estimated by using cluster approach (aerial data).
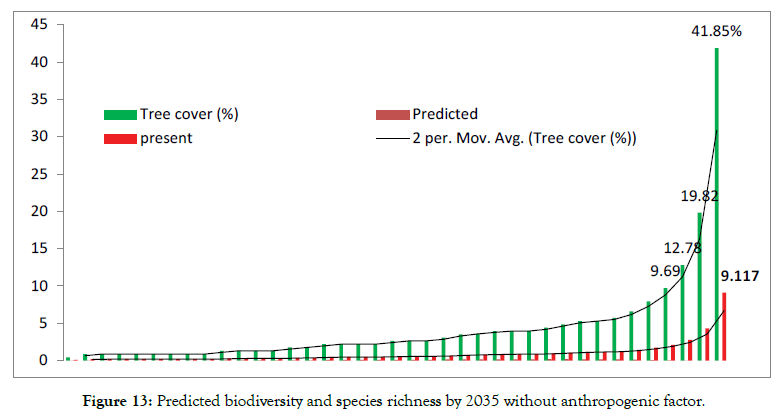
Figure 13: Predicted biodiversity and species richness by 2035 without anthropogenic factor.
An expansion of infrastructure and anthropogenic factor affects the disappearance of large fraction of plant and animal population in the research area Policy implication would be done in association with forest department and NGO, greatest concern is necessary for protection of biodiversity at KVAFSU (B) regional campus. On the basis of experimental approach, the biodiversity must be integrated in to private public partner. These sectors are directly dependent on biodiversity and positive association with productivity and sustainability of native plant species and also it can make significant contribution for restoring ecological balance at the larger extent.
• Growing demographic transition and urbanization less biodiversity would exist today (suboptimal in small area) had no communities University authority NGO government policy makers and to a growing extent positive action taken to conserve biodiversity, mitigate its loss and support its sustainable use.
• Species protection and recovery measures for threatened species -Considerable scope exists to conserve and use biodiversity sustainably through more effective management of individual species. Although “habitat based “approaches to species conservation are critical.
• Incept public awareness programme for public – Education and communication programs have both informed and changed preferences for biodiversity conservation and improved implementation of biodiversity responses improved communication through organization of mela conference and workshop for public are essential to achieve salient objectives of environmental conventions and its sustainable development.
• Prevent habitat loss from anthropogenic factors (human intervention) and strictly adhere the National environmental policy.
• Utilize existing green space connection.
• Use a native plant palette and plant appropriately.
• Support green economy and sustainable life style.
• Maintain spontaneous and novel urban habitats with associated species assemblages.
Authors acknowledge to the Hon’ble Vice Chancellor Prof. H.D. Narayanaswamy, Prof. H.M. Jayaprakasha, Campus Head, KVAFSU (B), Hebbal, Bengaluru and all key officers of Karnataka Veterinary Animals and Fisheries Sciences University, Bidar. We owing to extend our Special thanks to Prof K.N. Ganeshaiah Distinguished Former Professor and Botanist, UAS Bengaluru for his valuable inputs during study period, Personally we acknowledge Prof. Vivian K. Johannsen for his critical review and recommendation for small area estimation mathematical formula Further, we extend our heartfelt thanks to Prof. Dr. (Mrs) Bharathi Former Dean DSC and Prof. H.N. Narasimhamurthy. This research paper specially dedicated in the memory of our beloved teacher Prof. C.S. Patil Department of Silviculture College of Forestry Ponnampet.
Citation: Basavarajaiah DM, Narasimhamurthy B, Jayanaiak P, Gouri MD (2020) Biodiversity and Species Richness in Karnataka Veterinary Animal and Fisheries Sciences University Regional Campus. Fores Res. 9:240.
Received: 24-Sep-2020 Accepted: 08-Oct-2020 Published: 15-Oct-2020 , DOI: 10.35248/2168-9776.21.9.240
Copyright: © Basavarajaiah DM, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.