Indexed In
- Open J Gate
- Genamics JournalSeek
- CiteFactor
- Cosmos IF
- Scimago
- Ulrich's Periodicals Directory
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- Directory of Abstract Indexing for Journals
- OCLC- WorldCat
- Proquest Summons
- Scholarsteer
- ROAD
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Review Article - (2019) Volume 11, Issue 3
The Effects of Sodium-Glucose Co-Transporter-2 Inhibitors (SGLT-2-i) on Fatty Liver, Steato-Hepatitis, Liver Fibrosis, and Hepatocellular Carcinoma-A Review
Hyder Osman Mirghani, Thomas Antony Thaniyath* and Fakhralddin Abbas ElfakkiReceived: 05-Mar-2019 Published: 01-Apr-2019, DOI: 10.35248/0974-8369.19.11.459
Abstract
The drug category of Sodium-Glucose co-transporters 2 inhibitors (SGLT 2 inhibitors) is a newer class of pharmaceutical agents for the management of type-2 diabetes mellitus, with a proved promising cardio-protective effects. In addition to above beneficial effects, Sodium-Glucose co-transporters 2 inhibitors showed their effectiveness to reduce abdominal fat content, amount of fat in liver tissues and progression of inflammation and fibrosis in experimental as well as clinical studies. Non-Alcoholic Fatty Liver Diseases (NAFLD) are common among patients with type 2 diabetes patients and includes fatty liver, Non-Alcoholic Steato-Hepatitis (NASH) and cirrhosis and when present together these disorders exacerbates each other deleterious consequences. This review article is carried out with an objectives to find out the effects of Sodium-Glucose co-transporters 2 inhibitors on non-alcoholic fatty liver diseases, steato-hepatitis, fibrosis liver and hepatocellular carcinoma. The authors identified 21 relevant articles including twelve experimental and nine clinical studies with inclusion of 15152 patients, in study period ranges from 20-104 weeks. The analysis of these studies showed beneficial effects SGLT 2 inhibitors on fatty liver, liver function and NASH development, hepatic fibrosis and prevention and regression of hepatocellular carcinoma and concluded that SGLT-2 inhibitors may protect or reduce the incidence of non-alcoholic fatty liver diseases, steato-hepatitis and hepatic fibrosis in type 2 diabetes patients.
Keywords
SGLT-2-i; Non-alcoholic fatty liver; Steato-hepatitis; Hepatic fibrosis
Introduction
The US FDA recently approved Sodium-glucose cotransporter-2 inhibitors (SGLT-2-I) -canagliflozin, dapagliflozin and empagliflozinfor the treatment of patients with type 2 diabetes, who are not reaching the glycaemic control targets [1]. SGLT-2-I suppress renal glucose reabsorption by inhibiting the transporter protein (SGLT-2 protein) present in the S1 segment of proximal renal tubule, which is responsible for the majority of glucose reabsorption from renal tubule and thereby SGLT-2-I enhances the urinary glucose excretion [2,3] and controls the hyperglycaemia.
SGLT-2 inhibition offers several putative advantages. Acting independently of insulin, these agents should not confer a risk of hypoglycaemia and could be employed as monotherapy or in combination with other agents. Given their mechanism of action, these agents should be effective in patients with any degree of insulin resistance or β-cell dysfunction. Due to loss of glucose (calories) in urine and glucose-induced osmotic diuresis [3], the use SGLT-2-I are associated with loss of body weight in type 2 diabetes patients and their mild osmotic diuretic effect could potentially contribute to the reduction of systemic blood pressure [3]. Taken together, all these effects may have a beneficial impact on cardiovascular outcomes [4].
In addition, SGLT-2 Is has been shown their effect to increase glucagon/insulin ratio and shifting the metabolism from glucose to lipid oxidation and furthermore these classes of pharmaceutical agents causes a significant weight loss due to reduction in visceral fat content and body fat mass. Thus it seem to be reasonable that SGLT-2 inhibitors could be more beneficial for patients with concomitant type 2 diabetes and non-alcoholic fatty liver disease [5,6]. SGLT-2 inhibitors were also found effective to reduce the fat content in the liver, improve inflammation, and prevent progression of fibrosis in NAFLD model mice [7].
It is estimated that up to 70% of patients with type 2 diabetes mellitus are affected with non-alcoholic fatty liver disease [7]. The patients suffering from NAFLD may progress to the complications such as Steato-hepatitis, liver cirrhosis and hepatocellular carcinoma. Thus it seem to be reasonable that SGLT-2 inhibitors could be more beneficial for patients with concomitant type 2 diabetes and non-alcoholic fatty liver disease [5,6]. Considering the above information, this article, reviews the relevant studies with objective to determine the effect of SGLT-2 inhibitors in nonalcoholic fatty liver disease and hepatocellular carcinoma.
Materials and Methods
An electronic literature search was carried out in the following databases PubMed, MEDLINE and Embase. The key words used were SGLT-2 inhibitors-canagliflozin, dabagliflozin, empagliflozin, steato hepatitis, non-alcoholic fatty liver disease and hepatocellular carcinoma. The search was limited to articles published in English language and conducted in both humans and other animals, with time limit of 5 years from 2013.
To be included for review, the following criteria were considered: patient or animal model of type 2 diabetes and with altered hepatic functional parameters or hepatic injury administered with SGLT-2 inhibitors. Titles and abstracts were screened independently by two authors and full article retrieved for the manuscripts found relevant for the topic. Additional articles were searched and identified through hand searching of the bibliography. The retrieved full text articles were assessed for eligibility for inclusion and data were extracted by the authors using proforma. Any disagreement in selection of articles and data was discussed and solved between the researchers.
Results
The initial total number of 263 articles is identified. After removing duplications and irrelevant results, 34 articles are selected for further reading of abstract and after revision of the contents and application search criteria, a total of 21 evidences are retrieved and selected for final analysis and review (Figure 1). Out of 21 retrieved articles, twelve studies were experimental studies and nine were clinical studies, which included 15152 patients with type 2 diabetes mellitus and non- alcoholic liver diseases on SGLT-2 is with duration of treatment ranges form 20-104 weeks.
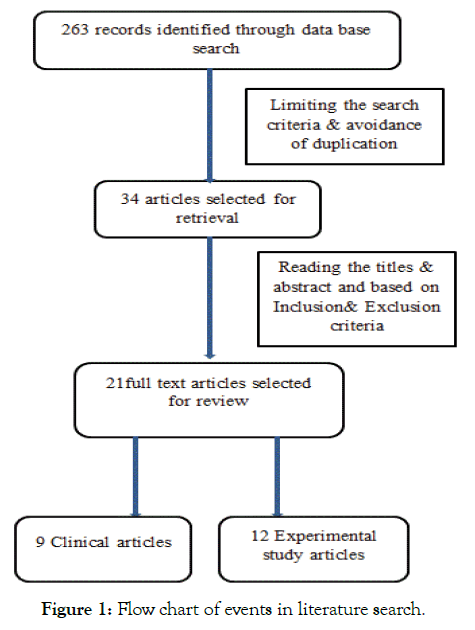
Figure 1: Flow chart of events in literature search.
Discussion
Non-alcoholic steato-hepatitis is a common complication among the patients with type 2 diabetes mellitus with a prevalence of 70% and they usually co-exist as manifestation of the metabolic syndrome, which is marked with high level of insulin resistance among these patients [7,8]. To add to this complication of diabetes mellitus, a meta-analysis report concluded that the patients with diabetes mellitus may be associated with elevated risk of hepatocellular carcinoma incidence and mortality, furthermore, patients with diabetes have poor prognosis compared to their counterparts [9,10]. Sodium glucose co-transporter 2 (SGLT-2) inhibitors lower blood glucose levels by inhibiting the reabsorption of glucose in the kidney and promoting the urinary glucose excretion and used to treat type 2 diabetes mellitus [11].
In addition to anti-hyperglycemic effect, SGLT-2 inhibitors have also been reported to have an effect to reduce abdominal fat (Table 1) [12]. SGLT2 inhibitors were also found effective to reduce the fat content in the liver, improve inflammation and prevent progression of fibrosis in NAFLD model mice [13].
Table 1: Effects of SGLT-2 inhibitors on Non-alcoholic Fatty Liver Disease and hepatocelluar carcinoma, clinical studies.
| Author | Year | Drug | No. of Patients | Study | Duration | Results |
|---|---|---|---|---|---|---|
| Kuchay et al. | 2018 | Empagloflozin | 50 | Trial | 20 weeks | Liver fat and ALT reduction |
| Sattar et al. | 2018 | Empagloflozin | 11042 | EMPA-REG OUTCOME® trial and other five | 24-104 weeks | Liver fat, ALT and AST reduction |
| Kawaguchi et al. | 2018 | Canagliflozin | 1 | Case report | Regression of hepatocellular carcinoma | |
| Seko et al. | 2017 | SGLT-2 | 45 | Case-control | 24 weeks | ALT, AST improved |
| Tobita et al. | 2017 | Dapagliflozin | 16 | A Prospective, Open-label, Uncontrolled Study | 24 weeks | ALT, AST improved |
| Ito et al. | 2017 | Ipragliflozin | 66 | Trial | 24 weeks | ALT, AST and hepatic/spleen ratio reduced |
| Levine et al. | 2016 | Dapagliflozin | 1 | Case-report | Raised ALT & AST | |
| Ohki et al. | 2016 | SGLT-2 & GLP-1 agonist | 130 | Retrospective | ALT and FIB-4 index reduction | |
| Leiter et al. | 2015 | Canagliflozin | 3801 | Data from four placebo-controlled & two active controlled | 26-52 weeks | ALT, AST improved |
Tahara et al. in 2013, studied the effect of ipragliflozin in type 2 diabetic model mice with hepatic steatosis and they observed improvement in hepatic function in animals and evidenced with diminished oxidative stress and inflammatory markers (Table 2) [14]. They examined the effect of ipragliflozin in streptozotocinnicotinamide- induced type 2 diabetic mice with high-fat diet which exhibit impaired insulin secretion, insulin resistance, hyperlipidemia, hepatic-steatosis and obesity.
Table 2: Effects of SGLT-2 inhibitors on Non-alcoholic Fatty Liver Disease and hepatocelluar carcinoma, experimental studies.
| Author | Year | Drug | Model | Result |
|---|---|---|---|---|
| Tahara et al. | 2013 | ipragliflozin | Type 2 diabetic mice | Improved hepatic steatosis |
| Hayashizaki-Someya et al. | 2015 | ipragliflozin | CDAA-diet rats | Hepatic TG accumulation and fibrosis prevention |
| Qiang et al. | 2015 | luseogliflozin | Steatohepatosis mice | Serum alanine aminotransferase, and fibrotic changes were all attenuated |
| Nakano et al. | 2015 | Remogliflozin | Obese male mice | Reduced ALT, AST, and liver weight |
| Komiya et al. | 2016 | ipragliflozin | Obese mice | Ameliorate fat accumulation in the liver, liver dysfunction, and inflammation |
| Honda et al. | 2016 | ipragliflozin | NASH mice | Improved liver injury, and fibrosis |
| Jojima et al. | 2016 | Empagliflozin | NASH mice | Attenuates development of NASH showing anti-steatotic and anti-inflammatory effects |
| JI et al. | 2017 | Canagliflozin | Obese mice | Reduced the liver weight and ameliorated liver fibrosis |
| Tang et al. | 2017 | Dapagliflozin | Type 2 diabetic mice | Decreased liver injury and fibrosis |
| Wang et al. | 2018 | Dapagliflozin | Obese mice | Decreased serum ALT, AST, hepatic lipid accumulation and hepatic fibrosis |
| Shiba et al. | 2018 | canagliflozin | NASH mice | attenuated hepatic steatosis, inhibited the development of hepatic fibrosis, and the number of liver tumors was significantly reduced |
| Kaji et al. | 2018 | Canagliflozin | Human liver cancer cells | Attenuates liver cancer cell growth and angiogenic activity |
Four-week repeated administration of ipragliflozin improved hepatic steatosis and obesity along with improvement in diabetic parameters.
In this experimental study, ipragliflozin also shows its effectiveness to reduce plasma and liver levels of oxidative stress biomarkers (thiobarbituric acid reactive substances and protein carbonyl) and inflammatory markers (interleukin 6, tumor necrosis factor α, monocyte chemotactic protein-1 and c-reactive protein) and improved liver injury as assessed by plasma levels of aminotransferases.
This positive effect of SGLT-2 inhibitor is proved by Qiang et al. in their study on rodent model with liseogliflozin [15] and it prevents accumulation of triglycerides and fibrosis in choline deficient L-amino acid defined diet rats [16].
Qiang et al. used mice treated with both nicotinamide and streptozotocin to induce diabetes and then fed a high fat diet containing trans fatty acids (HFDT) for 8 weeks and divide in to 2 groups. One group is treated with luseogliflozin and other one untreated, during this period. Luseogliflozin treated group showed improvement in fatty liver and reversal of elevated liver enzymes in addition to glycaemic control. Above those, luseogliflozin controlled, fibrotic change and increases in collagen deposition with upregulations of collagen1 and smooth muscle actin and inflammatory cytokine expressions observed in the HFDT-fed mouse livers.
In another experimental study, Nakano et al. in 2015 demonstrated the liver enzyme reducing effect of SGLT-2 inhibitor-remogliflozin in mice. After four weeks treatment with remoglifozin, there were marked reduction in plasma alanine aminotransferase (76%), aspartate aminotransferase (48%) and hepatic triglyceride content (40%). It also reduced the liver weight by 42% [17].
In 2015, Leiter et al. pooled data from four 26-week duration placebo controlled clinical trial, with cangliflozin 100 mg and 300 mg including of 2313 type 2 diabetic patients and from 2 studies of 52 weeks duration active controlled trials with canagliflozin 300 mg versus sitagliptin 100 mg in 1488 patients and found marked reduction in hepatic enzymes with canagliflozin than placebo group in 26 weeks trial and with canagliflozin 300 mg versus sitagliptin 100 mg (nominal p<0.001 for alanine aminotransferase, aspartate aminotransferase, gamma-glutamyl transferase and bilirubin, and p<0.01 for alkaline phosphatase) at week 52 [18].
Another retro-prospective study in 2016 by Ohki et al. enrolled 24 patients with type 2 diabetes mellitus with elevated ALT level administered with a combined treatment of SHLT-2 Inhibitor and glucagon like peptide-1(GLP-1) analogue or Dipeptidyl peptidase 4 (DPP-4) inhibitors. Out of 24 patients, 13 patients were administered a combination of SGLT-2 inhibitors with DPP-4 inhibitors, and 11 patients with combination of SGLT-2 inhibitors with GLP-1 analogues for a median dosing period of 320 days. At the end of the follow-up, body weight (p<0.01) and glycosylated hemoglobin levels (p<0.01) reduced markedly. Serum ALT levels also decreased significantly (p<0.01) and normalized in 14 patients (58.3%) [19]. In controversy to above findings, Levine et al. reported a case in 2016 with the drug dapagliflozin raised the liver enzymes in a type 2 diabetes patient with liver cirrhosis secondary to steatohepatitis and biopsy showed the features of drug induced liver injury [20].
Recently, in another animal study, Tang et al. demonstrated the protective effect of dapagliflozin against renal and liver fibrosis in type 2 diabetes model mice (Table 2) [21]. They conducted an experimental search the dapagliflozin in 18 weeks old db/db mice with objective to determine the hepato-protective effective of SGLT- 2 inhibitors and concluded that dapagliflozin improves the liver fibrosis by suppressing hyperglycaemia induced inflammation and oxidative stress.
The above observations of Tang et al. is supported by the findings of Wang et al. 2018 that the drug reduced the elevated hepatic enzyme in Western Diet induced obesity mice [22]. This protective role of dapagliflozin is also found in two separate clinical studies in patients with NASH and Type 2 DM by reducing the elevated serum liver enzymes in these patients [23,24]. In a recent randomized controlled clinical trial in patients with NAFLD and diabetes, Kuchay et al. observed a significant reduction in liver fat content in the arm of trial drug empagliflozin [25]. In this randomized controlled trial they compared the treatment group (standard treatment for type 2 diabetes plus empagliflozin 10 mg daily) with control group (standard treatment for type 2 diabetes without empagliflozin) for 20 weeks and observed significant reduction in fatty liver in empagliflozin group (p<0.0001) when compared to control group. This finding supports the observations of a randomized controlled trial with inclusion of the EMPA REG outcome trial showed the hepatic enzyme reducing effect of empagliflozin [26].
The authors searched for the effect of empagliflozin on aminotransferases, which correlates with fatty liver in type 2 diabetes mellitus patients and that empagliflozin significantly reduces aminotransferases in patients with type 2 diabetes patients, in a pattern (reductions in ALT>AST) that is potentially consistent with a reduction in liver fat, especially when ALT levels are high.
Conclusion
Above findings point to the evidence of SGLT-2 inhibitors for their effectiveness in the prevention and treatment of hepatocellular damage which is assessed with improvement of liver functional parameters and inflammatory markers. So we can conclude that SGLT-2 inhibitors have a protective role against hepatocellular damage in type 2 diabetes patients by providing glycemic control and suppressing the pro-inflammatory cytokines and oxidative stress. However, it needs a well-designed clinical trial with enrolment of large number of patients with type 2 diabetes and NAFLDs to make authoritative comment on its efficacy in this condition. The animal studies have given some promising evidence for the positive effect of SGLT-2 inhibitors for the treatment of hepatocellular carcinoma. But it requires a well conducted clinical study to prove this finding.
Declarations
Funding
None
Conflict of interest
No conflict of interest to be declared
Ethical approval
Not required
REFERENCES
- Halimi S, Vergès B. Adverse effects and safety of SGLT-2 inhibitors. Diabetes Metab. 2014;40(6):S28-S34.
- Mather A, Pollock C. Glucose handling by the kidney. Kidney Int. 2011;79:S1-S6.
- List JF, Whaley JM. Glucose dynamics and mechanistic implications of SGLT-2 inhibitors in animals and humans. Kidney Int. 2011;79: S20-S27.
- Foote C, Perkovic V, Neal B. Effects of SGLT-2 inhibitors on cardiovascular outcomes. Diab Vasc Dis Res. 2012;9(2):117-123.
- Taylor S, Blau J, Rother K. SGLT2 inhibitors may predispose to ketoacidosis. J Clin Endocrinol Metab. 2015;100(8):2849-2852.
- Patoulias D. SGLT-2 Inhibitors: Are they a promising treatment option in t2dm patients with NAFLD? Acta Medica (Hradec Kralove). 2017;60(4):167-170.
- Cusi K, Sanyal AJ, Zhang S, Hartman ML, Bue-Valleskey JM, Hoogwerf BJ, et al. Non-alcoholic fatty liver disease (NAFLD) prevalence and its metabolic associations in patients with type 1 diabetes and type 2 diabetes. Diabetes Obes Metab. 2017;19(11):1630-1634.
- Dharmalingam M, Yamasandhi PG. Nonalcoholic fatty liver disease and type 2 diabetes mellitus. Indian J Endocrinol Metab. 2018;22(3):421-428.
- Chen Y, Li X, Wu S, Ye W, Lou L. Metabolic syndrome and the incidence of hepatocellular carcinoma: a meta-analysis of cohort studies. Onco Targets Ther. 2018;11:6277-6285.
- Yang WS, Va P, Bray F, Gao S, Gao J, Li HL, et al. The role of pre-existing diabetes mellitus on hepatocellular carcinoma occurrence and prognosis: a meta-analysis of prospective cohort studies. PLoS One. 2011;6(12):e27326.
- Inagaki N, Kondo K, Yoshinari T, Takahashi N, Susuta Y, Kuki H. Efficacy and safety of canagliflozin monotherapy in Japanese patients with type 2 diabetes inadequately controlled with diet and exercise: a 24-week, randomized, double-blind, placebo-controlled. Phase III study. Expert Opin Pharmacother. 2014;15(11):1501–1515.
- Cefalu WT, Leiter LA, Yoon KH, Arias P, Niskanen L, Xie J, et al. Efficacy and safety of canagliflozin versus glimepiride in patients with type 2 diabetes inadequately controlled with metformin (CANTATA-SU): 52 week results from a randomised, double-blind, phase 3 non-inferiority trial. Lancet. 2013;382(9896):941-950.
- Inagaki N, Kondo K, Yoshinari T, Kuki H. Efficacy and safety of canagliflozin alone or as add-on to other oral antihyperglycemic drugs in Japanese patients with type 2 diabetes: a 52-week open-label study. J Diabetes Investig. 2015;6(2):210-218.
- Tahara A, Kurosaki E, Yokono M, Yamajuku D, Kihara R, Hayashizaki Y, et al. Effects of SGLT2 selective inhibitor ipragliflozin on hyperglycaemia, hyperlipidemia, hepaticsteatosis, oxidative stress, inflammation, and obesity in type 2 diabetic mice. Eur J Pharmacol. 2013;715(1-3):246-255.
- Qiang S, Nakatsu Y, Seno Y, Fujishiro M, Sakoda H, Kushiyama A, et al. Treatment with the SGLT2 inhibitor luseogliflozin improves nonalcoholic steatohepatitis in a rodent model with diabetes mellitus. Diabetol Metab Syndr. 2015;7(1):104.
- Someya HY, Kurosaki E, Takasu T, Mitori H, Yamazaki S, Koide K, et al. Ipragliflozin, an SGLT2 inhibitor, exhibits a prophylactic effect on hepatic steatosis and fibrosis induced by choline-deficient l-amino acid-defined diet in rats. Eur J Pharmacol. 2015;754:19-24.
- Nakano S, Katsuno K, Isaji M, Nagasawa T, Buehrer B, Walker S, et al. Remogliflozin etabonate improves fatty liver disease in diet-induced obese male mice. J Clin Exp Hepatol. 2015;5(3):190-198.
- Leiter LA, Forst T, Polidori D, Balis DA, Xie J, Sha S. Effect of canagliflozin on liver function tests in patients with type 2 diabetes. Diabetes Metab. 2016;42(1):25-32.
- Ohki T, Isogawa A, Toda N, Tagawa K. Effectiveness of Ipragliflozin, a sodium-glucose co-transporter 2 inhibitor, as a second-line treatment for non-alcoholic fatty liver disease patients with type 2 diabetes mellitus who do not respond to incretin-based therapies including glucagon-like peptide-1 analogs and dipeptidyl peptidase-4 inhibitors. Clin Drug Investig. 2016;36(4):313-319.
- Levine JA, Ann Lo A, Wallia A, Rogers M, Van Wagner LB. Dapagliflozin-induced acute-on-chronic liver injury. ACG Case Rep J. 2016;3(4):e169.
- Tang L, Wu Y, Tian M, Sjöström CD, Johansson U, Peng XR, et al. Dapagliflozin slows the progression of the renal and liver fibrosis associated with type 2 diabetes. Am J Physiol Endocrinol Metab. 2017;313(5):E563-E576.
- Wang D, Luo Y, Wang X, Orlicky DJ, Myakala K, Yang P, et al. The sodium-glucose cotransporter 2 inhibitor dapagliflozin prevents renal and liver disease in western diet induced obesity mice. Int J Mol Sci. 2018;19(1):137.
- Tobita H, Sato S, Miyake T, Ishihara S, Kinoshita Y. Effects of dapagliflozin on body composition and liver tests in patients with non-alcoholic steatohepatitis associated with type 2 diabetes mellitus: a prospective, open-label, uncontrolled study. Curr Ther Res Clin Exp. 2017;87:13-19.
- Seko Y, Sumida Y, Tanaka S, Mori K, Taketani H, Ishiba H, et al. Effect of sodium glucose cotransporter 2 inhibitor on liver function tests in Japanese patients with non-alcoholic fatty liver disease and type 2 diabetes mellitus. Hepatol Res. 2017;47(10):1072-1078.
- Kuchay MS, Krishan S, Mishra SK, Farooqui KJ, Singh MK, Wasir JS, et al. Effect of empagliflozin on liver fat in patients with type 2 diabetes and nonalcoholic fatty liver disease: a randomized controlled trial (E-LIFT Trial). Diabetes Care. 2018;41(8):1801-1808.
- Sattar N, Fitchett D, Hantel S, George JT, Zinman B. Empagliflozin is associated with improvements in liver enzymes potentially consistent with reductions in liver fat: results from randomized trials including the EMPA-REG OUTCOME® trial. Diabetologia. 2018;61(10):2155-2163.
Citation: Mirghani HO, Thaniyath TA, Elfakki FA (2019) The Effects of Sodium-Glucose Co-Transporter-2 Inhibitors (SGLT-2-i) on Fatty Liver, Steato-Hepatitis, Liver Fibrosis, and Hepatocellular Carcinoma-A Review. Biol Med (Aligarh) 11:459.
Copyright: © 2019 Mirghani HO, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.


