Research Article - (2020) Volume 11, Issue 11
Background: This is a prospective, open-label, drug and dose-controlled study. The primary objective of this study is to evaluate the safety and efficacy of cannabidiol (CBD) sublingual tablets for the treatment of patients with chronic diabetic neuropathic pain. The secondary objects are to evaluate the impact of CBD tablets on sleep quality and anxiety. We describe the effectiveness data at the completion of a 21-day study.
Methods: Subjects who met all inclusion/exclusion criteria and met the prespecified minimum Numerical Pain Rating Scale (NPRS) score were eligible for participation. Thirty-one subjects with chronic diabetic neuropathic pain were recruited. Patients were instructed to take CBD sublingual tablets three times a day and were taught to use a smart phone data collection application to record daily NPRS scores.
Results: At the conclusion of the study, patients reported significant reduction in pain from baseline. The secondary endpoints analysing the effects of the CBD tablets on sleep quality and anxiety score revealed statistically significant improvement in both. No adverse drug reactions were reported.
Conclusions: Our findings demonstrate that the sublingual application of CBD tablets can achieve significant reduction in pain, as well as significant improvement in sleep quality and a reduction in anxiety and without any adverse drug reactions in patients with diabetic peripheral neuropathy.
Cannabidiol; CBD; Diabetic peripheral neuropathy; Sublingual tablet
Diabetic peripheral neuropathy (DPN) is a type of nerve damage caused by diabetes that leads to pain in feet, legs, and/or hands (stocking glove distribution), and affects up to 50% of patients with diabetes [1]. Diagnosis of the disease can be complex, as patients report their experience of the condition differently, ranging from mild to extreme pain. In many cases, patients may present neuropathic deficits while being asymptomatic. A lack of patients’ awareness of DPN, coupled with a scarcity of disease specific treatments has led to inadequate care and a rise in health care costs [1]. Patients diagnosed with DPN, in addition to suffering from pain, are also at a higher risk of developing insensate foot ulceration, which could lead to gangrene, sepsis, and/or amputation.
Tight control of blood sugar and pharmacological management remains at the forefront of the treatment of diabetic neuropathic pain. The only FDA approved drugs for this indication are pregabalin (Lyrica) and duloxetine (Cymbalta); however, they both carry several adverse effects. Pregabalin and duloxetine can both cause dizziness, dry mouth, nausea, constipation, and fatigue, while also having their own unique side effects as well. Pregabalin may cause edema, weight gain, amnesia, tremors, reduced platelet counts, possibly heart failure, and the FDA recently added the warning of severe respiratory depression especially when taken in concert with opioids or other respiratory depressants and in patients with concurrent respiratory illnesses such as COPD [2]. Duloxetine may cause seizures and sexual dysfunction, and also carries a black box warning, the most severe warning that can be issued by the FDA, for suicidal ideation.
Tricyclic agents are currently the most commonly prescribed drugs to treat pain associated with DPN; however, due to the frequency and severity of their adverse effects, their use is often restricted [1]. For patients with mild pain, physicians often recommend nonsteroidal anti-inflammatory drugs (NSAIDs), although they are not very effective and may lead to renal impairment, heart attack, stroke, and gastric ulcer with use [3]. Other methods of pharmacological treatment may include tramadol, local anaesthetic anti-arrhythmic agents (lidocaine), or a combination of drugs. Opioids are not currently FDA approved for the treatment of chronic pain in DPN [1]. In conjunction with pharmaceutical drugs, patients may turn to physical treatments for pain relief, including percutaneous nerve stimulation, low-intensive laser therapy, static magnetic field therapy, monochromatic infrared light, and electrical spinal cord stimulation with mixed results. There is also a need for randomized controlled studies to investigate the effectiveness of these methods. Many patients have tried alternative pain-relieving methods (acupuncture, massage therapy etc.) again with mixed results.
The presence of medical cannabis in 33 states and the district of Columbia has stimulated interest in physicians and researchers to understand whether or not cannabis, cannabinoids, and specifically cannabidiol (CBD) can be effectively used for medicinal purposes. Cannabis sativa L contains over 400 bioactive molecules. Delta- 9-tetrahydrocannabinol (THC) and Cannabidiol (CBD) are two molecules of interest and are the most studied of the chemicals specific to the cannabis plant. Almost every living organism has an endocannabinoid system wherein THC interacts directly, and CBD interacts indirectly to cause a physiological effect [4]. CBD specifically is known to not cause an intoxicating feeling as does THC but CBD still has bioactive properties that may result in pain relief, reduction in anxiety, improvement in sleep, and reduction in inflammation. GW Pharma has FDA approval to use a botanically derived oral preparation of CBD for the treatment of seizures associated with Lennox-Gastout and Dravet Syndrome.
Animal research has examined cannabinoids for the treatment of glycine induced neuropathic pain with results that suggest that CBD may be an effective treatment for neuropathy [5]. Another animal research model showed success using CBD to prevent paclitaxel induced peripheral neuropathy [6]. Clinically, there is anecdotal evidence that suggests that CBD can reduce pain in patients with diabetic neuropathy [7].
Pure Green Pharmaceuticals is conducting a clinical research study to determine if a fixed dose of CBD can be used as an alternative to, or in conjunction with, traditional pain medicine for the treatment of DPN. Secondary endpoints include examining sleep improvements and reductions in anxiety throughout the trial. CBD sublingual tablets are a non-intoxicating cannabis formulation in a rapidly dissolving sublingual tablet. This study was approved by an independent ethics committee and was carried out in accordance with The Code of Ethics of the World Medical Association (Declaration of Helsinki). Written informed consent was obtained from all subjects prior to inclusion into the trial.
Chemicals and reagents
Thirty-one subjects (13 male; 18 female) between the ages of 23 and 73 and were diagnosed with diabetic peripheral neuropathy were recruited to a 3-week open label, dose-controlled clinical trial using CBD sublingual tablets for the treatment of diabetic neuropathic pain. The inclusion criteria for entry into the study were:
• Subject is at least 21 years of age.
• Subject has a diagnosis of chronic diabetic neuropathic pain condition as determined by the subject’s health care or allied health provider for which medications other than cannabis, cannabinoids, or cannabis-based medicines are currently utilized.
• Subject has a 7-day average pain scale score (recorded during the screening period) of ≥ 5.
• If female, subject is postmenopausal (>1 year), surgically sterile, or practicing an approved method of birth control throughout the study and for 5 months (150 days) after the last dose of study drug.
• If female and of childbearing potential, subject has a denied pregnancy and the desire to become pregnant.
• Subject will not take marijuana (cannabis) in any form, chemicals or extracts or foods or beverages or topical creams, lotions, gels, patches containing marijuana (cannabinoids, or and cannabis derivatives) including synthetic marijuana and/ or CBD for at least 30 days prior to this study., and you promise that you will not take marijuana (cannabis) in any form, chemicals or extracts or foods or beverages or topical creams, lotions, gels, patches containing marijuana (cannabinoids, or and cannabis derivatives) including synthetic marijuana and/ or CBD while participating in this study.
• Subject is willing and able to provide his/her written informed consent to participate in the study as stated in the informed consent document.
• Subject has access to a smart phone and knows how to use smart phone applications.
The exclusion criteria of the study were:
• Subject is pregnant or lactating.
• Subject has an allergy to cannabis, the Cannabaceae plant family (e.g., hemp, hops, hackberry), PEA, terpenes, peppermint.
• Subject has a known allergy to active or inert ingredients of Pure Green™ tablets.
• Subject is currently treating their pain with cannabis, cannabinoids, cannabis-base medicine.
• Subject is taking a concomitant medication or treatment that would complicate use or interpretation of the study drug’s effects (examples include: Cannabis or any cannabinoid products; Any drug or herbal product that influences the endocannabinoid system (ECS)).
• Subject is taking marijuana (cannabis) in any form, chemicals or extracts or foods or beverages or topical creams, lotions, gels, patches containing marijuana (cannabinoids, or and cannabis derivatives) including synthetic marijuana and/or CBD for at least 30 days prior to this study., and does not promise to not take marijuana (cannabis) in any form, chemicals or extracts or foods or beverages or topical creams, lotions, gels, patches containing marijuana (cannabinoids, or and cannabis derivatives) including synthetic marijuana and/or CBD while participating in this study.
• Subject is currently being treated with antibiotics for sinus, throat, or lung infections.
• Subject has shortness of breath associated with allergies.
• Subject has uncontrolled asthma.
• Subject has a fever and/or productive cough.
• Subject does not have access to a smart phone or does not know how to use a smart phone application.
The test product is a water-soluble, sublingual, 20 mg CBD tablet. Subjects enrolled in the study participated for a total of 21 days using an open-label study design, and received compensation for their participation. The primary objective of the study was to evaluate the safety and efficacy of CBD sublingual tablets for the treatment of patients with chronic diabetic neuropathic pain. The secondary objectives were to evaluate the impact of CBD tablets on sleep quality and anxiety.
Immediately upon enrollment into the trial, subjects were asked to complete a Pure Green™ pain questionnaire utilizing a numerical pain rating scale (NPRS), the Pittsburgh Quality of Life Index (PSQI), and were asked to be rated using the Hamilton Anxiety Rating Scale (HAM-A). Demographic information and baseline characteristics, including age, sex, race, and ethnicity, height, weight, medical history, and concomitant medications were also recorded. Subjects were asked to continue taking their prescribed medication as recommended by their primary care physician. The NPRS options ranged from 0 (no pain) to 10 (worst possible pain) in order to assess the level of pain experienced by the subjects over the 21-day treatment period. Subjects were assessed for overall pain reported throughout the trial, as well as highest reportable pain at any given time during the study. The HAM-A rater index was scored out of a total of 56, where each item was scored on a scale of 0 (not present) to 4 (severe). The PSQI assessed overall sleep quality and was scored out of 24. To calculate sleep quality, an inverse percentage score is used for reporting purposes in this manuscript.
Each subject received a 21-day supply of CBD sublingual tablets to be taken one tablet 3 times a day, 6 hours apart (morning, afternoon, and evening). Subjects were then instructed on the utilization of a smart phone data collection application (App) for recording study treatment, pain scores, adverse events, and concomitant medications. Following the 21-day treatment period, subjects were asked to return to the clinical site to recomplete the Pure Green™ pain questionnaire, HAM-A, and the PSQI. In addition, subjects were asked about their overall well-being, including a review of study drug tolerance and adverse events over the course of the treatment period. The NPRS, HAM-A, and PSQI were statistically assessed following completion of the trial using mean calculations, Student’s Paired T-test, Signed Rank test, and the Kruskal-Wallis test. This study was registered on ClinicalTrials.Gov, located at www.clinicaltrials.gov, with the following registration ID: NCT04088929.
The study took place on October 3, 2019 and concluded November 25, 2019. Table 1 summarizes clinical data of the study participants. The subjects had an average age of 52 years. There were no adverse events reported in the study and all 31 subjects completed the trial. The mean subject overall NPRS scores at baseline was 6.83 for subjects taking prescribed medication for pain, and the mean overall NPRS scores at baseline for subjects not currently taking prescription medication was 6.62, with a range of 3 to 9. Over the 21-day treatment period, monitored subjects experienced reduction in their overall pain and reported a mean overall NPRS score of 3.35 (with prescription medication) and 3.38 (without prescription medication) (Figure 1). All subjects experienced a significant reduction in overall pain (p<0.0001) over the 21-day treatment period (Table 2). There were no observable differences in subjects’ overall pain scores for subjects taking medication versus those reporting no medication use (p=0.9807) (Table 3).
| Row | Variable | Min. | Q1 | Median | Mean | Q3 | Max. |
|---|---|---|---|---|---|---|---|
| 1 | Age | 23 | 44 | 54 | 52 | 61 | 73 |
| 2 | Pre-Trial Overall Pain Score | 3 | 6 | 7 | 6.8 | 8 | 9 |
| 3 | Post-Trial Overall Pain Score | 0 | 1 | 4 | 3.4 | 5 | 7 |
| 4 | PSQI Pre (100% - ×/24) | 0.042 | 0.33 | 0.5 | 0.52 | 0.69 | 0.92 |
| 5 | PSQI Post (100% - ×/24) | 0.38 | 0.77 | 0.83 | 0.78 | 0.88 | 0.96 |
| 6 | HAM-A Pre (×/56) | 0.054 | 0.19 | 0.34 | 0.38 | 0.55 | 0.8 |
| 7 | HAM-A Post (×/56) | 0 | 0.045 | 0.071 | 0.13 | 0.18 | 0.61 |
| 8 | PSQI Pre Raw Score | 2 | 7.5 | 12 | 12 | 16 | 23 |
| 9 | PSQI Post Raw Score | 1 | 3 | 4 | 5.3 | 5.5 | 15 |
| 10 | HAM-A Pre Raw Score | 3 | 10 | 19 | 21 | 31 | 45 |
| 11 | HAM-A Post Raw Score | 0 | 2.5 | 4 | 7.1 | 10 | 34 |
| 12 | Difference of PSQI Raw Score | -14 | -9 | -6 | -6.3 | -3 | 0 |
| 13 | Difference of HAM-A Raw Score | -34 | -23 | -13 | -14 | -8 | 2 |
| 14 | Difference of Highest Pain Score | -8 | -5 | -3 | -3.6 | -2 | 0 |
| 15 | Difference of Overall Pain Score | -9 | -4.5 | -3 | -3.4 | -2 | 0 |
Table 1: Summary statistics of 31 subjects at pre- and post-trial after taking one CBD tablet three times a day for 21 days.
| Signed Rank Test | Paired t-Test | |||||
|---|---|---|---|---|---|---|
| Row | Response | Mean Difference | Test Stat. | p-Value | Test Stat. | p-Value |
| 1 | PSQI Raw Score | -6.29 | 0 | <0.0001 | -8.61 | <0.0001 |
| 2 | HAM-A Raw Score | -14.19 | 3 | <0.0001 | -8.63 | <0.0001 |
| 3 | Highest Pain Score | -3.61 | 0 | <0.0001 | -8.9 | <0.0001 |
| 4 | Overall Pain Score | -3.42 | 0 | <0.0001 | -8.31 | <0.0001 |
Table 2: Significance testing of the PSQI, HAM-A, and pain (highest and overall) of the mean differences between pre- and post-trial scores.
| Summary Statistics | Difference of Means | Kruskal-Wallis Test | t-Test | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Row | Response | Group | Level | N | Min. | Max. | Mean | Std. Dev. | Median | Cl Lower | CI Upper | Test Stat p-Value | p-Value | Test Stat | d.f. | p-Value |
| 1 | Difference of PSQI Raw Score | Gender | Male | 13 | -13 | 0 | -4.85 | 3.72 | -4 | -5.38 | 0.41 | 2.79 | 0.095 | -1.76 | 27.38 | 0.0891 |
| 2 | Difference of PSQI Raw Score | Gender | Female | 18 | -14 | 0 | -7.33 | 4.09 | -8 | |||||||
| 3 | Difference of PSQI Raw Score | CBD Activation | Immediate | 10 | -13 | 0 | -5 | 3.74 | -4 | -5.02 | 1.21 | 1.41 | 0.2344 | -1.28 | 19.64 | 0.2163 |
| 4 | Difference of PSQI Raw Score | CBD Activation | Delayed | 21 | -14 | 0 | -6.9 | 4.16 | -8 | |||||||
| S | Difference of PSQI Raw Score | Medication Taken | TRUE | 23 | -13 | 0 | -5.87 | 3.83 | -5 | -5.76 | 2.49 | 0.67 | 0.4142 | -0.88 | 10.35 | 0.4006 |
| 6 | Difference of PSQI Raw Score | Medication Taken | FALSE | 8 | -14 | -1 | -7.5 | 4.75 | -8 | |||||||
| 7 | Difference of HAM-A Raw Score | Gender | Male | 13 | -28 | 2 | -10.92 | 9.05 | -9 | -12.31 | 1.04 | 2.91 | 0.0879 | -1.74 | 25.41 | 0.0945 |
| 8 | Difference of HAM-A Raw Score | Gender | Female | 18 | -34 | -4 | -16.56 | 8.71 | -13 | |||||||
| 9 | Difference of HAM-A Raw Score | CBD Activation | Immediate | 10 | -26 | -4 | -14.4 | 8.25 | -13 | -6.7 | 7.31 | 0 | 0.9662 | 0.09 | 20.81 | 0.9287 |
| 10 | Difference of HAM-A Raw Score | CBD Activation | Delayed | 21 | -34 | 2 | -14.1 | 9.75 | -11 | |||||||
| 11 | Difference of HAM-A Raw Score | Medication Taken | TRUE | 23 | -28 | 1 | -14.09 | 8.59 | -13 | -10.14 | 9.32 | 0.02 | 0.8919 | -0.09 | 9.99 | 0.9265 |
| 12 | Difference of HAM-A Raw Score | Medication Taken | FALSE | 8 | -34 | 2 | -14.5 | 11.26 | -11.5 | |||||||
| 13 | Difference of Highest Pain Score | Gender | Male | 13 | -7 | 0 | -2.77 | 2.05 | -3 | -3.05 | 0.14 | 2.81 | 0.0939 | -1.86 | 27.44 | 0.073 |
| 14 | Difference of Highest Pain Score | Gender | Female | 18 | -8 | -1 | -4.22 | 2.26 | -4 | |||||||
| 15 | Difference of Highest Pain Score | CBD Activation | Immediate | 10 | -8 | -1 | -5.3 | 2.41 | -S.5 | 0.67 | 4.32 | 7.09 | 0.0077 | 2.94 | 13.56 | 0.0112 |
| 16 | Difference of Highest Pain Score | CBD Activation | Delayed | 21 | -6 | 0 | -2.81 | 1.72 | -3 | |||||||
| 17 | Difference of Highest Pain Score | Medication Taken | TRUE | 23 | -8 | 0 | -3.78 | 2.37 | -3 | -1.16 | 2.47 | 0.32 | 0.5694 | 0.77 | 14.74 | 0.4521 |
| 18 | Difference of Highest Pain Score | Medication Taken | FALSE | 8 | -6 | 0 | -3.13 | 1.96 | -3 | |||||||
| 19 | Difference of Overall Pain Score | Gender | Male | 13 | -6 | 0 | -2.77 | 1.79 | -3 | -2.71 | 0.47 | 1.44 | 0.2302 | -1.44 | 28.99 | 0.1605 |
| 20 | Difference of Overall Pain Score | Gender | Female | 18 | -9 | 0 | -3.89 | 2.54 | -3 | |||||||
| 21 | Difference of Overall Pain Score | CBD Activation | Immediate | 10 | -9 | -3 | -5.3 | 2.26 | -5 | 1.04 | 4.51 | 10.51 | 0.0012 | 3.43 | 14.16 | 0.004 |
| 22 | Difference of Overall Pain Score | CBD Activation | Delayed | 21 | -7 | 0 | -2.52 | 1.72 | -2 | |||||||
| 23 | Difference of Overall Pain Score | Medication Taken | TRUE | 23 | -9 | 0 | -3.48 | 2.31 | -3 | -1.88 | 2.34 | 0.01 | 0.9087 | 0.24 | 11.97 | 0.8177 |
| 24 | Difference of Overall Pain Score | Medication Taken | FALSE | 8 | -7 | 0 | -3.25 | 2.38 | -3 | |||||||
Table 3: Significance testing of difference scores of the PSQI, HAM-A, and pain (highest and overall) by factor between pre- and post-trial scores. Factors include gender, CBD activation (immediate v. delayed reaction), and whether subjects were concomitantly taking prescription medication.
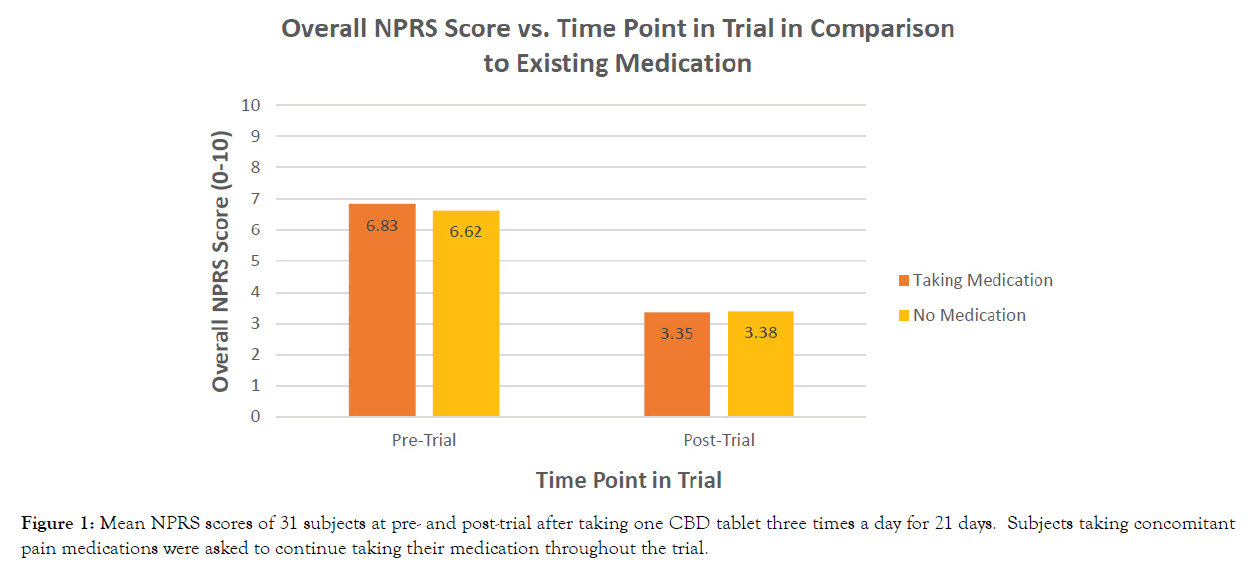
Figure 1: Mean NPRS scores of 31 subjects at pre- and post-trial after taking one CBD tablet three times a day for 21 days. Subjects taking concomitant pain medications were asked to continue taking their medication throughout the trial.
Subjects’ highest reportable pre-trial pain scores were 9.04 (with prescription medication) and 8.75 (without prescription medication). All subjects’ highest reportable pre-trial pain scores also significantly decreased post-trial with a mean NPRS score of 3.35 (with prescription medication) and 3.38 (without prescription medication) (p<0.0001) (Figure 2). Differences in pain scores between male and female subjects were not observed (p=0.0939), and no notable differences were identified in subject’s highest reported pain scores for subjects taking medication versus those reporting no medication use (p=0.5694) (Table 3).
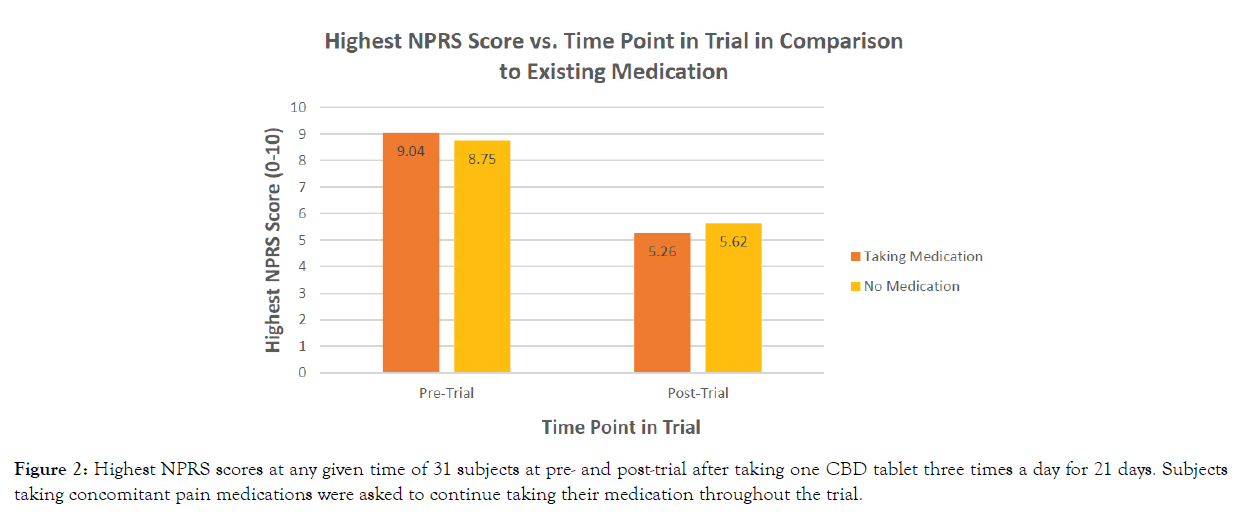
Figure 2: Highest NPRS scores at any given time of 31 subjects at pre- and post-trial after taking one CBD tablet three times a day for 21 days. Subjects taking concomitant pain medications were asked to continue taking their medication throughout the trial.
Initial scores on the HAM-A were shown to drop 3-fold by the end of the trial. The mean HAM-A pre-trial scores were 21.83 (with prescription medication) and 19.88 (without prescription medication). Following the 21-day treatment period, HAM-A post trial scores dropped to 7.74 (with prescription medication) and 5.38 (without prescription medication) (Figure 3). Subjects’ overall anxiety recorded on the HAM-A was shown to significantly decrease over the duration of the trial (p<0.0001) (Table 2), with no difference in whether subjects were taking prescription pain medications (p=0.8919) (Table 3).
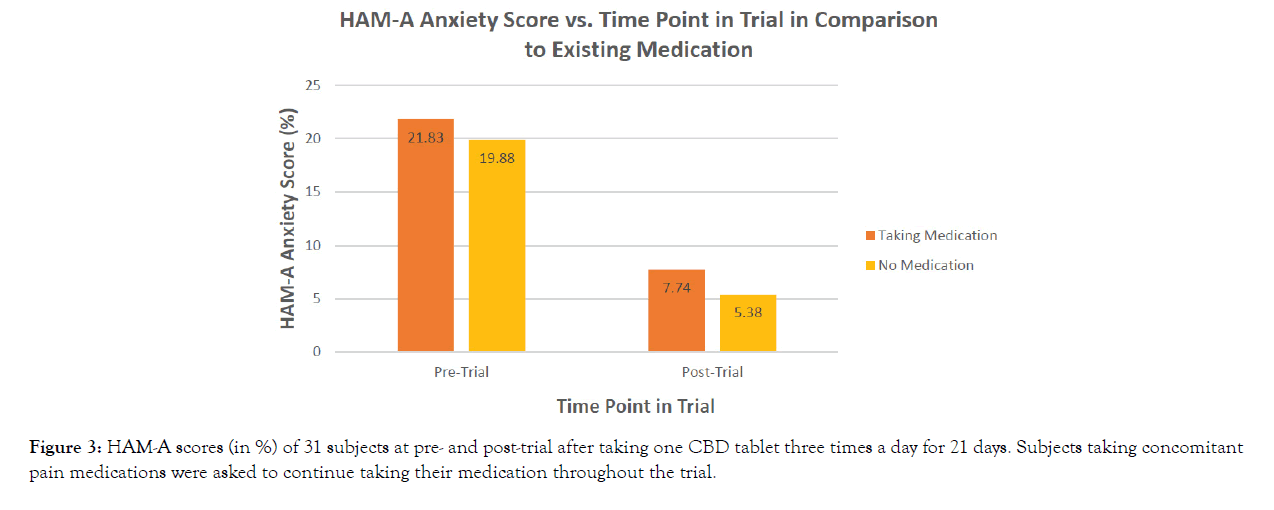
Figure 3: HAM-A scores (in %) of 31 subjects at pre- and post-trial after taking one CBD tablet three times a day for 21 days. Subjects taking concomitant pain medications were asked to continue taking their medication throughout the trial.
The mean of all subjects’ baseline PSQI scores is 52.9% (with prescription medication) and 48.96% (without prescription medication). Subjects demonstrated a minimum of a 24.4% improvement in sleep quality at the conclusion of the study, with the highest percentage increase valued at 31.3%. Following the 21-day treatment period, post-trial scores revealed 77.36% (with prescription medication) and 80.21% (without prescription medication) improvement in sleep quality (Figure 4). Differences in pre-treatment v. post-treatment scores recorded on the PSQI were statistically significant (p<0.0001) (Table 2). As shown in Table 3, scores did not significantly differ between subjects on or off prescription medications for diabetic neuropathic pain (p=0.4142).
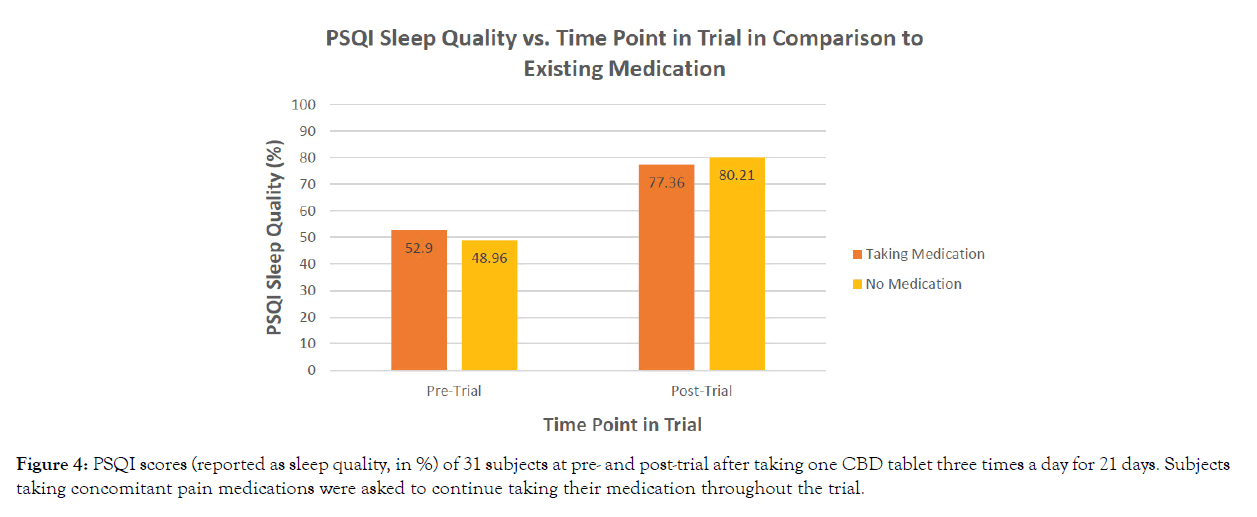
Figure 4: PSQI scores (reported as sleep quality, in %) of 31 subjects at pre- and post-trial after taking one CBD tablet three times a day for 21 days. Subjects taking concomitant pain medications were asked to continue taking their medication throughout the trial.
A post-treatment interview found that roughly one-third of subjects reported an immediate response to the CBD tablets, with results visibly noticeable within 24 hours of taking the first dose of study treatment. Individuals who identified as “delayed responders” reported a noticeable change after an average of 7 days into the 21-day treatment period. No differences were observed in subject’s age, medications, or demographics that could discern between immediate and delayed respondents. Although there was no statistically significant difference between scores in anxiety (p=0.9662) or sleep quality (p=0.2334), there was some observable difference between the immediate versus delayed respondents in overall pain scores (p=0.0012), but not statistically significant according to data analysis performed post-trial (Table 3).
The 31 subjects tested had an overall average pre-test pain NPRS score of 6.8. At the completion of the 3-week trial using the 20 mg CBD sublingual tablets at a fixed dosing regimen of one tablet three times a day the average NPRS score was 3.4. No subject required additional pain medications. Anecdotally, the 23 subjects taking concomitant pain medications requested to reduce or stop their existing prescription medications but were advised against it for this trial.
It is interesting to note that two thirds of the subjects had a delayed response to the pain-relieving effects of the tablets. Although there was a delay in response to treatment, the delayed responders had a statistically significant reduction in their NPRS score and there was no statistically significant difference between the immediate responders and the delayed responders, as both had statistically significant pain reduction. In this trial, more men than women experienced a delayed response. However, we cannot make a generalization regarding this as the power of the sample is too small to determine a correlation.
Subjects reported that their anxiety levels were noticeably reduced throughout their day and that they felt calmer and experienced a noticeable reduction of stress. This was reflected in the HAM-A scores where all but two subjects had a statistically significant reduction in their anxiety scores. The two subjects that did not experience a reduction in anxiety verbally reported no change in their anxiety level at their exit interview. Although the HAM-A scores of these two subjects, (subjects 3 and 13) had increased by one and two points out of a total of 56 points respectively, it was not a statistically significant increase nor was it a noticeable increase in anxiety to the subject.
Sleep quality was evaluated in this trial using the PSQI. At the completion of the trial subjects reported an overall improvement in their sleep by 33.6% that was reflected in the PSQI as a statistically significant improvement. Subjects with neuropathic pain often complain of poor sleep quality due to pain, stress and anxiety [8]. This effect is multi-factorial and is due to primary pain as well as pain induced stress and anxiety. The lack of quality sleep will in turn enhance pain. We postulate that since the subjects in the trial had a statistically significant improvement in both pain and anxiety it is likely that these improvements are interrelated to the measured improvement of sleep quality.
As this was an open label trial, it has the limitations inherent to the lack of randomization. Future studies will include a dose escalation in attempt to reduce the number of delayed responders, extending the length of treatment time, and feature a double-blind design.
The opioid epidemic has shined a bright light on the paucity of available safe and effective therapeutic options for the treatment of DPN. Current pain medications, either over the counter or prescription fall short in proving to be an effective solution. The current therapeutic options are also fraught with adverse effects including stroke, heart attack, gastric ulcers, and suicide. As evidenced by the subjects participating in this clinical trial, patients continue to take medications even if they are ineffective. With thirty-three states and the district of Columbia now having some sort of legal medical cannabis program, patient access to medical cannabis, and CBD, is increasing. However, there are no specific formulations or dosing guidelines for therapeutic indications. Physicians and other health care providers are not formally educated in traditional medical education platforms on the endocannabinoid system or how cannabis interacts within the endocannabinoid system, thereby leaving but a few self-taught healthcare providers with the understanding that medical cannabis could be a viable therapeutic option for the treatment of pain and specifically diabetic neuropathy. As referenced above, cannabis and CBD have been studied for various indications and have been shown to reduce inflammation, pain, and seizure activity, to name a few [4-7].
In addition, patients in this study reported significant positive changes in secondary endpoints, such as improved sleep quality and anxiety reduction. Understanding how cannabis and cannabinoids interact within the endocannabinoid system may explain these positive results [4]. With no serious adverse effects reported, the results in this clinical study may assist in understanding the impact of consistent and stable cannabis-derived formulations and dosing on diabetic neuropathic pain.
In this trial, the sublingual tablet formulation using CBD was used to treat diabetic neuropathic pain as the primary endpoint and improve the quality of sleep and reduce anxiety as secondary endpoints. The three-week trial revealed a statistically significant improvement of each of the three endpoints. The benefits of this study demonstrate a potential for advancement of safe alternative treatment for patients with chronic diabetic neuropathic pain condition.
The data used to support the findings of this study are included within the article. Access to individual subject data is restricted due to patient privacy and confidentiality.
Design and analyses: D.K., M.C., S.G.; manuscript writing: A.K., D.K., M.C., S.G.; manuscript revising for intellectual content: S.G. All authors approved the final version of the manuscript.
The authors are associated with Pure Green Pharmaceuticals, LLC, which developed and manufactured the Pure Green® CBD 20 mg tablets.
This work was solely financed by Pure Green Pharmaceuticals, LLC.
The authors would like to recognize and thank Heather Denham, who contributed significantly to this study. The authors would also like to thank all investigators, study teams, and patients for participating in this study.
Citation: Kimless D, Caloura MK, Kirakosyan A, Goldner S (2020) The effects of cannabidiol-based sublingual tablets on diabetic neuropathic pain. J Diabetes Metab. 11:860.
Received: 21-Sep-2020 Published: 11-Dec-2020
Copyright: �?�© 2020 Kimless D, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.