PMC/PubMed Indexed Articles
Indexed In
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- Ulrich's Periodicals Directory
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Research Article - (2020) Volume 11, Issue 1
Inhibitory Effect of Hybrid Liposomes on the Growth of NP2 Glioma Cells
Yoko Matsumoto*, Keiji Kuwabara and Hideaki IchiharaReceived: 04-Feb-2020 Published: 25-Feb-2020, DOI: 10.35248/2157-2518.20.11.344
Abstract
The inhibitory activity of hybrid liposomes (HLs), composed of 90 mol% L-α-dimyristoyl-phosphatidylcholine (DMPC) and 10 mol% polyoxyethylene (25) dodecyl ether (C12(EO)25), was investigated in human glioma NP2 cells. HLs with a hydrodynamic diameter below 100 nm persisted for 4 weeks. The inhibitory effect of HLs on the proliferation of NP2 cells was evaluated. Induction of apoptosis in NP2 cells treated with HL was measured through a PI assay and the TUNEL method. HLs caused apoptosis in NP2 cells through the mitochondrial pathway. An increase of AIF protein expression was observed in HL-treated cells. Cellular membrane fluidity of NP2 cells was also increased as revealed by the fluorescence depolarization method. Enhanced HL accumulation in the membrane of NP2 cells was observed.
Keywords
Hybrid Liposomes; Glioma; Apoptosis
Abbrevations
AIF: Apoptosis Inducing Factor; C12(EO)25: Polyoxyethylene(25)Dodecyl Ether; DMPC: L-α- dimyristoylphosphatidylcholine; HL: Hybrid Liposomes composed of DMPC and C12(EO)25; NBDPC: 1-palmitoyl-2-[12- [(7-nitro-2-1,3-benzoxadiazol-4-yl)amino]-dodecanoyl]-sn-glycero-3 phosphocholine; TUNEL: Terminal deoxynucleotidyl tranferasemediated dUTP-biotin nick end labeling.
Introduction
According to the latest pathological classification, there are over 100 types of brain tumors [1]. Treatment of patients with malignant brain tumors is not limited to surgery and employs radiotherapy and chemotherapy as well. The brain has glial cells that surround and support nerve cells. Astrocytoma, oligodendroglioma, and ependymoma are examples of gliomas, which account for approximately 25% of brain tumors. Brain tumors are classified into 4 grades of malignancy on the basis of pathological characteristics. In comparison to grade 1 pilocytic astrocytomas and grade 2 oligodendrogliomas, anaplastic astrocytomas and anaplastic oligodendrogliomas of grade 3 and grade 4 glioblastomas are highly malignant.
We have produced hybrid liposomes (HLs) that can be easily prepared by sonicating a mixture of vesicular and micellar molecules in buffer solution [2]. Inhibitory activity of hybrid liposomes composed of L-α-dimyristoylphosphatidylcholine (DMPC), polyoxyethylene (20) sorbitan monolaurate (Tween 20) and antitumor drugs such as 1, 3-bis (2-chloroethyl)-1-nitrosourea on the growth of glioma cells have been previously described both in vitro and in vivo [3]. HLs, composed of DMPC and polyoxyethylene (25) dodecyl ethers (C12 (EO)25), are effective in inhibiting the growth of various cancer cells in vitro [4-8], in vivo [9-12] and for clinical applications [13]. HLs discriminate between the more fluid membranes of cancer cells and less fluid membranes of normal cells, preferentially fusing and accumulating in cancer cell membranes [14]. However, the inhibitory activity of HLs on the proliferation of glioma cells has not yet been elucidated.
In the current study, we investigated apoptosis induction for human glioma NP2 (anaplastic astrocytoma; grade 3) cells by HLs.
Methodology
Preparation of HL
HLs were prepared using a sonicator (VS-N300; VELVO, Tokyo, Japan) after DMPC (purity>99%; NOF Co. Ltd., Tokyo, Japan) and C12(EO)25 (Nikko Chemicals Co., Ltd., Tokyo, Japan) were mixed in 5% glucose solution at 45°C, 300 W and filtered with a 0.20 μm filter.
Dynamic light scattering measurements
The diameter (dhy) of HLs was measured by a light scattering spectrometer (ELS-8000, Otsuka Electronics, Osaka, Japan) using a He−Ne laser (633 nm) at a scattering angle of 90°. dhy was calculated using the Stokes-Einstein formula by the below equation, where κ is the Boltzmann constant, T is the absolute temperature, η is the viscosity and D is the diffusion coefficient:
Cell culture
The human glioma (anaplastic astrocytoma, grade 3) NP2 cell line was purchased from Riken Cell Bank (Ibaraki, Japan). NP2 cells were cultured in MEM medium (Gibco, Thermo Fisher Scientific, Inc. MA, USA) containing 10% fetal bovine serum (FBS, HyClone Laboratories Inc., UT, USA), penicillin (100 unit/ml) and streptomycin (50 μg/ml). Cells were kept in a humidified incubator at 37°C and 5% CO2.
Assessment of the half-maximal inhibitory concentration (IC50) of HL in vitro
The half-maximal inhibitory concentration (IC50) of HLs on the growth of NP2 cells was determined on the basis of a WST-8 [2-(2-methoxy-4-nitrophenyl)-3-(4-nitrophenyl)-5-(2, 4- disulfophenyl)-2H tetrazolium, monosodium salt] assay (Cell Counting Kit-8, Dojindo Laboratories, Kumamoto, Japan) [15]. NP2 (5.0 × 103 cells/ml) cells were seeded in 96-well plates and cultured in a 5% CO2 humidified incubator at 37°C for 24 h. Cells were cultured for a further 48 h after the addition of DMPC (0 ~ 2.0 × 10-3 M) and HLs (0 ~ 1.4 × 10-3 M depending on DMPC concentration). WST-8 solution was then added and the incubated for 3 h. The absorbance at a wavelength of 450 nm was measured using a microplate reader (Thermo, CA, USA). The inhibitory activity of HLs on the growth of NP2 cells was evaluated by Amean/Acontrol, where Amean and Acontrol denote the absorbance of water-soluble formazan in the presence and absence of HL, respectively.
Analysis of apoptosis induction by HLs on the basis of a PI assay using flow cytometry
Cells were seeded at a density of 5.0 × 103 cells per 100 ml dish and incubated in a humidified atmosphere of 5% CO2 at 37°C for 24 h. HLs (0.1 ~ 5.0 × 10-4 M depending on DMPC concentration) were added into each dish and the dishes were incubated. Cells were centrifuged and suspended in PBS (-), containing 1 mg/ml RNase, 0.1% Triton X-100 and 40 μg/ml propidium iodide (PI, Molecular Probes, Eugene, OR, USA), in a dark room. The percentage of apoptotic cells was analyzed using a flow cytometer (Epics XL system II, Beckman Coulter, Brea, CA, USA).
Terminal deoxynucleotidyl tranferase-mediated dUTPbiotin nick end labeling (TUNEL) assay
Detection of apoptotic cells was performed by the TUNEL method using the In Situ Cell Death Detection Kit (Roche Diagnostics, Basel, Switzerland). NP2 cells (5 × 103 cells/ml) were seeded in dishes and cultured for 24 h. HLs ([DMPC]=3.0×10-4 M, [C12(EO)25]=3.33 × 10-5 M) or DMPC liposomes were added and cells were cultured for another 48 h. 4% paraformaldehyde solution was added for fixation. After formalin was removed, cells were washed with Hank's Balanced Salt Solution (HBSS). Impregnated solution was added and cells were stored in darkness at 4°C for 2 min. After cells were washed, TUNEL reaction mixture was added, and cells were then incubated at 37°C for 60 min within a humectant box. Stained cells were observed under a confocal laser microscope (TCS-SP; Leica Microsystems, Wetzlar, Germany) with a 488 nm Ar laser line for TUNEL (detection at 515–565 nm).
Measurement of caspase activity through flow cytometry
NP2 cells (1.0 × 105 cells/ml) cells were treated with HLs. After 24 h of incubation, each substrate solution (PhiPhiLux-GID2, CaspaLux 8-LID2, CaspaLux 9-MID2 for caspase-3, caspase-8, and caspase-9; OncoImmunin Inc., Gaithersburg, MD, USA) was added to NP2 cells [16]. NP2 cells were then incubated for 45 min at 37°C. After washing twice with 1 ml of ice-cold PBS (-), NP2 cells were resuspended in 1 ml fresh PBS (-). Caspase activity was analyzed using a flow cytometer.
Mitochondrial membrane potential
3, 3-Dihexyloxacarbocyanine iodide [DiOC6 (3)] (Molecular Probes, Oregon, USA) was added to NP2 cells (1.0 × 106 cells/ ml), previously treated with HLs ([DMPC]=3.0 × 10-4 M, [C12 (EO)25]=3.33 × 10-5 M), to evaluate mitochondrial transmembrane potential (Δψm). Stained NP2 cells were examined through flow cytometry with the use of a 15 mW 488 nm air-cooled Ar laser and FL1 sensor (505–545 nm).
Assessment of AIF by flow cytometry
NP2 cells (5.0 × 103 cells/ml) cells were seeded in dishes and incubated for 24 h. NP2 cells were treated with HLs (3.0 × 10-4 M) and incubated for 24 h. Cells were then fixed with 10% formaldehyde for 30 min in wells, followed by incubation with anti-AIF Alexa Fluor® 488-conjugated antibody (5 μg/ml) (Abcam plc, Cambridge, UK) in a shaded box at 4°C for 1 h. Stained NP2 cells were analyzed by flow cytometry.
Fusion and accumulation of HL in the cell membrane
HL fusion and accumulation in the cell membrane was assessed using confocal laser microscopy (TCS-SP; Leica Microsystems, Berlin, Germany) and a fluorescent probe ((1-palmitoyl-2-[12-[(7- nitro-2-1,3-benzoxadiazol-4-yl)amino]-dodecanoyl]-sn-glycero-3- phosphocholine (NBDPC; Avanti Polar Lipids, Alabama, USA)). NP2 cells (2.0 × 104 cells/ml) were incubated in a humidified incubator at 37°C and 5% CO2 for 24 h. Cells were then treated with HLs/NBDPC ([DMPC]=99.6 μM, [C12 (EO)25]=1.11 μM, [NBDPC]=0.4 μM) for 10–60 min and were viewed under a confocal laser microscope using a 488 nm Ar laser line (detection at 505–555 nm).
Measurement of cellular membrane fluidity
The membrane fluidity of NP2 cells was measured using a spectrophotometer (F-7100; HITACHI) on the basis of the fluorescence depolarization method [12]. NP2 cells were labeled with DPH (1, 6-diphenyl-1, 3, 5-hexatriene, Nacalai Tesque, Japan). The fluorescence depolarization (P) value of DPH in cells treated with HLs (1.0 × 10-4 M) was measured.
Statistical Analysis
Data are presented as mean ±S.D. For statistical comparison, a Student ’ s t-test was performed. p<0.05 was considered to represent a statistically significant difference.
Results and Discussion
Physical properties of HL
We examined the morphology of HLs on the basis of dynamic light scattering. HLs with a hydrodynamic diameter less than 100 nm remained viable for 4 weeks. In contrast, DMPC liposomes were unstable and precipitated at 14 days after preparation. These results suggest that HLs with a diameter below 100 nm can avoid recognition by the reticular endothelial system (RES) [17], and, therefore, would be appropriate for administration through intravenous injection both in vivo and in the clinic.
Inhibitory effect of HLs on the growth of NP2 cells
With respect to the inhibitory activity of HLs on the growth of human glioma NP2 cells, we examined the half-maximal inhibitory concentration (IC50) of HLs on the basis of a WST-8 assay. The results are shown in Figure 1. The IC50 value of HLs was 233.8 ± 9.8 μM, whereas that of DMPC liposomes was 806.6 ± 19.5. It was evident that HL potently inhibited the growth of NP2 cells.
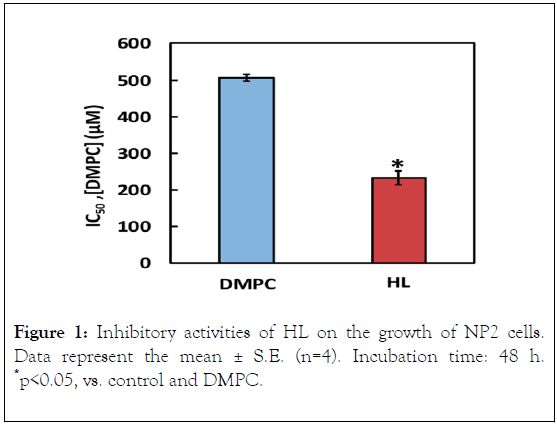
Figure 1. Inhibitory activities of HL on the growth of NP2 cells. Data represent the mean ± S.E. (n=4). Incubation time: 48 h. *p<0.05, vs. control and DMPC.
Apoptosis induction by HLs
We evaluated the apoptotic fraction of NP2 cells treated with HL on the basis of a PI assay using flow cytometer. Results are shown in Figure 2A. Apoptotic cell fraction increased after HL treatment, in a dose-dependent manner. The apoptotic fraction reached high levels (more than 95% of cells). Next, the induction of apoptosis of HL-treated NP2 cells was evaluated on the basis of the TUNEL method using confocal laser microscope. Results are shown in Figure 2B. Green fluorescence was observed in cells treated with HL using TUNEL staining. In contrast, green fluorescence was not observed in the case of DMPC liposome-treated cells. These results indicate that HLs induced apoptosis in NP2 cells.
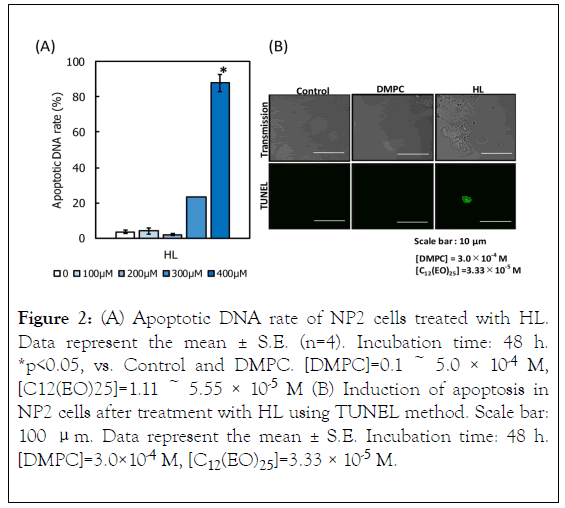
Figure 2. (A) Apoptotic DNA rate of NP2 cells treated with HL. Data represent the mean ± S.E. (n=4). Incubation time: 48 h. *p<0.05, vs. Control and DMPC. [DMPC]=0.1 ~ 5.0 × 10-4 M, [C12(EO)25]=1.11 ~ 5.55 × 10-5 M (B) Induction of apoptosis in NP2 cells after treatment with HL using TUNEL method. Scale bar: 100 μm. Data represent the mean ± S.E. Incubation time: 48 h. [DMPC]=3.0×10-4 M, [C12(EO)25]=3.33 × 10-5 M.
Apoptotic pathway in HL-treated NP2 cells
To investigate the apoptotic pathways triggered by HL treatment of NP2 cells, we examined caspase activation through flow cytometric analysis, as shown in Figure 3. Activities of caspase-3, 8, and 9 were not detected, suggesting that HLs induced apoptosis without their activation. Next, we investigated the mitochondrial pathway of apoptosis in HL-treated NP2 cells. The results are shown in Figure 4. After HL treatment, decreased mitochondrial transmembrane potential was observed in cells. Apoptosis inducing factor (AIF) is involved in initiating the caspase-independent pathway of apoptosis. Thus, AIF in NP2 cells treated with HL was measured through immunostaining and subsequent analysis by flow cytometer. The results are shown in Figure 5. AIF detected in NP2 cells after HL treatment was more abundant when compared to the levels detected in DMPC liposome-treated cells. Thus, AIF levels were increased in HL-treated cells. The data indicate that AIF was involved in the induction of apoptosis caused by HLs. These results suggest that HL induced apoptosis via AIF, through a caspase-independent pathway.
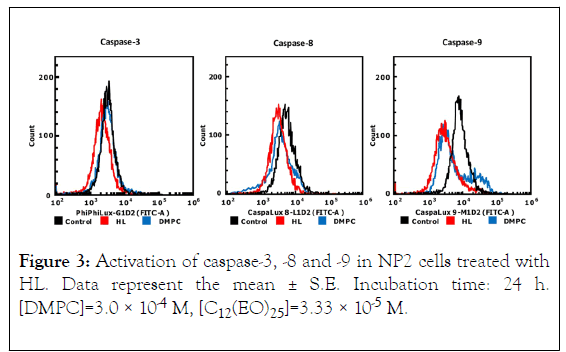
Figure 3. Activation of caspase-3, -8 and -9 in NP2 cells treated with HL. Data represent the mean ± S.E. Incubation time: 24 h. [DMPC]=3.0 × 10-4 M, [C12(EO)25]=3.33 × 10-5 M.
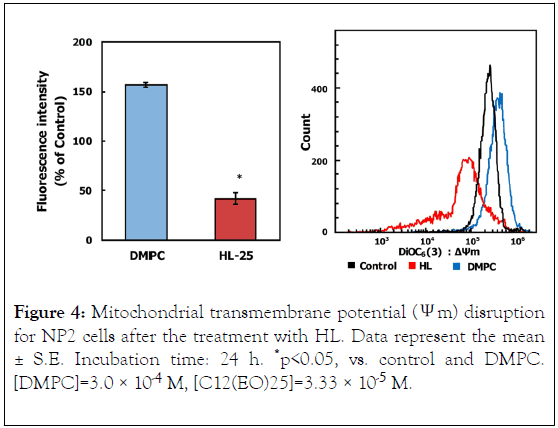
Figure 4. Mitochondrial transmembrane potential (Ψm) disruption for NP2 cells after the treatment with HL. Data represent the mean ± S.E. Incubation time: 24 h. *p<0.05, vs. control and DMPC. [DMPC]=3.0 × 10-4 M, [C12(EO)25]=3.33 × 10-5 M.
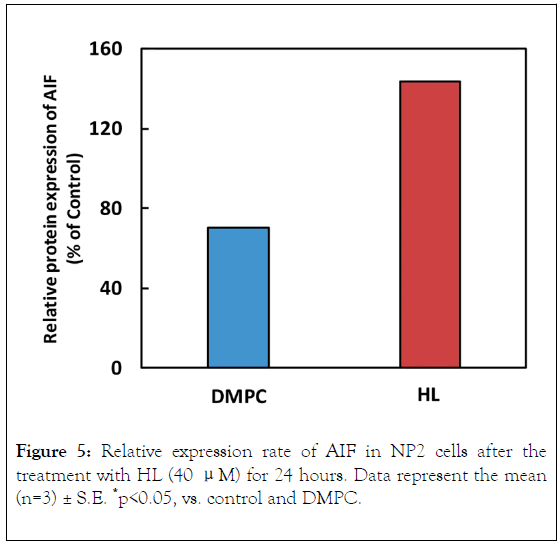
Figure 5. Relative expression rate of AIF in NP2 cells after the treatment with HL (40 μM) for 24 hours. Data represent the mean (n=3) ± S.E. *p<0.05, vs. control and DMPC.
HL fusion and accumulation in NP2 cells
The fusion and accumulation of HL in NP2 cells was evaluated through flow cytometer. The results are shown in Figure 6. A time-dependent increase in HL accumulation was observed using NBDPC as a fluorescence probe.
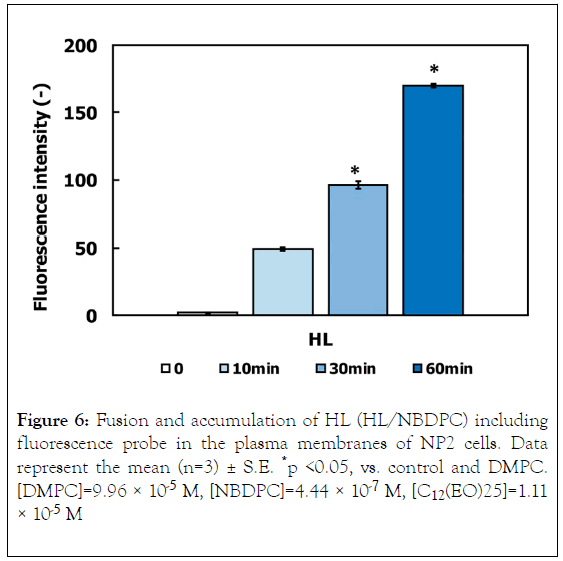
Figure 6. Fusion and accumulation of HL (HL/NBDPC) including fluorescence probe in the plasma membranes of NP2 cells. Data represent the mean (n=3) ± S.E. *p <0.05, vs. control and DMPC. [DMPC]=9.96 × 10-5 M, [NBDPC]=4.44 × 10-7 M, [C12(EO)25]=1.11 × 10-5 M
We examined membrane fluidity of NP2 cells on the basis of the fluorescence polarization (P) analysis. The results are shown in Figure 7. The P value decreased in NP2 cells treated with HL, indicative of increased membrane fluidity. These results suggest that the increase in membrane fluidity due to HL accumulation could be involved in the early events of HL-induced apoptosis.
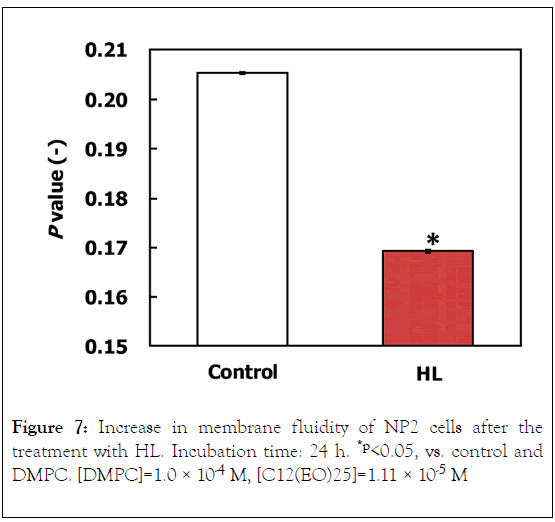
Figure 7. Increase in membrane fluidity of NP2 cells after the treatment with HL. Incubation time: 24 h. *p<0.05, vs. control and DMPC. [DMPC]=1.0 × 10-4 M, [C12(EO)25]=1.11 × 10-5 M
Conclusion
The current study demonstrated the inhibitory effect of hybrid liposomes (HLs) on the growth of human glioma NP2 (anaplastic astrocytoma) cells underlain by apoptosis induction through the accumulation of HL into cell membranes. The noteworthy findings are as follows: (a) HL with a hydrodynamic diameter less than 100 nm persisted for 4 weeks. (b) The inhibitory effect of HL on the growth of NP2 cells was observed, and it was driven by increased apoptosis. (c) HL caused apoptosis through the mitochondrial pathway. (d) HL induced apoptosis in NP2 cells via the release of AIF, a caspaseindependent pathway. (e) HL fused into NP2 cell membranes increasing their fluidity. A remarkably high inhibitory effect of HLs on NP2 cell growth was observed in vitro.
Acknowledgment
We thank Aya Yamashita for her technical assistance. This work was supported in part by a Grant-in-Aid for Science Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan (Nos. 17K01383 and 17K05944).
REFERENCES
- Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, Jouvet A, et al. The 2007 WHO Classification of Tumours the Central Nervous System. Acta Neuropathol. 2007;114(2):97-109.
- Ueoka R, Matsumoto Y, Moss RA, Swarup S, Sugii A, Harada K, et al. Membrane matrix for hydrolysis of amino acid esters with marked enantioselectivity. J Am Chem Soc. 1988;110(5):1588-1595.
- Kitamura I, Kochi M, Matsumoto Y, Ueoka R, Kuratsu J, Ushio Y. Intrathecal chemotherapy with 1,3-bis(2-chloroethyl)-1-nitrosourea encapsulated into hybrid liposomes for meningeal gliomatosis: An experimental study. Cancer Res. 1996;56(17):3986-3992.
- Matsumoto Y, Kato T, Iseki S, Suzuki H, Nakano K, Iwahara M, et al. Remarkably enhanced inhibitory effects of hybrid liposomes on the growth of specific tumor cells. Bioorg Med Chem Lett. 1999;9(14):1937-1940.
- Matsumoto Y, Iwamoto Y, Matsushita T, Ueoka R. Novel mechanism of hybrid liposomes-induced apoptosis in human tumor cells. Int J Cancer. 2005;115(3):377-382.
- Iwamoto Y, Matsumoto Y, Ueoka R. Induction of apoptosis of human lung carcinoma cells by hybrid liposomes containing polyoxyethylenedodecyl ether. Int J Pharm. 2005; 292(2):231-239.
- Nagami H, Nakano K, Ichihara H, Matsumoto Y, Ueoka R. Two methylene groups in phospholipids distinguish between apoptosis and necrosis for tumor cells. Bioorg Med Chem Lett. 2006;16(4):782-785.
- Komizu Y, Matsumoto Y, Ueoka R. Membrane targeted chemotherapy with hybrid liposomes for colon tumor cells leading to apoptosis. Bioorg Med Chem Lett. 2006;16(23):6131-6134.
- Kitajima H, Komizu Y, Ichihara H, Goto K, Ueoka R. Hybrid liposomes inhibit tumor growth and lung metastasis of murine osteosarcoma cells. Cancer Med. 2013;2(3):267-276.
- Ichihara H, Ueno J, Umebayashi M, Matsumoto Y, Ueoka R. Chemotherapy with hybrid liposomes for acute lymphatic leukemia leading to apoptosis in vivo. Int J Pharm. 2011;406(2):173-178.
- Ichihara H, Hino M, Umebayashi M, Matsumoto Y, Ueoka R. Intravenous injection of hybrid liposomes suppresses the liver metastases in xenograft mouse models of colorectal cancer in vivo. Eur J Med Chem. 2012;57(1):143-148.
- Ichihara H, Okumura M, Matsumoto Y, Tsujimura K. Theranostics with hybrid liposomes in an orthotopic graft model mice of breast cancer. Anticancer Res. 2018;38(9):5645-5654.
- Ichihara H, Nagami H, Kiyokawa T, Matsumoto Y, Ueoka R. Chemotherapy using hybrid liposomes along with induction of apoptosis. Anticancer Res. 2008;28(2):1187-1196 .
- Ueoka R, Matsumoto Y, Goto K, Ichihara H, Komizu Y. Membrane targeted chemotherapy with hybrid liposomes for tumor cells leading to apoptosis. Curr Pharm Des. 2011;17(17):1709-1719.
- Tominaga H, Ishiyama M, Ohseto F, Sasamoto K, Hamamoto T, Suzuki K, et al. A water-soluble tetrazolium salt useful for colorimetric cell viability assay. Anal Commun. 1999;36(12):47–50.
- Telford WG, Komoriya A, Packard BZ. Detection of localized caspase activity in early apoptotic cells by laser scanning cytometry. Cytometry. 2002;47(2):81-88.
- Allen TH, Hansen C, Martin F, Redemann C, Yau-Young A. Liposomes containing synthetic lipid derivatives of poly(ethylene glycol) show prolonged circulation half-lives in vivo. Biochim Biophys Acta. 1991;1066(1):29-36.
Citation: Kuwabara K, Ichihara H, Matsumoto Y (2020) Inhibitory Effect of Hybrid Liposomes on the Growth of NP2 Glioma Cells. J Carcinog Mutagen. 11:344. DOI: 10.35248/ 2157-2518.20.11.344
Copyright: © 2020 Matsumoto Y, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Sources of funding : This work was supported in part by a Grant-in-Aid for Science Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan (Nos. 17K01383 and 17K05944).


