Indexed In
- Open J Gate
- Genamics JournalSeek
- Academic Keys
- JournalTOCs
- ResearchBible
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Proquest Summons
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- MIAR
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
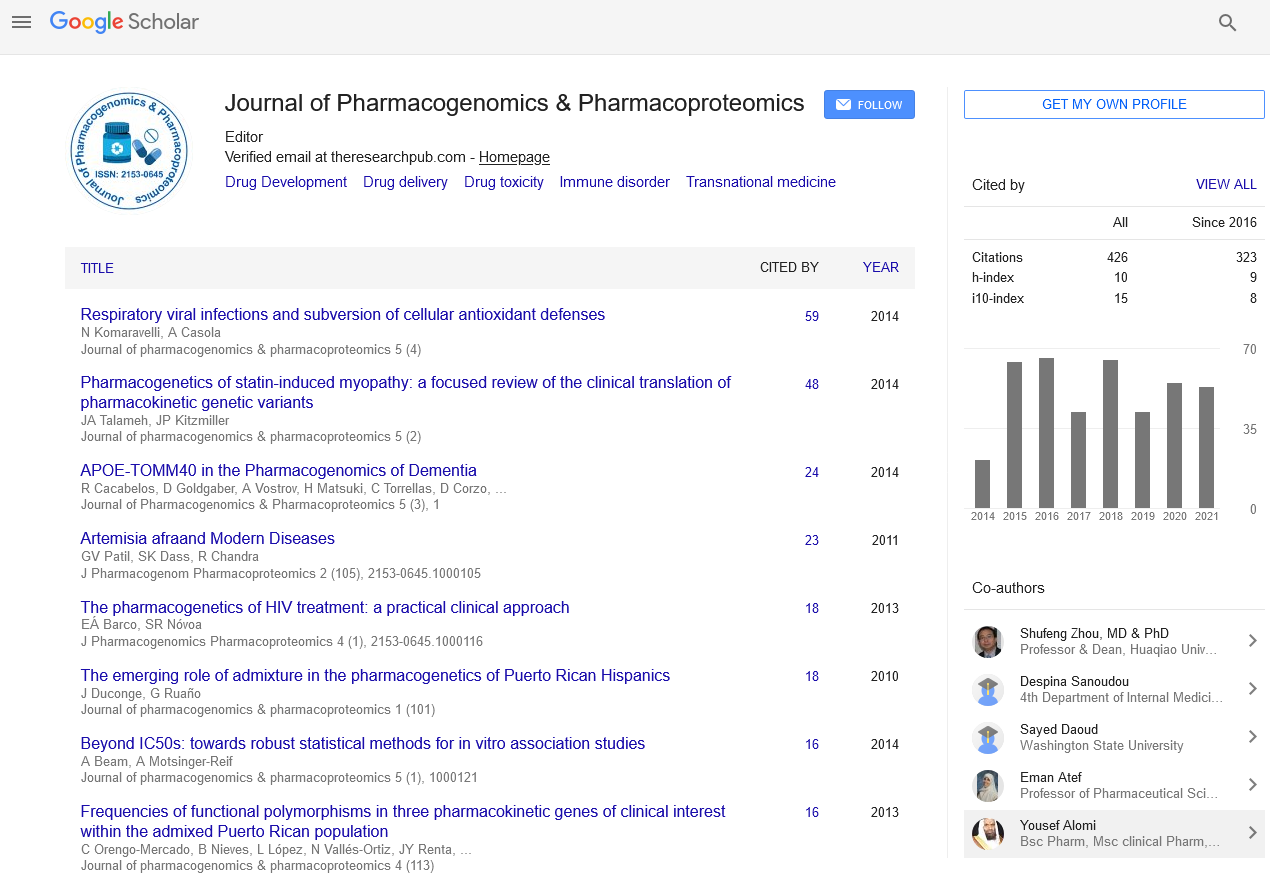
Abstract
Pharmacokinetics and Safety of Sublingual Flumazenil (CRLS035) in Healthy Adults (Potential Therapy for Hepatic Encephalopathy)
Saadi T, Kramskay R, Zilberman Peled B, Katz N, Peled N and Baruch Y
Flumazenil, a GABAA receptor antagonist, has a significant clinical benefit especially in overt hepatic encephalopathy patients, although it requires intravenous access. A novel highly concentrated sublingual spray formulation of flumazenil (CRLS035) was developed by Coeruleus Ltd. The aim of this study was to determine the single dose safety and pharmacokinetics of sublingual CRLS035 versus flumazenil intravenously (IV) in healthy volunteers.
Ten healthy adult volunteers participated in the study. CRLS035 was administered sublingually in two doses (1.1 mg and 2.2 mg) vs. IV flumazenil (0.2 mg). Subjects were evaluated after a high-fat diet and water consumption. Blood samples were collected pre- and post-dose at eight time points. Flumazenil levels were analyzed for Cmax, Tmax, Cmin, Tmin, AUC0-∞, AUC0-t and T1/2. Safety variables included local oral area and assessment of systemic adverse events.
The estimated bioavailability of the two sublingual doses was 14% and 11%, respectively. The bioequivalence of the 1.1 mg sublingual dose was similar to the 0.2 mg IV dose. Water consumption and the high-fat diet did not change the pharmacokinetic parameters significantly. No associated adverse events were reported across the study.
The pharmacokinetics of sublingual flumazenil is comparable to intravenous administration and the drug is safe. The sublingual approach allows convenient and better treatment availability for patients with hepatic encephalopathy.