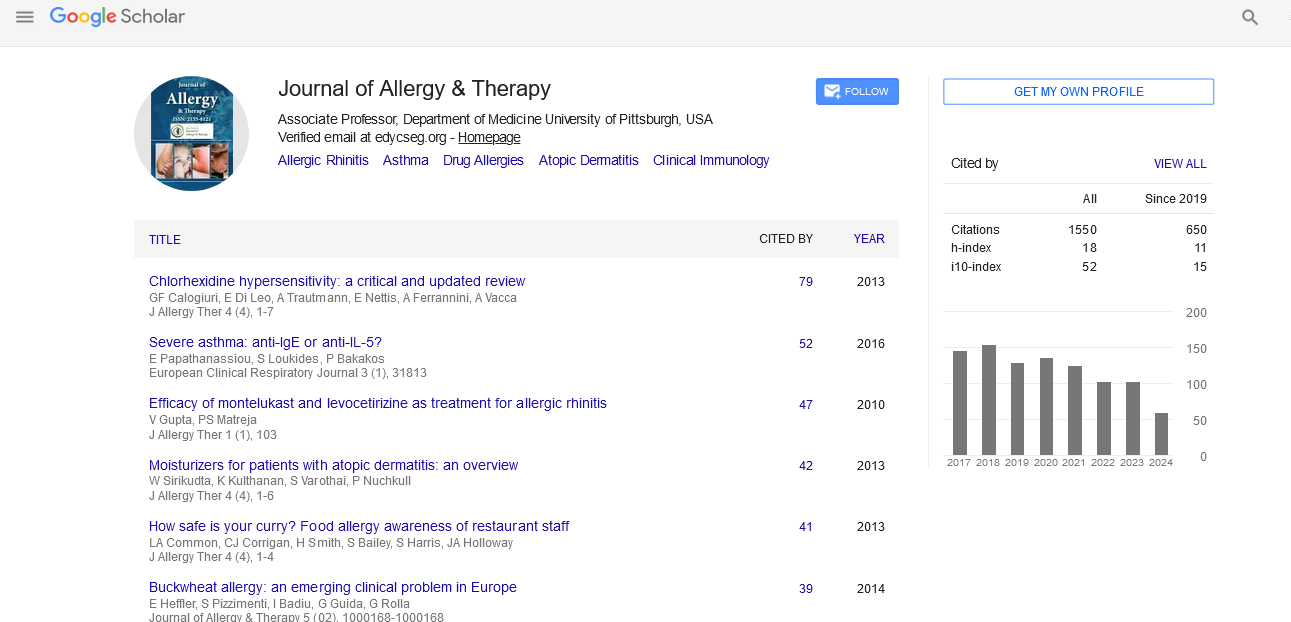
PMC/PubMed Indexed Articles
Indexed In
- Academic Journals Database
- Open J Gate
- Genamics JournalSeek
- Academic Keys
- JournalTOCs
- China National Knowledge Infrastructure (CNKI)
- Ulrich's Periodicals Directory
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Abstract
Clinical and Immunologic Effects of Allergen-Specific Sublingual Immunotherapy in a Canine Model of Atopic Dermatitis: A Double Blind, Randomized, Controlled Study
Marsella R and Ahrens K
Sublingual Allergen Specific Immunotherapy (SLIT) has been advocated for the treatment for allergies. Controlled studies are difficult to perform in humans due to the variety of allergies, diet, and allergenic exposure. This prospective, randomized, controlled study evaluated clinical and immunological effects of one year of SLIT using an experimental model of atopic dermatitis in dogs. Eighteen Beagles, sensitized to dust mites, timothy grass and ragweed were divided into control (n=6, vehicle) and active (n=12, 3 allergens) groups. Allergen challenge and scoring of clinical signs was done before and at the end of one year of SLIT. Blood was drawn at baseline, 4,8, and 12 months of SLIT and 2 months after stopping SLIT to measure allergen-specific IgE, IL-10, and TGF-beta.
After 12 months of SLIT, ANOVA showed significant decrease of clinical scores for both groups (p<.0001) but no significant differences between groups. T tests within each group comparing pre vs. post treatment scores showed statistically significant decrease in the control (p=0.042) and in the SLIT group (p=0.00027). Effect size using Cohen’s d was 1.182 for control and 2.1 for allergen group. Thus, decrease from baseline to post-treatment was nearly twice as large in the allergen as in the control group.
Mixed results were found for allergen-specific IgE with significant decrease for dust mites (p=0.0242) and increase for ragweed (p=0.0074) at the end of the study. SLIT induced significant increase of TGF-beta (p=0.03) and IL-10 (p=0.0009) after ragweed stimulation compared to baseline and to the control group. TGF-beta increase abated after SLIT discontinuation results consistent with SLIT induced T regulatory response. Interestingly a significant increase for IL-10 after timothy stimulation was seen for both groups at the end of study (p<.0001).It is concluded that this experimental model is useful to investigate treatments for atopic dermatitis and their immunologic effects.